ObesityWeek® 2024 Full Report –
Executive Highlights
- ObesityWeek® 2024 was back in Texas again this year – this time in San Antonio, just steps from the Riverwalk! The energy was absolutely electric during the five days, and rooms were truly packed – GLP-1 has certainly made its mark on the field, and indeed, we’d also add that so many medicines for people with T1D and T2D used to be associated with weight gain (read: TZDs Actos and Avandia, various insulins) and now it’s been terrific to see more focus on weight neutrality and weight loss.
- During the presidential plenary, Dr. Jamy Ard (Wake Forest School of Medicine), President of The Obesity Society, delivered the opening remarks, celebrating this year’s theme: “Advancing Equitable Care – A Patient Journey.” Following his remarks, Dr. Sean Wharton (McMaster University, Ontario, Canada) moderated an impactful panel discussion on equity in obesity care. The panelists included Drs. Claudia Fox (University of Minnesota), Priya Sumithran (Monash University, Victoria, Australia), Leah Schumacher (Temple University, Philadelphia, PA), Ellen Schur (University of Washington), and patient advocate Ms. Sarah Le Brocq, who shared her experiences on various themes, including: (i) moving beyond past biases in obesity care; (ii) understanding the genetic contributions to obesity; and (iii) advancing equity in obesity care.
- Triple G alert! Dr. Chisom Kanu (Lilly) presented additional findings from Lilly’s phase 2 trial (n=338) for “triple G” retatrutide (GIP/GLP-1/glucagon RA) in people with obesity, focusing on health-related quality of life (HRQoL) and eating behavior. Retatrutide demonstrated generally positive effects in improving HRQoL and eating behavior, though not all factors measured were statistically significant between participants on retatrutide and placebo. On HRQoL, participants on retatrutide experienced significant improvements in the domains of physical functioning, general health, and vitality at 48 weeks. On eating behavior, participants on retatrutide saw significant dose-dependent improvements in the perceived hunger and disinhibition, but not in cognitive restraint. On HRQoL, participants on retatrutide experienced significant improvements in the domains of physical functioning, general health, and vitality at 48 weeks. On eating behavior, participants on retatrutide saw significant dose-dependent improvements in the perceived hunger and disinhibition, but not in cognitive restraint.
- New ICD-10 codes for obesity! In a very well-attended session moderated by the esteemed Dr. Donna Ryan (Pennington Biomedical Research Center), Drs. Alyson Goodman (CDC), Angela Golden (NP From Home, LLC), and Lydia Alexander (Enara Health) discussed the new ICD-10 codes for obesity, which officially took effect on October 1, 2024. Notably, the new codes EE66.811, EE66.812, and E66.813 correspond to the CDC’s BMI categories for Class 1, 2, and 3 obesity, respectively. These new ICD-10 codes for obesity were designed to improve diagnostic specificity and accuracy, facilitate better healthcare data collection and research, support appropriate reimbursement and insurance coverage, and reduce stigma.
- SURMOUNT-1 three-year outcomes were a major highlight from the week. Results for Lilly’s dual GIP/GLP-1 RA tirzepatide in prediabetes and obesity showed that tirzepatide confers up to 23% weight loss in people with prediabetes and obesity vs. 2% in placebo over 176 weeks. This corresponds to an astounding 24.6 kg (54.2 lbs) weight loss from a baseline of 107.4 kg (237 lbs). Nearly 95% of participants on tirzepatide reverted to normoglycemia vs. 60% in placebo. Notably, reversion to normoglycemia was similar across tirzepatide doses, highlighting tirzepatide’s potential weight loss-independent glycemic benefits.
- Dr. Daniel Drucker (University of Toronto), recipient of the TOPS Research Achievement Award, delivered an award presentation on the future of GLP-1 RAs and other incretin-based therapies. Dr. Drucker’s research on incretins has been instrumental to the development of GLP-1 RA therapies. He said highlighted that the story of GLP-1 RAs has been a 40-year one, dating back to the early 1980s with the discovery of the biologically active GLP-1 peptide sequence and its ability to stimulate glucose-dependent insulin secretion. His website glucagon.com has been very closely watched for decades. Over that time, highly effective therapies such as sitagliptin, (invented at Toronto’s Drucker Lab) semaglutide, and tirzepatide have been developed, and a “slew” of molecules are in development – 60 to 70, according to Dr. Drucker’s estimates. Looking ahead, Dr. Drucker said, “The next decade is going to be as or more exciting as the last 40 years,” as GLP-1 RAs become even more fine-tuned. For one, Dr. Drucker suggested that in 10 years, injections may no longer be necessary, especially with promising oral formulations in development. Companies are also exploring gene editing and gene therapy, which could potentially even change treatment from once-daily/once-weekly dosing to a “one-and-done” approach.
- Dr. Timothy Garvey (University of Alabama at Birmingham) presented full OASIS 4 results (n=307) for high dose once-daily oral semaglutide (25 mg) in people with obesity or overweight without diabetes. Oral semaglutide 25 mg significantly decreased body weight by 13.6% from a baseline of 105.9 kg (233 lbs) vs. 2.2% in placebo at 64 weeks. Categorically, 79% of participants achieved ≥5% weight loss vs. 31% in placebo. Notably, half of participants in the semaglutide achieved ≥15% weight loss vs. 6% in placebo, and 30% achieved ≥20% weight loss vs. 3% in placebo. A1c also significantly decreased by 0.29 percentage points vs. 0.06 in placebo (baseline of 5.7%). Additionally, 71% of participants who had prediabetes at baseline had reverted to normoglycemia, compared to 33% in placebo.
- Dr. Linong Ji (Peking University People’s Hospital, China) presented phase 2b study results of GLP-1 RA GZR18 in overweight or obesity. As background, GZR18 is being developed by China-based Gan & Lee, and its phase 1b/2a study showed that weight loss with once-weekly and bi-weekly administration of GZR18 was 17.8% and 12.8%, respectively, with 35 weeks of treatment. In this phase 2b trial, at 30 weeks, once-weekly GZR18 24 mg and bi-weekly GZR18 48 mg conferred weight loss of 1817.8% and 17.3%, respectively, vs. 1.0% in placebo. In the highest dose group (GZR18 48 mg), 9897.8% of participants lost ≥5% body weight, 82.2% lost ≥2010%, 60% lost ≥15%, and 3837.8% lost ≥20%.
- In a session dedicated to the SELECT trial for Novo Nordisk’s GLP-1 RA semaglutide in obesity, former head of TOS Dr. Robert Kushner (Northwestern University) presented an exploratory post hoc analysis showing that individuals treated with semaglutide were 11% less likely to experience hospitalization (p<0.0001) and 12% less likely to be hospitalized due to a severe adverse event (p<0.0001). Dr. Michael Lincoff (Cleveland Clinic) presented an analysis demonstrating that, in people with heart failure, semaglutide confers more pronounced (though not statistically significant) reductions in MACE, CV death, and hospitalization risk. Dr. Ildiko Lingvay (UT Southwestern) said highlighted that highlighted that semaglutide confers a 22% risk reduction in five-component kidney composite endpoint compared to placebo, driven primarily by a 20% decreased risk of persistent macroalbuminuria.
- Drs. Philip Schauer (Pennington Biomedical Research Center) and Beverly Tchang (Weill Cornell) squared off in a friendly debate on metabolic surgery vs. pharmacotherapy for managing obesity. While having a debate on this topic may insinuate imply that there is a divide between the two approaches, Drs. Schauer and Tchang both noted that in practice, there is significant alignment and cooperation between surgeons and endocrinologists. Both speakers presented convincing arguments. Dr. Schauer delivered a data-heavy presentation highlighting the durability and efficacy of metabolic surgery, demonstrated by long-term data from the oft-cited Swedish Obese Subjects study, as one example, for example. Meanwhile, Dr. Tchang outlined compelling points in favor of pharmacotherapy, such as the ability for it to directly target mechanisms driving obesity.
