FDA approves Provention Bio’s teplizumab under brand name Tzield for the delay of clinical type 1 diabetes in people with stage 2 type 1 diabetes –
Executive Highlights
- The FDA approved the first disease-modifying therapy for type 1 diabetes, anti-CD3 teplizumab, under the cool brand name Tzield to delay the onset of clinical (stage 3) type 1 diabetes in people ≥8 years old with stage 2 type 1 diabetes (≥2 T1D autoantibodies with dysglycemia). Through a once-daily intravenous infusion for 14 consecutive days, Tzield delays the destruction of beta cells by inactivating killer T cells that attack pancreatic beta cells and increasing the proportion of regulatory T cells that facilitate the proper recognition of beta cells as self-cells. The FDA’s press release preceded Provention Bio’s press release by a few hours.
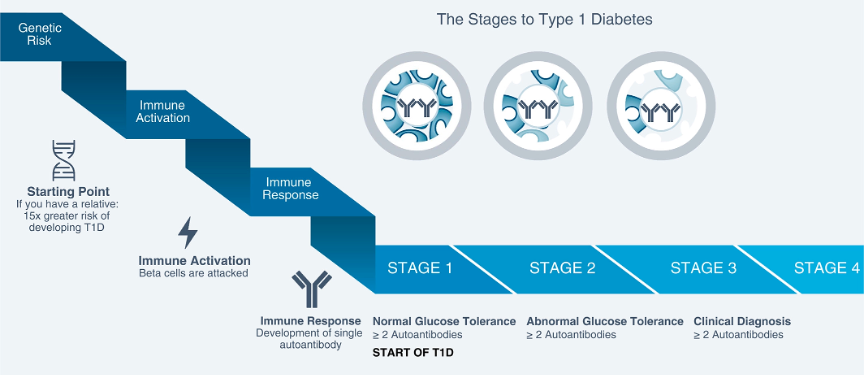
- For background, as of 2015, type 1 diabetes is classified into three stages (see appendix for a visual):
- Stage 1: ≥2 diabetes-related autoantibodies and normoglycemia. A 44% risk of progression to stage 3 in five years and lifetime risk approaches 100%
- Stage 2: ≥2 autoantibodies with dysglycemia but no symptoms. Dysglycemia is defined fasting plasma glucose ≥100 mg/dL and <125 mg/dL, two-hour plasma glucose ≥140 mg/dL and <200 mg/dL, or oral glucose tolerance test value ≥200 mg/dL. 75% risk of progression to stage 3 in five years
- Stage 3: a clinical diagnosis of type 1 diabetes (T1D) – this is, for laypeople, the “regular” T1D that has been known about forever
- For background, as of 2015, type 1 diabetes is classified into three stages (see appendix for a visual):
- Provention Bio will discuss Tzield’s approval in a conference call tomorrow, November 18 at 8 am ET – register here (it took us less than 20 seconds).
- The approval is based on results from the TN-10 (n=76) study, which showed a median two-year delay (three-year delay in a follow-up analysis) for a clinical T1D diagnosis in people with stage 2 T1D and a relative with T1D. The indication differs from the trial population in that Tzield’s use is not limited to people with a family history of type 1 diabetes – others who had autoantibodies could be included. See our detailed coverage of Tzield’s clinical data, benefits/risks, and experts discussion from the May 2021 FDA Advisory Committee meeting and the FDA’s briefing documents. In line with TN-10’s population, Provention’s initial target population for Tzield is the estimated 30,000 people in the US who have stage 2 type 1 diabetes and a relative with type 1 diabetes. From our view, this sounds like a very meaningful number, and we know, as always, it’ll all be about how well the earlier screening and diagnoses (of stage 2 T1D) goes.
