Adocia 1Q25 – Phase 3 BioChaperone Lispro topline readout expected in mid-2025; priority to secure partnerships –
France-based biopharmaceutical company Adocia shared its 1Q25 results today (see press release). As background, Adocia is focused on developing innovative delivery solutions for peptide therapies (e.g., insulin, amylin, and GLP-1) that address current limitations, such as short-acting nature, need for injections, and fragility. The company has four proprietary platforms (see image below) and over half a dozen products under investigation for indications such as diabetes, obesity, and MASH. As indicated on Adocia’s website, the company has formed partnerships for some of these products and is seeking partners for the others.
Source: Adocia Feburary 2025 Investor Presentation
Financial positioning and cashflow
In 1Q25, Adocia ended the quarter with €12.2 million ($13.2 million USD), nearly doubling from the previous quarter at €7.5 million ($7.8 million USD). The increase was mainly from the €9.7 million ($10.5 million USD) private placement[1] for 2,125,000 new shares, completed in February 2025 – these were sold to Armistice Capital (€7 million), Vester Finance (€0.9 million), and Adocia cofounder Gerard Soula (€0.5 million), among others. With the upcoming $10 million in milestone payment from Tonghua Dongbao for completing the clinical portion of the phase 3 trial of BioChaperone Lispro in people with T2D, Adocia has secured funding until 2Q26.
Of note, the topline results for the phase 3 trial are expected in mid-2025, and Tonghua Dongbao plans to submit BioChaperone Lispro for regulatory review in China, pending results. Marketing authorization would prompt additional milestone payment of $20 million and double-digit royalties on sales to Adocia.
As with 4Q24, Adocia continues to prioritize establishing a partnership with Sanofi for M1Pram (fixed combination amylin/insulin) and identifying a partner for BioChaperone CagriSema. The company will also present new preclinical data at ADA 2025 on AdoShell Islets, designed to provide immunoprotection for cell therapy in people with diabetes; first-in-human trial submission planned for 2H25.
Adocia’s pipeline leverages proprietary platforms to innovate the delivery of existing drugs for diabetes, obesity, and MASH
Adocia’s pipeline consists of two clinical-stage and four preclinical products under investigation for diabetes, obesity (including obesity in people with diabetes), and MASH. The company also has two products in its pipeline not shown below: BioChaperone Combo (pre-mixed glargine/lispro) and BioChaperone LisPram (combination insulin lispro/pramlintide), both in phase 1 development.

- BioChaperone Lispro (ultra-rapid insulin lispro): BioChaperone Lispro is being investigated in collaboration with Tonghua Dongbao in two phase 3 studies for diabetes. In T1D study (n=509), the final patient was dosed in January 2025, and topline results are expected in mid-2025. In the T2D study (n=978), the last patient completed final dosing in December 2024, prompting $10 million in milestone payments from Tonghua Dongbao to Adocia. As background, Adocia licensed BioChaperone Lispro to Tonghua Dongbao in April 2018.
- M1Pram (pramlintide/insulin analog): M1Pram is a fixed dose combination of pramlintide and M1, a mealtime insulin analog, for people with obesity on intensive insulin therapy. Following the July 2023 agreement between Sanofi and Adocia, Sanofi has the exclusive right to negotiate a partnership for €10 million (10.8 million USD); conversations are ongoing to establish a global partnership.
- BioChaperone CagriSema (cagrilintide/semaglutide): BioChaperone CagriSema is a fixed combination of cagrilintide and semaglutide in preclinical development designed to optimize the manufacturing process (e.g., lower costs, increase capacity). As background, Novo Nordisk’s CagriSema requires cagrilintide and semaglutide to be housed in separate chambers in its single-use injection pen. One of Adocia’s priorities this year is to establish a partnership for BioChaperone CagriSema.
- BioChaperone Combo (pre-mixed insulin glargine/insulin lispro): BioChaperone Combo is a fixed combination of long-acting insulin glargine and rapid-acting insulin lispro. Adocia regained full rights for BioChaperone Combo after Tonghua Dongbao decided to discontinue this program in July 2024.
- BioChaperone LisPram (pramlintide/insulin lispro): BioChaperone LisPram is a co-formulation of pramlintide and insulin lispro in phase 1 development for people with diabetes and overweight or obesity. In contrast to M1Pram, BioChaperone LisPram is designed to be delivered via insulin pump. Sanofi has also the exclusive right to negotiate a global partnership on this product.
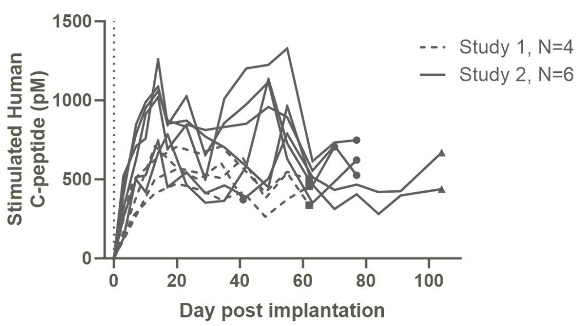
- AdoShell Islets: AdoShell Islets aims to enable islet transplantation in people with T1D without the need for immunosuppression. AdoShell Islets uses an immune-protective biomaterial containing endocrine cells, as Dr. Jay Skyler (University of Miami) highlighted at Keystone 2023. Adocia aims to complete IND-enabling studies in 2025. A first-in-human trial submission is planned for 2025. In preclinical data presented at ATTD 2025, in vivo studies in minipig showed that human C-peptide secretion was maintained for three months. Additional preclinical data will be presented at ADA 2025, IPITA World Congress, European Islet Study Group, and the International Society for Cell and Gene Therapy.
- AdOral GLP-1: AdOral GLP-1 is an oral delivery technology for peptides aimed at reducing injection burden and lowering manufacturing costs. It is in preclinical development on track for human studies in 2H25. Adocia has an R&D collaboration agreement for its AdOral technology; therefore, all costs related to this product’s development are covered by the partner.
- AdoGel GLP-1: AdoGel GLP-1 is a ready-to-use injectable hydrogel for long-acting drug delivery. It is in preclinical development for once-monthly or quarterly dosing of semaglutide. The AdoGel Sema has a controlled release mechanism characterized by nearly zero-order kinetics with no initial burst, ensuring steady release of the semaglutide into the bloodstream. This technology could potentially also enable quarterly injections.
Close Concerns’ Questions
- What are the company’s expectations for the upcoming phase 3 results for BioChaperone Lispro?
- Does Adocia hope to seek approval and commercialize BioChaperone Lispro in other countries?
- What is the amylin to GLP-1 RA ratio for BioChaperone CagriSema?
- What are ongoing discussions with Sanofi about the potential partnership for M1Pram and LisPram? What criteria is Sanofi seeking?
- Can the BioChaperone platform be used for combinations for three or more peptides (e.g., GLP-1 RA, GIP, and glucagon)?
- Why is BioChaperone LisPram intended to be delivered via insulin pump? Can it be administered as an injection as well?
--by Kat Moon, Monica Oxenreiter, and Kelly Close
[1] Sales of stocks or bonds to a small, pre-selected number of investors.