- Dr. Tina Thethi (AdventHealth) presented a post hoc analysis of the SURMOUNT-1 (n=1,775) and SURMOUNT-2 (n=609) studies assessing the association between early weight reduction and body weight and A1c reductions. On body weight management, early responders experienced a ~9% greater body weight reduction at Week 72 compared to non-early responders in both SURMOUNT-1 and SURMOUNT-2 studies (p<0.001). On A1c reduction, significantly more early responders achieved an A1c ≤5.7% (i.e., normoglycemia) compared to non-early responders among SURMOUNT-2 participants (~70% and ~41% of participants, respectively).
- In a joint symposium titled, “What's in a Name? Controversy Surrounding What to Call Obesity Medication,” panelists from The Obesity Society, European Association for the Study of Obesity, European Coalition for People living with Obesity, and Obesity Canada discussed the need to change the term “anti-obesity medications.” All panelists and audience members emphasized said that the term “anti” has stigmatizing implications, impacting both patients with obesity and broader public health outreach. Dr. Sanjeev Sockalingam, Scientific Director of representing Obesity Canada, noted that the field does not label treatments as “anti-obesity behavioral interventions” or “anti-obesity surgery; “therefore, medications should not be uniquely subjected to this stigmatizing language. Panelists suggested using the terms “obesity medications” or“medications for obesity.”
- During an especially compelling poster session, Dr. Joel Neutel (Orange County Research Center) presented new phase 1 data on Viking’s oral formulation of dual GIP/GLP-1 RA VK2735. As background, participants initially received oral VK2735 at doses ranging from 2.5 mg to 40 mg. Topline results reported in March 2024 revealed an encouraging safety and tolerability profile, as well as a 3.3% placebo-adjusted weight loss. Given these findings, Viking escalated dosage testing to 60 mg, 80 mg, and 100 mg. Results showed that VK2735 also demonstrated an encouraging safety and tolerability profile at these higher doses. Placebo-adjusted weight loss was as high as 6.8% (seen in the 100 mg dose group). Weight loss maintenance was also observed up to four weeks after treatment cessation.
- The exhibit hall simply buzzed with conversation during the welcome reception early in the meeting and throughout the rest of the gathering. There was a mix of larger booths, including Novo Nordisk, Lilly, and AstraZeneca, as well as smaller, more intimate setups. We were pleasantly surprised to see Dexcom here at ObesityWeek® for the first time (to our knowledge). Dexcom’s booth primarily featured its OTC CGM Stelo (launched in August for adults not on insulin therapy), and Dexcom’s G7 was also on show. This is significant for multiple reasons – most of all, in our view, to expand education and awareness about glycemic health and the point that it is addressable and changeable. Attendees were also attracted to the poster session in the exhibit hall. Many attendees were crowded around Viking’s poster on the phase 1 trial results for the oral formulation dual GLP-1 RA/GIP RA VK2735.
ObesityWeek® 2024 was yet another success, this time in San Antonio! Read on for our top highlights and see our Resource Hub!
Table of Contents []
-
Top Highlights
- Dr. Ard presents compelling opening remarks, and leads compelling panel with multiple celebrated clinicians and advocates
- New positive phase 1 data on oral formulation of Viking’s dual GIP/GLP-1 RA VK2735
- AstraZeneca moves three phase 1 compounds for obesity into phase 2
- New ICD-10 codes for obesity diagnostic codes signal a milestone for care, diagnosis, and reimbursement– all of which met with applause at Obesity Week 2024!
- Barriers to long-term anti-obesity medications use and insights from real-world evidence
- Real-world study (n=159) of GLP-1 RA discontinuation reveals access, not side effects, as primary reasons drop dropping coverage
- Dr. Cornelia Griggs discusses the underuse of metabolic and bariatric surgery in teens
- Dr. Daniel Drucker on the future of GLP-1 RAs: “The next decade is going to be as (or more) exciting as the last 40 years”
- SURMOUNT-1 three-year full results show tirzepatide confers 23% weight loss in people with prediabetes and obesity; ~95% of participants reverted to normoglycemia
- New SELECT analyses on the cardiovascular and renal benefits of semaglutide
- Dr. Louis Aronne shares clinical pearls on the balance between patient’s and provider’s target weights
- Additional analyses of phase 2 trial for Lilly’s retatrutide in obesity highlight improvements in quality of life and eating behaviors
- Additional analyses of phase 2 trial for Lilly’s retatrutide in obesity highlight improvements in quality of life and eating behaviors
- Early responders to tirzepatide in SURMOUNT-1 and SURMOUNT-2 trials experience greater body weight and A1c reductions than non-early responders
- Dr. Stephen Hursting on the potential role of tirzepatide in attenuating obesity-driven tumor progression – albeit with animal data
- Drs. Philip Schauer and Beverly Tchang square off in lively debate on metabolic surgery vs. anti-obesity medications
- OASIS 4 full results: Oral semaglutide 25 mg confers nearly 14% weight loss in people with obesity; 71% reverted from prediabetes to normoglycemia
- Novel GLP-1 RA GZR18 confers over 17% weight loss in phase 2b trial in Chinese people with obesity or overweight
- Consensus achieved on the need to drop “anti” from “anti-obesity medications” due to its associated stigma
Top Highlights
Dr. Ard presents compelling opening remarks, and leads compelling panel with multiple celebrated clinicians and advocates
Dr. Jamy Ard (Wake Forest School of Medicine), President of The Obesity Society, delivered the opening remarks for the presidential plenary, celebrating this year’s theme: “Advancing Equitable Care – A Patient Journey.”Following his remarks, Dr. Sean Wharton (McMaster University) moderated an impactful panel discussion on equity in obesity care. The panelists included Drs. Claudia Fox (University of Minnesota), Priya Sumithran (Monash University), Leah Schumacher (Temple University), Ellen Schur (University of Washington), and highly-regarded patient advocate Ms. Sarah Le Brocq, who shared her experiences on various themes, including: (i) moving beyond biases in obesity care; (ii) understanding the genetic contributions to obesity; and (iii) advancing equity in obesity care.
- On moving beyond past biases, Ms. Le Brocq said how, while she felt different due to her body shape since a young age, she did not initially view obesity as a major issue. Dr. Fox highlighted recent strides in treatment options for adolescents with obesity, including the anti-obesity medications liraglutide, semaglutide, and phentermine topiramate for children over 12 years old. While some parents accept these new treatments, others remain hesitant. Dr. Fox said that many parents still view obesity as a matter of willpower rather than as a disease with complex biological underpinnings. She noted that parents who can overcome this bias and are willing to address obesity as a disease will have more treatments available. Ms. Le Brocq agreed and emphasized the importance of shifting perspectives away from the outdated and overly simplistic idea of “eat less, move more.”
- On understanding the genetic contributions to obesity, Dr. Sumithran discussed how, despite research published in NEJM 13 years ago showing the long-term persistence of counterregulatory hormones to weight loss, public awareness of these factors has only recently gained traction. Dr. Sumithran said that understanding of the science behind obesity will help people understand that it is a chronic disease, and that it is expected for weight loss maintenance to be challenging.
- Ms. Le Brocq described how Dr. Kevin Hall’s viral publication on set point theory, “Obesity Energetics: Body Weight Regulation and the Effects of Diet Composition,” reshaped her understanding about obesity, helping her realize that her diagnosis was not her fault. Empowered by this knowledge, she pursued genetic testing, discovering she had the FTO gene, predisposing her to obesity, and the MC4 receptor, leading a potential propensity for certain food groups. This was a relieving reminder that her obesity was not her fault, though she expressed uncertainty around testing her daughter for these genetic factors, knowing positive results would not currently lead to any meaningful modifications in how she is raised.