- This is a momentous approval as Tzield is the first approved therapy to modify T1D progression and the first new therapy for the condition beyond insulin and Symlin (pramlintide). A two- to three-year delay in the diagnosis of clinical type 1 diabetes is certainly clinically meaningful for many people, which the FDA Advisory Committee members agreed with. This point is supported by Provention’s market research as well as dQ&A’s survey data (n=1,112) that was presented at FDA back in May, 2021 by analyst Jackie Tait.
- According to a survey by Provention, a huge group of surveyed endocrinologists (n=847) would prescribe Tzield to, on average, 93% of eligible patients. Among patients, caregivers, and at-risk individuals (n=200), 81% said they would take / would have taken Tzield with their HCP’s recommendation. How many endos have knowledge of any people with stage 2 T1D is a question, of course, as obviously is price, ability for people to take off time to receive the (ostensibly one-time) treatment, etc.
- dQ&A survey data from 1,112 patients (presented at the FDA AdComm) found that about half of respondents believed Tzield’s delay in diagnosis would be valuable, providing precious time to prepare a healthcare team, educate themselves, and emotionally mature. Notably, about a quarter of respondents also shared comments that raised other barriers, noting concerns about heightened levels of emotional stress or pushing T1D diagnosis to a more challenging time in their lives (e.g., in some cases, puberty, etc.). The pros/cons of Tzield clearly will require clear, informed discussions between families and clinicians, similar to discussions on autoantibody screening. Given that those surveyed were all PWD who had already been diagnosed, we imagine we might hear even more enthusiasm from siblings and family members who haven’t already been diagnosed and have an opportunity to delay the disease!
- No details were shared on pricing, but Provention sees a “significant price potential,” according to May 2022 investor event. Identifying the perceived clinical value of teplizumab on a five-point scale (1: no value, 5: major value), payers reported a perceived clinical value of 4.2, between “important” and “major” clinical value. Scores above four are only given to a few therapies, such as gene therapies. Notably, payers, prescribers, and KOLs said there is no price-point comparator for Tzield – we’d agree with this from what we know, especially since, as we understand it, it’s likely to be a one-time treatment, but there could be some who take it again. Notably, Provention has a patient support program and a co-pay assistance program to support access to Tzield – we will report more on this as we learn more.
- See more on immunotherapies being developed for type 1 diabetes in our immunotherapy competitive landscape. At Keystone 2022, JDRF’s CEO Dr. Aaron Kowalski said the future of type 1 diabetes disease-modifying therapies is “very, very promising,” highlighting a few promising therapies in clinical trials: anti-thymocyte globulin, verapamil, iscalimab, IMCY-0098, baricitinib, and ustekinumab.
In highly anticipated news, Provention Bio’s anti-CD3 antibody Tzield (teplizumab-mzwv) received FDA approval this afternoon to delay the onset of clinical (stage 3) type 1 diabetes in people ≥8 years of age with stage 2 type 1 diabetes (≥2 T1D autoantibodies with dysglycemia). This approval follows Tzield’s initial FDA submission in November 2020, a May 2021 FDA Advisory Committee meeting that voted 10:7 in favor of approval, a July 2021 Complete Response Letter, and a February 2022 Biologics License Application (BLA) resubmission.
Table of Contents []
- Tzield leads to an approximately three-year delay in clinical type 1 diabetes; safety warnings and precautions in line with immunotherapies
- Largest barrier to Tzield’s use is 14 days of infusion, but Provention aims to offer multiple infusion options
- Tzield’s commercialization and coverage
- Approval has the potential to catalyze autoantibody screening, thereby increasing eligible population for ongoing/future T1D clinical trials
- Provention has plans for several potential indication expansions for Tzield
- Tzield’s mechanism of action and a brief history of anti-CD3 antibodies in type 1 diabetes
- KOL commentary
- Teplizumab development timeline (June 2009 – present)
- Close Concerns Questions
- Appendix: Stages of Type 1 Diabetes
Tzield leads to an approximately three-year delay in clinical type 1 diabetes; safety warnings and precautions in line with immunotherapies
To assess Tzield’s efficacy, the May 2021 FDA Advisory Committee meeting considered two data sets: (i) the phase 2 TN-10 study (n=76) in relatives of people with type 1 diabetes who had stage 2 type 1 diabetes, and (ii) a post hoc meta-analysis of five other trials of Tzield in stage 3 type 1 diabetes: PROTÉGÉ, PROTÉGÉ ENCORE, STUDY 1, AbATE, and Delay.