- Dr. Schur added that over 1,000 genetic markers are linked to obesity and related comorbidities, though much remains to be understood about the function of those changes at a molecular level. Even without detailed counseling on these genetic factors, which Dr. Schur still said is imperative for the future, recognizing the genetic component can reassure patients.
- On advancing equitable obesity care, Dr. Schumacher said it must start with patient involvement in care decisions, especially as treatment options expand and experiences with obesity treatment become increasingly diverse. Educating patients and providers that obesity is a chronic disease is essential, she said, but progress requires action on multiple levels, including improving insurance coverage, addressing social determinants of health, and shifting public narratives to influence policy. Ms. Le Brocq said that while the newest therapies for obesity are technically approved and available through the NHS in the UK, funding and access pathways make it challenging, raising questions of whether stigma plays a role in the lack of access to medications for obesity in particular compared to other chronic diseases. Dr. Schur added that understanding the biology of obesity is only part of the picture; truly understanding individuals requires listening to their stories. Ms. Le Brocq concluded that putting patient voices at the forefront allows for more informed, impactful clinical guidelines. She advocated for a future where patients feel empowered to voice their needs in support and care, emphasizing that confidence in advocacy is key to equitable care.
New positive phase 1 data on oral formulation of Viking’s dual GIP/GLP-1 RA VK2735
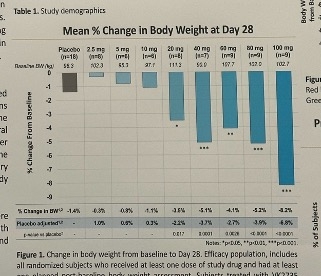
In the poster hall, Dr. Joel Neutel (Orange County Research Center) presented a poster with new phase 1 data (n=92) on Viking’s oral formulation of dual GIP/GLP-1 RA VK2735. As background, participants initially received oral VK2735 at doses ranging from 2.5 mg to 40 mg (quite a range!) for 28 days. Participants were medically healthy adults with BMI ≥30 kg/m2. Topline results reported in March 2024 revealed an encouraging safety and tolerability profile, as well as a 3.3% placebo-adjusted weight loss from a baseline of about 200 lbs. Given these findings, Viking escalated dosage testing to 60 mg, 80 mg, and 100 mg. Of note, this is the first-in-human trial for an oral dual GIP/GLP-1 RA therapy.
- Safety. Overall, VK2735 demonstrated a promising safety and tolerability profile, even at higher doses. The majority (99%) of adverse events were mild or moderate, and all GI-related adverse events were mild or moderate. Nausea was reported in both the treatment group (32%) and placebo group (11%) – all nausea was reported as mild, and we’d love to understand the difference between this nausea and what is typically reported as “mild to moderate.” Specifically, it was good to see that vomiting was reported in just three participants in the treatment group (3%) compared to zero in placebo.
- Weight loss. As shown below, placebo-adjusted weight loss was as high as 6.8% (seen in the 100 mg dose group). At 28 days, weight loss had not plateaued, suggesting that greater weight reduction may be possible. Weight loss maintenance was also observed up to four weeks after treatment cessation. The degree to which this may be additive or synergistic with other approaches is a question to watch.
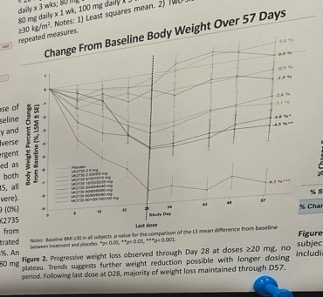
- As shared in 3Q24, Viking sees oral VK2735 as maintenance therapy following successful treatment with subcutaneous VK2735. Viking has also said that this oral formulation could serve as a potential “lead-in” to an injectable, particularly in a primary care setting. Viking plans to initiate a 13-week phase 2 trial in 4Q24.
AstraZeneca moves three phase 1 compounds for obesity into phase 2
AstraZeneca compounds saw encouraging results for AZD5004 (oral GLP-1 RA), AZD6234 (long-acting amylin), and AZD9550 (GLP-1/glucagon RA) for overweight/obesity and cardiometabolic health, all of which are moving forward into phase 2. Over 60% of people with obesity also suffer from one or more comorbidities, and AstraZeneca estimates that the company’s impressive pipeline of treatments could address nearly two-thirds of these conditions. Dr. Barr highlighted that AstraZeneca’s portfolio of therapies focuses on three key patient populations: (i) people living with obesity who may or may not have other severe co-morbidities; (ii) people who are overweight that require weight loss and organ protection; and (iii) people with T2D who may or may not be living with obesity/overweight.
- Phase 2 trials are next for AZD5004 (n=19, oral GLP-1 RA) following fantastic results for phase 1 results that support favorable safety, tolerability, and once-daily dosing profile. Data demonstrated that AZD5004 can be taken with or without food, unlike some other oral GLP-RA, which require fasting. Dr. Barr emphasized that, as an oral treatment, AZD5005 is also well-designed to be used in combination with other therapies. Data from the multiple ascending dose study showed a dose-dependent increase in adverse events (primarily nausea and vomiting) and a dose-dependent reduction in body weight across all treatment groups. On Day 28, data showed a robust reduction of fasting glucose across all treatment groups, with a fairly astounding mean reduction of 76.6 mg/dL from a baseline glucose of 189 mg/dL. Given these very promising results, AstraZeneca has two new phase 2b trials: (i) VISTA trial (n=304) for people with obesity/overweight and one comorbidity (started October 8); and (ii) SOLSTICE trial (n=384) for people with T2D. These trials are expected to complete in December 2025 and January 2026, respectively. We very much hope that with these kinds of glycemic results – a 40% drop in fasting glucose! – that continuous glucose monitoring will be used so that it is possible to see the glycemic impact for people with diabetes.
- Phase 2 studies also just began for AZD6234 (n=74, long-acting amylin) after phase 1 results provided encouraging data as a non-incretin-based therapy. These results demonstrated a favorable safety and tolerability profile and significant weight reduction at all doses studied. The pharmacokinetics also support once-weekly dosing with encouraging weight loss after a single dose. Furthermore, AZD6234 demonstrated quality weight loss, reducing fat mass while retaining lean mass in preclinical studies. The phase 2b APRICUS trial (n=231) is currently recruiting participants with obesity or overweight and the trial has two primary endpoints: (i) percent change in body weight at baseline vs. 26 weeks; and (ii) percent of participants achieving ≥5% weight loss. This trial is expected to complete in February 2026.
- A phase 2 fixed dose study trial of therapy is planned for AZD9550 (n=52, GLP-1/glucagon RA), following favorable phase 1 results shown at Obesity Week that support once-weekly dosing and providing confidence for combination therapy with AZD6234. Dr. Danielson discussed the potential for the combination of AZD9550 and ASD6234 to target three complementary mechanisms for weight loss via amylin and GLP-1/glucagon RA. Amylin targets fat-specific weight loss while preserving lean muscle mass, and GLP-1/glucagon RA confers end-organ protection. Dr. Barr emphasized the growing need for different management options and combination therapies to manage comorbidities, reduce the risk of complications, and tailor therapies to meet individualized goals and provide better health outcomes. While we look forward a phase 2b fixed dose combination study with AZD9550 and AZD6234, we will also be very interested to hear how the most frequently reported adverse events of nausea, vomiting, and headache emerge – while reduced appetite is another side effect seen, we are less concerned about satiety. It was very good to hear that the therapy demonstrated a favorable safety profile, but we will be most interested to know how tolerability emerges.