The TN-10 study, summarized by the NEJM in this 110-second video, served as the “one adequate and well-controlled clinical investigation,” according to the Advisory Committee discussion. It enrolled people ages 8.5 to 49.5 years old and found that Tzield led to a median two-year delay in type 1 diabetes diagnosis compared to placebo. A follow-up analysis found that Tzield led to a median three year delay (33 months) in clinical type 1 diabetes diagnosis. Over a median follow-up of 51 months, Tzield led to a significant 69% reduced risk of a clinical type 1 diabetes diagnosis compared to placebo (HR: 0.41; 95% CI: 0.22-0.78; p=0.0066).
The Advisory Committee did not find the meta-analysis compelling as the meta-analysis population was distinct from the TN-10 study (all of whom had clinical type 1 diabetes), and it involved an unvalidated surrogate endpoint (C-peptide levels).
Per Provention’s press release, the most common side effects of Tzield in the TN-10 study were decreased levels of lymphocytes (73% on Tzield vs. 6% on placebo), rash (36% on Tzield and 0% on placebo), decreased levels of leukocytes (21% on Tzield vs. 0% on placebo), and headache (11% on Tzield vs. 6% on placebo). Tzield has the following warnings and precautions:
- Cytokine Release Syndrome (CRS) which occurs when immune system responds too aggressively to infection but can also occur with immunotherapy
- Risk of serious infections
- Decreased levels of lymphocytes
- Risk of hypersensitivity reactions
- The need to administer all age-appropriate vaccinations prior to starting Tzield
- Avoiding concurrent use of live, inactivated and mRNA vaccines with Tzield
Tzield’s clinical dataset and safety database includes over 800 people and more than 1,500 patient-years of follow-up. Importantly, Tzield’s most common adverse events are mechanism-based and expected for immunotherapies, and since Tzield is a one-time treatment the adverse events are transient.
See our highlights from the Advisory Committee meeting for discussions of TN-10’s small study size, strength of the study data, safety concerns and unknown long-term risks, and Tzield’s indication.
Largest barrier to Tzield’s use is 14 days of infusion, but Provention aims to offer multiple infusion options
At Provention’s May 2022 commercialization investor event, the company characterized the largest barrier to therapy use as the 14 days of consecutive infusion. To assist patients and families in managing this period of infusion, Provention will offer three infusion options:
- In an infusion center or out-patient hospital setting
- Partially in an infusion center/hospital and partially in-home
- Entirely in-home
Provention has partnered with two specialty pharmacy networks that have in-home infusion capabilities in all US states. The company anticipates that most patient starts will be done entirely in-hospital or in an infusion center since teplizumab is a new immunotherapy.
Tzield’s commercialization and coverage
Provention estimates that in the US 100,000 people have stage 1 type 1 diabetes and 200,000 people have stage 2 type 1 diabetes. Provention further estimates that 15% of people with stage two have direct relatives with type 1 diabetes, for a total of 30,000 people with stage two type 1 diabetes and a relative with type 1 diabetes. These 30,000 people are the initial target population for Tzield’s launch.
Tzield’s launch will be greatly supported by Provention and Sanofi’s October 2022 co-promotion agreement. Sanofi will co-promote Tzield, and Provention will contract Sanofi’s customer-facing field teams in the US. This agreement will expand the commercial footprint of Tzield, with more resources devoted to education, clinician outreach, and awareness. During Provention’s 3Q22 update, management shared that Sanofi’s outreach team will focus predominantly on adult endocrinology clinics, while Provention’s team will focus more heavily on pediatrics.