New ICD-10 codes for obesity diagnostic codes signal a milestone for care, diagnosis, and reimbursement– all of which met with applause at Obesity Week 2024!
In a well-attended session moderated by the esteemed Dr. Donna Ryan (Pennington Biomedical Research Center), Drs. Alyson Goodman (CDC), Angela Golden (NP From Home, LLC), and Lydia Alexander (Enara Health) discussed the new ICD-10 codes for obesity and how they may potentially impact patient care, diagnosis, and reimbursement. Dr. Ryan recognized the leadership and advocacy of Drs. Goodman and William Dietz (Director, STOP Obesity Alliance) in the creation of these new codes, which officially took effect on October 1, 2024. These codes were designed to address limitations in the old codes and reduce stigma in obesity-related healthcare.
- The old ICD-10 codes for obesity did not reflect the complexity of obesity or its different levels of severity.These codes also used stigmatizing and outdated language. For example, codes E66.0, E66.01, and E66.09[1] all imply that obesity is solely “due to excess calories.”
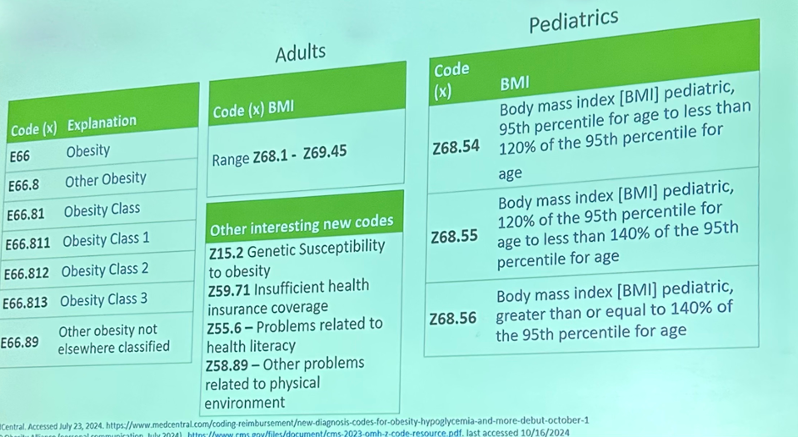
- The new ICD-10 codes for obesity in adults and children are aligned with the CDC’s obesity categories. As shown below, the table on the left summarizes the new codes for adults and children. Notably, codes EE66.811, EE66.812, and E66.813 correspond to the CDC’s BMI categories for Class 1, 2, and 3 obesity, respectively. Dr. Goodman aptly pointed out that the CDC is responsible for developing and modifying these codes, which may have been a motivating factor for the CDC to develop new codes that align with its BMI categories.
- There are also three new Z-codes[2] for children based on age- and gender-specific BMI percentiles. Z codes can be used in conjunction with E codes to provide more specificity in a diagnosis.
- The new ICD-10 codes for obesity were created to improve diagnostic accuracy, improve coding practice, enhance the usefulness of data for research, and reduce stigma. All three presenters highlighted how these codes improve the specificity and accuracy of diagnosis, facilitate better healthcare data collection and research, support appropriate reimbursement and insurance coverage, and reduce stigma as they reflect current, patient-centered approaches to obesity. On top of the benefits for patients, we were particularly excited to hear how the new codes will benefit data quality so that clinical researchers can better understand the effectiveness of interventions, such as GLP1 RAs, surgery, or lifestyle interventions. All three speakers emphasized the importance of raising awareness of these new codes by updating coding practices in health records and educating clinicians and patients on their utility.
Barriers to long-term anti-obesity medications use and insights from real-world evidence
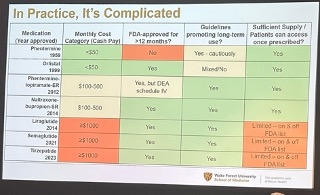
To a packed room, Dr. Kristina Lewis (Wake Forest School of Medicine) discussed the barriers to long-term anti-obesity medication (AOM) use and emphasized the importance of using real-world evidence to better understand AOM efficacy and safety. Dr. Lewis said that using AOMs long-term is complicated by factors beyond treatment efficacy. As shown below, one of these factors is the cost of medications, which can range from under $50/month for older medications such as phentermine to over $1,000/month for the latest incretin-based medications. Other factors include the availability of these medications (e.g., there have been global, intermittent shortages of semaglutide and tirzepatide in the past few years), as well as the drug indications themselves. For example, Dr. Lewis pointed out that phentermine is not FDA-approved for use beyond 3 months.
- Dr. Lewis emphasized the importance of using real-world evidence on AOM use to gain a more accurate understanding of safety signals, effectiveness, and barriers. She pointed out that the real-world setting differs significantly from clinical trials, which have highly selected patient populations, controlled treatment environments, and no access concerns (i.e., medications are free and available). For example, Dr. Lewis shared a study using real-world datafrom 2008-2017 from the VA’s MOVE! Program, revealing that of the nearly 500,000 participants in the program, only 1.6%, or 8,500 participants, were on an AOM. In addition, most participants (83%) were on orlistat, one of the older medications, highlighting that access to AOMs – particularly the latest, most efficacious ones – can vary based on healthcare coverage. In summary, Dr. Lewis said real-world evidence currently shows that AOM treatment durability is poor and that the amount of weight loss achieved with AOMs is less than amounts observed in clinical trials – though this difference is smaller among patients who are able to continue treatment for up to a year, and who are taking the medications for the indication of obesity, as opposed to diabetes.
Real-world study (n=159) of GLP-1 RA discontinuation reveals access, not side effects, as primary reasons drop dropping coverage
In a standing-room-only oral session, Dr. Jenna Napoleone (Omada Health) presented findings from a real-world survey study (n=159) on the demographics and characteristics of patients who recently discontinued GLP-1 RAs therapy. Study participants came from Omada Health’s digital health lifestyle programs for weight management or hypertension. Mean age was 46 years old, and the majority of participants were White (65%) and female (87%). All participants had used a GLP-1 RA for at least eight weeks and discontinued treatment within four weeks of the study. Novo Nordisk’s Wegovy (semaglutide) and Lilly’s Zepbound (tirzepatide) were the most common GLP-1 RAs participants reported using.
- GLP-1 RA treatment discontinuation was primarily due to access barriers, including high costs, loss of insurance coverage, and/or drug shortages. Specifically, 75% of participants reported discontinuing treatment due to access barriers. Dr. Napoleone said that even participants experiencing treatment side effects were often willing to stay on the medication to reach their weight loss goals.
Dr. Cornelia Griggs discusses the underuse of metabolic and bariatric surgery in teens
Dr. Cornelia Griggs (Mass General) discussed the underuse of metabolic and bariatric surgery (MBS) in pediatric obesity treatment, emphasizing that the “watchful waiting” approach is no longer appropriate. The efficacy of bariatric surgery in this population has been demonstrated in studies such as Teen-LABS, which showed a 26% reduction in body weight and a 100% reduction in diabetes medications use in teens. Dr. Griggs also pointed out that the American Academy of Pediatrics’ clinical practice guidelines recommend MBS for those aged 13 years or older with a BMI ≥120% of the 95th percentile for age and sex.
- Dr. Griggs highlighted new 10-year follow-up data from the Teen-LABS study, published in NEJM last week, which supports the long-term safety and efficacy of pediatric MBS. This study showed that patients who underwent sleeve gastrectomy and gastric bypass maintained ~20% and ~22% BMI reduction from baseline, respectively, after 10 years. For reference, the Teen-LABS study initially found a ~25% reduction in BMI six months after surgery. Dr. Griggs also noted the continued remission of coexisting conditions, including type 2 diabetes (T2D). At baseline, 60% of participants had T2D, which decreased to just over 50% after 10 years. Especially considering that pediatric MBS has a strong safety profile that is also consistent with that of adults, Dr. Griggs said MBS use in this population should be higher.