In its press announcement, Provention highlighted its COMPASS patient support program, which has staff dedicated to help navigate coverage, reimbursement, and access for Tzield. While specifics weren’t shared in the press announcement, Provention will offer copay assistance to eligible patients.
Provention’s expected payer mix is approximately 60% commercial, 35% Mediciad, and 5% other. Provention has >75 individuals payers engaged, representing over 270 million people. Provention’s field market access team expects Tzield to receive coverage in six to nine months post-approval.
Identifying at-risk individuals through autoantibody screening is one of the main challenges facing Tzield’s launch. As a result, over the past few years Provention has been dedicated to education efforts to increase autoantibody screening. The company has launched disease-awareness campaigns for patients and HCPs (Type 1 Tested and Connected by T1D), collaborated with a number of advocacy groups on screening education, and identified T1D immunotherapy centers of excellence.
Approval has the potential to catalyze autoantibody screening, thereby increasing eligible population for ongoing/future T1D clinical trials
Teplizumab’s approval has the potential to catalyze the use of autoantibody screening. As detailed in the Milken Institute’s and Helmsley Charitable Trust’s highly-regard 2021 report “Type 1 Diabetes Autoantibody Screening: A Roadmap for Pediatric Policy Implementation,” a key barrier to the uptake of autoantibody screening has been the lack of an approved therapy to modify type 1 diabetes progression. With Tzield’s approval and the availability of screening options, more HCPs may recommend screening, leading more people to get screened. Provention’s market research found that 76% of endocrinologists would increase screening for first degree relatives of people with type 1 diabetes following Tzield’s approval. Likewise, among 200 patients, caregivers, and at-risk individuals, 89% expressed a willingness to get screened for type 1 diabetes autoantibodies if recommended by their HCP. When only analyzing people with type 1 diabetes and caregivers, 96% would get screened or would have gotten screened if recommended by an HCP.
Reaching the 85% of people who develop type 1 diabetes without a family history of type 1 diabetes will require broad population screening, which has been implemented in a handful of local US programs and in the UK. There are also commercial options screening options: JDRF’s T1Detect program offers at-home tests through Enable Biosciences, and LabCorp, Quest Diagnostics, and Mayo Laboratories offer laboratory tests. The availability of screening options does not seem to be a major barrier, and Enable Biosciences offers a $10 screening option for those with financial need (typically $55/test). Still, insurance coverage for screening would likely boost the accessibility of screening, and we imagine the approval of Tzield may push payers to reconsider autoantibody screening.
One concern some have raised is whether the availability of Tzield would make trial recruitment for other type 1 diabetes therapies even more difficult. Just like in the TN-10 study, type 1 diabetes therapy trials have often faced challenges in recruiting patients, as JDRF’s CEO Dr. Aaron Kowalski explained at Keystone 2022. It’s possible that people who opt to receive Tzield may be disqualified from participating in trials for candidate type 1 diabetes disease modifying therapies, further limiting the pool for eligible participants. We believe that the approval of Tzield would increase autoantibody screening to such an extent that the population eligible for type 1 diabetes trials would increase more than the population of individuals who may be disqualified from such trials by receiving Tzield treatment.
Provention has plans for several potential indication expansions for Tzield
Looking ahead, Provention already has plans for Tzield indication expansions. Based on the ongoing phase 3 PROTECT study (exp. Completion May 2023), Provention may pursue a newly diagnosed type 1 diabetes indication. The company also plans to explore Tzield’s use in children ages two to eight years old, repeated dosing, subcutaneous administration, use with beta cell transplantation, and combination use with other therapies. In fact, JDRF, Yale University, and an early-stage immunotherapy company NexImmune are investigating combination use of a mouse version of teplizumab and NexImmune’s Artificial Immune Modulation nanoparticles.