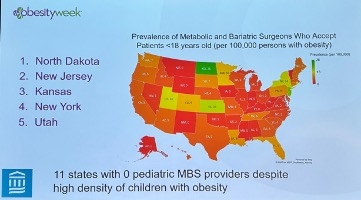
- Provider reluctance remains a notable barrier to greater adoption of MBS in pediatric care. Indeed, a recent study in the Journal of Surgical Research study revealed a significant mismatch between states with the highest prevalence of pediatric obesity and the those with MBS surgeons willing to accept pediatric patients. Alarmingly, Dr. Griggs said that, based on the American Society for Metabolic and Bariatric Surgery member registry, 11 states have do not have any MBS surgeons willing to treat pediatric patients, though she acknowledged that the registry may not be a completely accurate proxy. On using GLP-1 RA therapy as “adjunctive therapy” for those undergoing surgery, Dr. Griggs called for more research on this front and we wonder if long-term research on the ROI would be particularly helpful. Short-term, while surgery and GLP-1 may sound very expensive, long-term, the optimal way to approach those who do elect surgery may well be to use combination approaches. As usual, knowing in whom to use combination approaches is the question of the day - we look forward to more intelligence on this front at this meeting.
Dr. Daniel Drucker on the future of GLP-1 RAs: “The next decade is going to be as (or more) exciting as the last 40 years”
Dr. Dan Drucker (University of Toronto), recipient of the prestigious TOPS Research Achievement Award, delivered an award presentation on the outlook of GLP-1 RAs and other incretin-based therapies. Dr. Drucker’s research on GLP-1 was instrumental to the development of GLP-1 RA therapies. He highlighted that the story of GLP-1 RAs has been a 40-year one, dating back to the early 1980s with the discovery of the biologically active GLP-1 peptide sequence and its ability to stimulate glucose-dependent insulin secretion. For more reading on the history of GLP-1 RAs, we point you to Dr. Drucker’s article in NEJM from mid-September, from mid-September, titled “Discovery of GLP-1–Based Drugs for the Treatment of Obesity.” Since then, highly effective therapies such as sitagliptin and exenatide and semaglutide and tirzepatide have been developed, and a “slew” of molecules are in development – 60 to 70, according to Dr. Drucker’s estimates. As well, he said that around 10 different molecules originally discovered for muscle-related disorders (e.g., muscular dystrophy) are now being investigated to combat muscle mass loss seen in weight loss, which was a higher number than we imagine many expected.
Looking ahead, Dr. Drucker said, “The next decade is going to be as or more exciting as the last 40 years,” as GLP-1 RAs become even more fine-tuned. For one, Dr. Drucker suggested that in 10 years, injections may no longer be necessary, especially with promising oral formulations in development (e.g., Lilly’s GLP-1 RA orforglipron). While we don’t think people “mind” injections so much, it would certainly be easier and less costly to manufacture “true” oral therapies. Second, he said highlighted that he highlighted that companies are also exploring gene editing and gene therapy, which could potentially even change treatment from once-daily/once-weekly dosing to a “one-and-done” approach. Dr. Drucker’s presentation highlighted three important areas of discussion on GLP-1 RAs: (i) what mediates the pleiotropic benefits of GLP-1 RAs?; (ii) do GLP-1 RAs need to be taken for life?; and (iii) what’s next in GLP-1 RA research?
Challenging the intuitive notion that the benefits of GLP-1 RAs are solely weight-loss mediated: Dr. Drucker said that while weight loss is important, “at the end of the day, we are all trying to improve health,” whether that is reducing kidney disease progression, CVD risk, etc. GLP-1 RAs have demonstrated not only significant weight loss benefits, but also benefits in heart failure (STEP-HFpEF/STEP-HFpEF DM) and kidney disease (FLOW), with potentially more to come (e.g., semaglutide is also being investigated for peripheral artery disease [STRIDE]). While weight loss may mediate some of these observed cardio-renal benefits, Dr. Drucker highlighted evidence suggesting that these benefits are also partially weight-independent.
Dr. Drucker pointed out that in GSK’s Harmony Outcomes trial, GLP-1 RA albiglutide demonstrated a 22% relative risk reduction in MACE despite having only modest effects on A1c and body weight reduction. Dr. Drucker also mentioned Novo Nordisk’s ESSENCE trial for semaglutide in people with MASH – positive headline results were announced just days ago. He said that while these benefits may intuitively seem solely weight-loss mediated, lab evidence suggests otherwise. For example, in animal studies, if GLP-1 receptors in the brain are knocked out, effectively preventing weight loss, the liver still demonstrates improvement with treatment. Wow!
Challenging the “dogma” that GLP-1 RAs are a lifelong therapy: Much of the current thinking around incretin-based therapies is that patients need to take them for the rest of their lives to avoid weight regain. While this is true for some patients and has been shown in clinical trials, Dr. Drucker said pointed out that said pointed out that not everyone experiences weight regain. He referenced the SURMOUNT-4 trial, in which 46% of participants were able to maintain at least 10% weight loss and 26% were able to maintain 15% weight loss a year after stopping treatment. Dr. Drucker said understanding which patients can maintain weight loss without continuing treatment stands as an important research opportunity as patients may appreciate knowing that they may only need to take a medication for a finite period – we imagine if they know if they are “more or less likely” to be able to go “off” therapy would be valuable, even if it’s not a certainty.
Questions and opportunities for the future: Special populations, safety, and “fine-tuning” GLP-1 RAs. Dr. Drucker noted that there are still many areas for obesity and GLP-1 RA research. For example, there is a lack of data on the use of these therapies in special populations, such as kids. How obesity affects growth, development, and puberty in this population is also not well characterized. The safety of GLP-1 RAs can also be further reinforced. Dr. Drucker said noted that noted that in contrast to some past drugs that were withdrawn by the FDA due to safety concerns (e.g., Arena’s lorcaserin for increased cancer risk and Abbott’s sibutramine for elevated CV risk), GLP-1 RAs have shown to be very safe therapies. Dr. Drucker highlighted Novo Nordisk’s SELECT trial for semaglutide, pointing out that there were no risks that offset the observed benefits, such as the 20% relative risk reduction in MACE and 19% reduction in all-cause mortality. Dr. Drucker also acknowledged that GLP-1 RAs may not be for everyone, and a great area for future research is understanding who can tolerate these therapies and benefit most from them.
SURMOUNT-1 three-year full results show tirzepatide confers 23% weight loss in people with prediabetes and obesity; ~95% of participants reverted to normoglycemia
In a well-attended session, Drs. Leigh Perreault (University of Colorado), Louis Aronne (Weill Cornell), Ania Jastreboff (Yale), Sean Wharton (University of Toronto), and Carel le Roux (University College Dublin) presented the full SURMOUNT-1 three-year outcomes results for Lilly’s dual GIP/GLP-1 RA tirzepatide in obesity. These results are hot off the press and will results are hot off the press and will be published in NEJM next week. For those who are unfamiliar, SURMOUNT-1 (n=2,539) was a 72-week trial for tirzepatide in people with overweight or obesity, without diabetes. Results (presented at ADA 2022) showed that tirzepatide confers up to 23% weight loss vs. 2% in placebo; additionally, up to 96% of participants in the tirzepatide group achieved ≥5% weight loss vs. 28% in placebo – of note, all participants in the trial engaged in a reduced calorie diet and increased physical activity. At baseline, 41% of participants had prediabetes (n=1,032), and at 72 weeks, over 95% of these participants reverted to normoglycemia.