Tzield’s mechanism of action and a brief history of anti-CD3 antibodies in type 1 diabetes
As a humanized anti-CD3 monoclonal antibody, Tzield binds to the CD3 receptor on T cells as a partial agonist and ultimately reduces autoimmunity. Specifically, Tzield temporarily inactivates self-reactive cytotoxic (or killer) T cells that are involved in the destruction of pancreatic beta cells, turning them into exhausted T cells, which are associated with slower type 1 diabetes progression. Additionally, Tzield enhances the function and proliferation of regulatory T cells, which suppress the action of cytotoxic T cells and facilitate the proper recognition of beta cells as self-cells. Through these two mechanisms, Tzield delays the destruction of beta cells and, ultimately, delays the progression of type 1 diabetes.
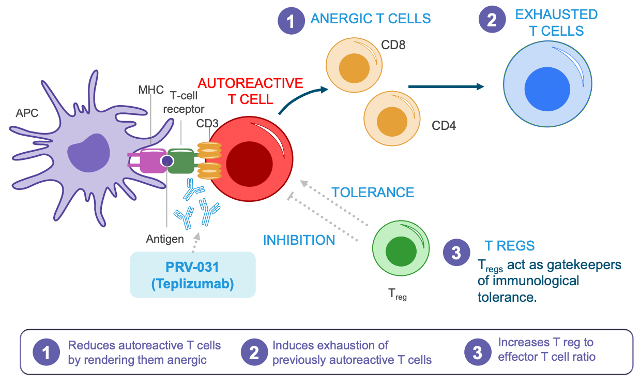
Anti-CD3 antibodies in type 1 diabetes became an attractive therapeutic option following the recognition of type 1 diabetes as an autoimmune disease in the second half of the 20th century, the first production of monoclonal antibodies in 1975, and the 1986 approval of the first monoclonal antibody for renal transplantation (anti-CD3 orthoclone OKT3). 1989 brought the first evidence that a single injection of an anti-CD3 antibody in early mouse development leads to a reduction in insulitis and insulin-dependent diabetes. Over the following decade, studies indicated that short-term anti-CD3 antibody treatment for type 1 diabetes seemed to specifically inactivate diabetogenic T cells, while leaving the rest of the immune response intact.
See more on immunotherapies being developed for type 1 diabetes in our immunotherapy competitive landscape, which we will be further updating over the coming weeks. At Keystone 2022, JDRF’s CEO Dr. Aaron Kowalski said the future of type 1 diabetes disease-modifying therapies is “very, very promising,” highlighting a few promising therapies in clinical trials: anti-thymocyte globulin, verapamil, iscalimab, IMCY-0098, baricitinib, and ustekinumab.
KOL commentary
- “This is a great day for all those having an interest in type 1 diabetes, from patients and their family members, researchers and care providers, and funding agencies, but perhaps most importantly, those at high risk for developing the disease. This is a testimony to the notion of persistence and how therapies are not developed or approved overnight. Today is a culmination of a 30-year effort to achieve this goal. From studies in animal models, drug development, numerous clinical trials, and then the regulatory process. It takes time.” – Dr. Mark Atkinson, University of Florida
- “The recent decision represents a turning point in the field. First, it identifies a way in which an immune therapy to stop the disease process might be combined with cell replacements in those with type 1 diabetes. It also suggests that it is time to more broadly screen to identify those at risk for type 1 diabetes, since now there is a therapy that can change its course.” – Dr. Kevan Herold, Yale University
- “Today’s FDA decision gives people at risk of developing type 1 diabetes the gift of time. For the first time ever, we have a way to change the course and slow the development of T1D. This is a huge win for the T1D community and the latest example of how JDRF’s research and advocacy contribute to improving lives today and tomorrow. I am tremendously proud of the role JDRF played in bringing this therapy to market, and we look forward to working with other stakeholders to ensure Tzield is accessible to those who need it.” – Dr. Aaron Kowalski, CEO of JDRF