This three-year -outcomes three-year outcomes trial for SURMOUNT-1 only included the 41% of participants who had prediabetes at baseline. In other words, participants with normoglycemia at baseline were only studied up to 72 weeks, and those with prediabetes continued until three years. Topline results presented in August 2024 revealed that tirzepatide reduces the risk of T2D progression by a whopping 94% over 176 weeks. For comparison, semaglutide demonstrated a 73% reduced risk over 156 weeks in a pre-specified analysis of the SELECT trial.
Study design. Participants (n=1,032) were randomized to one of four groups: (i) tirzepatide 5 mg (n=247), tirzepatide 10 mg (n=262), tirzepatide 15 mg (n=253), or placebo (n=270). They were on treatment for 176 weeks, plus by a 17-week off treatment period, for a total of 193 weeks. The study was conducted in nine countries[3] across four continents; 45% of study participants were from the US.
The objectives of the 176-week period were to demonstrate that percent change in body weight with tirzepatide 10 mg and/or 15 mg is superior to placebo and that time to onset of T2D with tirzepatide (all doses) is superior to placebo. The objective of the 193-week period was also to demonstrate superiority in time to T2D onset for tirzepatide. Additional endpoints included reversion to normoglycemia and changes in cardiometabolic risk factors (e.g., blood pressure, lipids) and health-related quality of life.
Baseline characteristics. Mean age was 48 years old, and 64% of participants were female. By race, 73% of participants were White, 10% were Asian (a higher percentage than typically seen, which Dr. Aronne pointed out was due to study sites in multiple Asian countries), 8% were Black/African American, and 7% were American Indian/Alaska Native. By ethnicity, 47% were Hispanic or Latino, also reflective of there being study sites in multiple South American countries. Baseline weight, BMI, and A1c were 107 kg (236 lbs), 39 kg/m2, and 5.8%, respectively. The most common reason for study treatment discontinuation was “withdrawal by subject.” This was seen most in the placebo group (52%), which Dr. Aronne said usually occurs due to lack of efficacy.
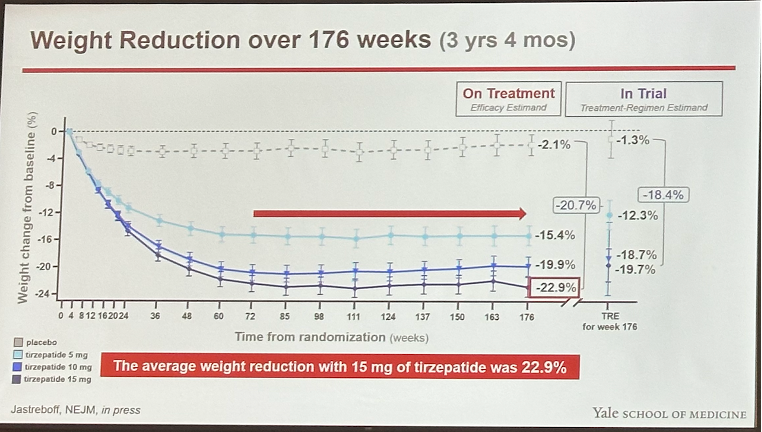
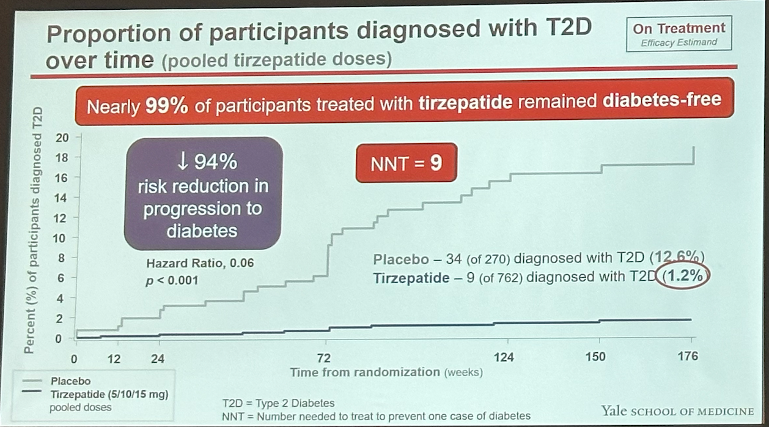
Results at 176 weeks. At 176 weeks, tirzepatide conferred up to 23% weight loss vs. 2% in placebo, similar to weight loss observed at 72 weeks. This corresponds to 24.6 kg (54.2 lbs) weight loss from a baseline of 107.4 kg (236.8 lbs). Impressively, in the highest dose group (tirzepatide 15 mg) 95% of participants lost ≥5% body weight, 79% lost ≥15%, and 43% lost ≥25%. On T2D progression, nine participants (1.2%) on tirzepatide progressed to T2D vs. 34 participants (13%) in placebo – a 94% risk reduction. In other words, 99% of participants treated with tirzepatide remained diabetes free. An analysis showed that 55% of this risk reduction in T2D progression was mediated by weight reduction.
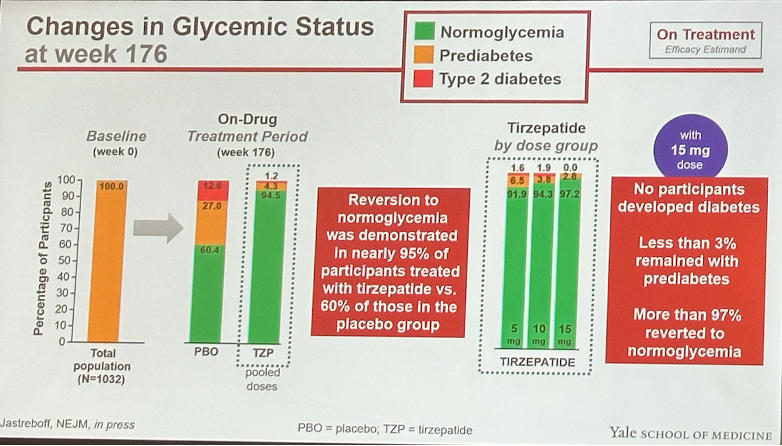
Changes in glycemia. At 176 weeks, nearly 95% of participants on tirzepatide reverted to normoglycemia vs. 60% in placebo. Notably, reversion to normoglycemia was similar across tirzepatide doses, highlighting tirzepatide’s potential weight loss-independent glycemic benefits. A1c decreased by up to 0.65 percentage points in the tirzepatide group vs. 0.14 percentage points in placebo from a baseline of 5.8%.
Changes in waist circumference, blood pressure, lipids. Waist circumference was reduced by up to 20 cm (8 inches) in the tirzepatide group vs. 3 cm (1 inch) in placebo from a baseline of 117 cm (46 inches). Reductions in systolic and diastolic blood pressure superior to placebo were not dose dependent. Tirzepatide also significantly increased HDL cholesterol by over 14% and reduced triglycerides by over 32%.
Changes in health-related quality of life. Participants on tirzepatide demonstrated significantly greater improvements in all eight domains of the SF-36 questionnaire, including physical functioning, bodily pain, social functioning, emotional role functioning, and mental health.
Results at 193 weeks (176 weeks + 17-week off drug period). At 193 weeks, participants saw an estimated average 7% weight gain. In the tirzepatide 15 mg group, participants maintained 18% weight loss (compared to 23% at treatment cessation at 176 weeks). Eight additional participants in the tirzepatide group progressed to T2D, bringing the total to 17. A1c also increased toward baseline levels – in the tirzepatide 15 mg group, A1c was 5.4% (compared to 5.1% at 176 weeks). Systolic and diastolic blood pressure also increased toward baseline levels.
Safety. GI-related adverse events were the most common adverse events and observed more in the tirzepatide groups than placebo. These were mostly mild to moderate in severity and occurred primarily during the dose escalation period. Discontinuation from the study drug due to adverse events were higher in the tirzepatide groups (up to 12%, or 31 participants) vs. placebo (6%, or 16 participants). GI-related adverse events were the most common reason for discontinuation – in the tirzepatide 15 mg group, nausea and diarrhea reported by six (2.4%) and four (1.6%) participants, respectively, led to treatment discontinuation.
New SELECT analyses on the cardiovascular and renal benefits of semaglutide
The famed Dr. Donna Ryan (Pennington Biomedical Research Center) underscored the importance of implementing learnings from the SELECT trial (n=17,604) into standards of care for cardiovascular disease management. She reminded the audience of the remarkable rigor, precision, and length of the inaugural trial, emphasizing very eloquently that its results, as we’ve heard from many others, legitimized obesity treatment as a pathway to disease modification. Dr. Ryan invited several other impressive speakers to present data to reinforce the seemingly never-ending positive effects of semaglutide.
Dr. Robert Kushner (Northwestern University), who is a past president of TOS, presented an exploratory post hoc analysis from the SELECT trial showing that individuals treated with semaglutide were 11% less likely to experience hospitalization (p<0.0001) and 12% less likely to be hospitalized due to a severe adverse event (p<0.0001). Dr. Kushner said that semaglutide-treated patients spent, on average, 19 fewer days hospitalized per 100 patient-years compared to placebo (157.2 days and 176.2 days, respectively; p=0.01). There were two fewer admissions per 100 patient-years than those on placebo (18.3 admissions and 20.4 admissions, respectively; p<0.001). However, he said noted that he noted that semaglutide did not significantly reduce the duration of each hospitalization (13 days with semaglutide vs. 13.2 days with placebo).
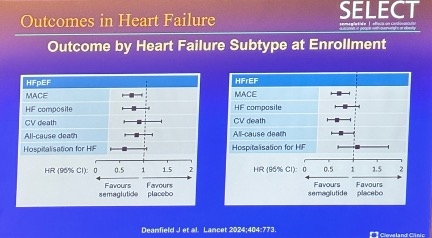
Dr. Michael Lincoff (Cleveland Clinic), also very well-known also very well-known in the field, reported that semaglutide reduced the risks of MACE, CV death, and hospitalization for people with a history of heart failure (HF). Dr. Lincoff said that of the 17,604 enrolled in SELECT, 4,286 had HF at baseline. While semaglutide reduced the overall risk of MACE, the reduction was more pronounced – although not significantly – in patients with HF, who had 28% risk reduction compared to 16% reduction in those without HF. Dr. Lincoff noted a similar, non-significant effect across HF subtypes: 35% MACE risk reduction in patients with HFrEF and 31% reduction in those with HFpEF. He said highlighted that He highlighted that participants with HFpEF taking semaglutide demonstrated a favorable risk profile for MACE, HF composite outcomes, CV death, all-cause death, and hospitalization. Specifically, he said only those with HFrEF taking semaglutide had a marginally increased risk of hospitalization for HF (see figure below).
Dr. Ildiko Lingvay (UT Southwestern) presented compelling findings showing that semaglutide confers a 22% risk reduction in five-component kidney endpoint[4] compared to placebo. The incidence of these endpoints was 1.8% for semaglutide-treated patients and 2.2% for those on placebo (p<0.05). Dr. Lingvay said that this reduction was primarily driven by a 20% decreased risk of persistent macroalbuminuria, with an incidence of 1.6% in the semaglutide group compared to 2% in the placebo group (p=0.05). There were no significant risk reductions observed in initiation of chronic kidney replacement therapy or in the onset of persistent eGFR <15 mL/min/1.73 min2. Dr. Lingvay noted a numerical, but not statistically significant, reduction in the risk of persistent ≥50% reduction in eGFR for those on semaglutide c0ompared to placebo.
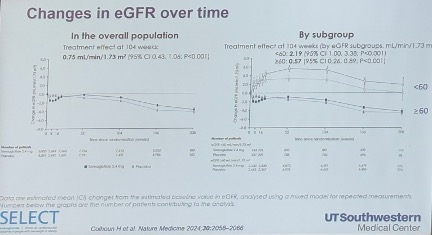
Dr. Lingvay also reported that the eGFR decline was less pronounced in the semaglutide arm than the placebo arm at 104 weeks. Specifically, eGFR declined by 0.86 mL/min/1.73 min2 in the semaglutide arm versus 1.61 mL/min/1.73 min2 in the placebo arm, reflecting a net benefit of 0.75 mL/min/1.73 min2 in the overall population. This treatment effect was particularly pronounced in patients with poorer baseline kidney function, which makes sense as it is “easier” to is “easier” to show benefit in this group. Dr. Lingvay said that among participants with baseline eGFR <60 mL/min/1.73 min2, those on semaglutide experienced an increase of 5.28 mL/min/1.73 min2, compared to an increase of just 3.09 mL/min/1.73 min2 in the placebo group, yielding a net advantage of 2.19 mL/min/1.73 min2 (p<0.001) in the “treated” group. By contrast, Dr. Lingvay said those with a baseline eGFR ≥60 mL/min/1.73 min2 had a smaller net improvement of 0.57 mL/min/1.73 min2 with semaglutide (p<0.001) – unsurprising since it is harder to show benefit in this group since they are doing better overall.
Dr. Lingvay said noted that Lingvay noted that semaglutide exhibited a similar adverse event profile across different baseline eGFR levels. Notably, patients taking semaglutide experienced a reduction in severe adverse events, regardless of baseline eGFR. This also makes sense, since having a better or worse starting point for kidney health is unlikely to be associated with more or less nausea or vomiting. She highlighted a significant reduction in adverse events leading to trial product discontinuation, primarily with gastrointestinal effects, independent of baseline eGFR (see figure below).

Dr. Louis Aronne shares clinical pearls on the balance between patient’s and provider’s target weights
Dr. Louis Aronne (Weill Cornell) discussed the nuances of optimal weight goals and the potential for discrepancies between healthcare providers and patients in the era of “miracle” weight loss drugs. Referencing the DREAM and OBSERVE studies, Dr. Aronne highlighted the sometimes stark differences between the weight goals set by patients versus those recommended by providers, often the result of varying prioritization of cosmetic desires and medical priorities. “This is a problem that needs more attention,” he noted, noting emphasizing that he noted, emphasizing that the increasing accessibility of effective weight-loss drugs has begun encouraging patients to pursue excessively low target weights. Instead, Dr. Aronne advocated for obesity treatment to target weight loss that specifically reduces obesity-related complications.
Dr. Aronne shared valuable “clinical pearls” for explaining optimal weight loss to patients in a manner that respects their personal priorities. He shared a figure (shown below) for clinicians to use when encouraging patients to maintain a stable weight rather than pursue further weight loss. The figure illustrates that the “lowest risk” curves for obesity-related complications (ORCs) typically occur within a BMI range of 22-25 kg/m2, after which risks increase again. On a personal note, he said he supports a BMI of 22 kg/m2 as the lower limit for patients with obesity, acknowledging that individual factors, including ethnic diversity in body composition, require case-by-case considerations.