- “Tzield is the first medication to delay the onset of T1D. Our high risk patients will now be given the option to receive treatment. My hope is that during the delay there are other therapies that are developed that will delay onset longer, and once day patients can say, “I was at very high risk for developing T1D and never did.” – Dr. Natalie Bellini, R&B Medical Group, NY
Teplizumab development timeline (June 2009 – present)
- June 2009: MacroGenics/Lilly announce enrollment completion of teplizumab’s PROTÉGÉ phase 2/3 study and initiation of phase 3 PROTÉGÉ ENCORE study
- October 21, 2010: MacroGenics/Lilly’s phase 3 trial for teplizumab’s failed to meet primary composite endpoint of daily insulin dosage and A1c levels in patients with recent-onset type 1 diabetes
- May 30, 2018: Provention acquires teplizumab from MacroGenics
- June 9, 2019: Results from phase 2 TN-10 study presented at ADA 2019 and published in NEJM with an editorial
- August 6, 2019: FDA grants Breakthrough Therapy Designation to teplizumab
- October 24, 2019: Teplizumab receives PRIME Designation by the European Medicines Agency
- April 16, 2020: Provention commenced rolling BLA submission for teplizumab
- June 15, 2020: Follow-up results for TN-10 study presented at ADA 2020; published in Science Translational Medicine in March 2021
- November 2, 2020: Provention completes rolling submission of teplizumab BLA
- January 4, 2021: FDA accepts teplizumab BLA under Priority Review
- April 9, 2021: FDA identified deficiencies in BLA for teplizumab and requested additional PK/PD data
- April 23, 2021: Provention and FDA had an informal meeting to discuss comparability between Provention’s intended AGC Biologics-manufactured product and Lilly’s historical product
- May 25, 2021: FDA release briefing documents for Advisory Committee meeting
- May 27, 2021: FDA Advisory Committee meeting voted 10:7 in favor of teplizumab approval to delay the onset of clinical type 1 diabetes in at-risk individuals
- July 2, 2021: FDA issued Complete Response Letter for teplizumab BLA
- July 12, 2021: Teplizumab receives Innovation Passport in the UK
- September 13, 2021: Topline PD data from PROTECT trial, completed FDA meeting about product quality and manufacturing, plans for FDA meeting about PK modelling
- October 1, 2021: Provention submitted briefing documents and requested Type A meeting with FDA
- November 18, 2021: Type A meeting with FDA about PK modelling
- November 22, 2021: Provention shares top-line PK data, plans to schedule Type B pre-BLA resubmission meeting
- January 27, 2022: Provention announces takeaways from Type B pre-BLA resubmission meeting, plans to resubmit BLA in 1Q22
- February 22, 2022: Provention resubmits BLA
- March 21, 2022: FDA accepts BLA resubmission, sets August 17, 2022 PDUFA date
- May 19, 2022: Provention hosts investor event on teplizumab launch
- June 30, 2022: FDA PDUFA extended to November 17, 2022
- October 6, 2022: Sanofi and Provention enter into co-promotion agreement
Close Concerns Questions
- What will Tzield’s insurance coverage look like? What is the range of potential co-pays? When will details on pricing be available? What does the impact of the ultimate price and the 20% or 10% likely co-pay in the US mean?
- Is insurance coverage expected to align with the Tzield’s label or align with TN-10’s patient population (i.e., only people with relatives with type 1 diabetes)?
- How may the approval of Tzield influence type 1 diabetes autoantibody screening recommendations? The ADA currently recommends T1D autoantibody screening in research setting says it “can be considered an option” for people who have a first-degree relative with T1D.
- To what degree is the very impressive JDRF T1Detect screening program likely to expand based on this approval?
Appendix: Stages of Type 1 Diabetes
--by Ashwin Chetty and Kelly Close