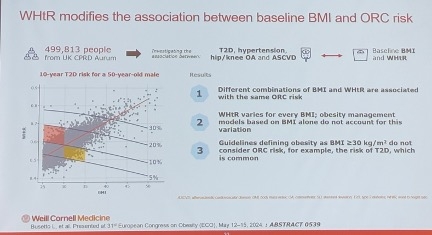
Dr. Aronne also presented data from ECO 2024 that established target values for BMI and the waist-to-height ratio (WHtR) to mitigate the risk of four ORCs (n=45,899). This prospective study aimed to evaluate the 10-year risk relationship between changes in BMI and WHtR due to unintentional weight loss and the risk of developing: (i) type 2 diabetes (T2D); (ii) hypertension; (iii) hip or knee osteoarthritis; and (iv) ASCVD. Outcomes were assessed over a 10-year period following a three-year baseline observation in the UK.
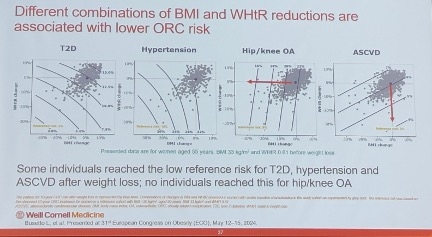
Dr. Aronne said explained that Aronne explained that different reductions in BMI and WHtR were associated with decreased risk for each of the ORCs. Reduced BMI was almost entirely associated with reduced risk for hip and knee osteoarthritis, while almost all the benefits for ASCVD were associated with reduced WHtR. Dr. Aronne said that reduced risk of T2D and hypertension was seen through variable combinations of reductions in both BMI and WHtR (see figure below). Based on these findings, the study identified a BMI target of 27 kg/m2 and a WHtR target of 0.53, which were shown to be good indicators of low 10-year incidence of all four obesity-related complications.
Additional analyses of phase 2 trial for Lilly’s retatrutide in obesity highlight improvements in quality of life and eating behaviors
Dr. Chisom Kanu (Lilly) presented additional findings from Lilly’s phase 2 trial (n=338) for “triple G” retatrutide (GIP/GLP-1/glucagon RA) in people with obesity, focusing on health-related quality of life (HRQoL) and eating behavior. This builds on the initial results presented at ADA 2023, which found that, at 48 weeks, retatrutide conferred up to 24% weight loss from a baseline of 237 lbs (108 kg) vs. 2% in placebo. Additionally, 100% of participants on retatrutide achieved ≥5% weight loss, 93% achieved ≥10% weight loss, 83% achieved ≥15% weight loss, 63% achieved ≥20% weight loss, 48% achieved ≥25% weight loss, and 26% achieved ≥30% weight loss.
In this current analysis, HRQoL was evaluated using the Short Form-36 Health Survey Version 2 (SF-36v2) form, which assesses physical and mental factors across eight domains (physical functioning, role-physical, bodily pain, general health, mental health, role-emotional, social functioning, and vitality). Eating behavior was evaluated using the Eating Inventory, which measures: (i) Cognitive Restraint of Eating; (ii) Perceived Hunger; and (iii) Disinhibition.
Results. Retatrutide demonstrated generally positive effects in improving HRQoL and eating behavior, though not all factors measured were statistically significant between participants on retatrutide and placebo.
HRQoL. Participants on retatrutide saw significantly greater improvements in HRQoL compared to placebo, as demonstrated by greater increases in SF-36v2 scores in certain domains. Specifically, in Week 24, participants on retatrutide had higher scores in: (i) physical domains combined; (ii) bodily pain domain; (iii) general health domain; (iv) vitality; and (v) role-emotional. In At Week 48, the physical functioning domain also became significant, in favor of retatrutide. Interestingly, at Week 48, the bodily pain and role-emotional domain scores were no longer significantly different between retatrutide and placebo.
HRQoL. Participants on retatrutide saw significantly greater improvements in HRQoL compared to placebo, as demonstrated by greater increases in SF-36v2 scores in certain domains. Specifically, at Week 24, participants on retatrutide had higher scores in: (i) physical domains combined; (ii) bodily pain domain; (iii) general health domain; (iv) vitality; and (v) role-emotional. At Week 48, the physical functioning domain also became significant, in favor of retatrutide. Interestingly, at Week 48, the bodily pain and role-emotional domain scores were no longer significantly different between retatrutide and placebo.
Eating behavior. Participants on retatrutide saw significant dose-dependent improvements in the Perceived Hunger and Disinhibition components of the Eating Inventory at both Week 24 and 48. However, no significant improvements were seen in the Cognitive Restraint component.
Prior analyses of this phase 2 trial for retatrutide in obesity: At ESC2024, a post hoc analysis found that retatrutide improves lipoprotein and apolipoprotein levels in people with overweight or obesity. Specifically, retatrutide reduced all atherogenic apoB and apoC-III particles and improved lipoprotein particle profile, including reductions in total and large triglyceride-rich lipoprotein particles (by up to 75%), total (24%) and small lipoprotein particles (32%), and total (8%) and small high-density lipid particles (13%).
Phase 3 plans for retatrutide in obesity: Retatrutide is now in phase 3 investigation for obesity in the TRIUMPH program:
TRIUMPH-1 (est. completion May 2026), investigating people with obesity/overweight, including subsets of people with knee osteoarthritis or obstructive sleep apnea;
TRIUMPH-2 (est. completion May 2026), investigating people with obesity/overweight and T2D;
TRIUMPH-3 (est. completion February 2026), investigating people with obesity/overweight and established CVD;
TRIUMPH-4 (est. completion March 2026), investigating people with obesity/overweight and knee osteoarthritis; and
TRIUMPH-OUTCOMES (est. completion February 2029), investigating cardiovascular and renal outcomes in people with obesity/overweight and ASCVD and/or CKD.
Additional analyses of phase 2 trial for Lilly’s retatrutide in obesity highlight improvements in quality of life and eating behaviors
Dr. Chisom Kanu (Lilly) presented additional findings from Lilly’s phase 2 trial (n=338) for “triple G” retatrutide (GIP/GLP-1/glucagon RA) in people with obesity, focusing on health-related quality of life (HRQoL) and eating behavior. This builds on the initial results presented at ADA 2023, which found that, at 48 weeks, retatrutide conferred up to 24% weight loss from a baseline of 237 lbs (108 kg) vs. 2% in placebo. Additionally, 100% of participants on retatrutide achieved ≥5% weight loss, 93% achieved ≥10% weight loss, 83% achieved ≥15% weight loss, 63% achieved ≥20% weight loss, 48% achieved ≥25% weight loss, and 26% achieved ≥30% weight loss.
In this current analysis, HRQoL was evaluated using the Short Form-36 Health Survey version 2 (SF-36v2) form, which assesses physical and mental factors across eight domains (physical functioning, role-physical, bodily pain, general health, mental health, role-emotional, social functioning, and vitality). Eating behavior was evaluated using the Eating Inventory, which measures: (i) Cognitive Restraint of Eating; (ii) Perceived Hunger; and (iii) Disinhibition.
Results. Retatrutide demonstrated generally positive effects in improving HRQoL and eating behavior, though not all factors measured were statistically significant between participants on retatrutide and placebo.
HRQoL. Participants on retatrutide saw significantly greater improvements in HRQoL compared to placebo, as demonstrated by greater increases in SF-36v2 scores in certain domains. Specifically, at Week 24, participants on retatrutide had higher scores in: (i) physical domains combined; (ii) bodily pain domain; (iii) general health domain; (iv) vitality; and (v) role-emotional. At Week 48, the physical functioning domain also became significant, in favor of retatrutide. Interestingly, at Week 48, the bodily pain and role-emotional domain scores were no longer significantly different between retatrutide and placebo.
Eating behavior. Participants on retatrutide saw significant dose-dependent improvements in the Perceived Hunger and Disinhibition components of the Eating Inventory at both Week 24 and 48. However, no significant improvements were seen in the Cognitive Restraint component.
Prior analyses of this phase 2 trial for retatrutide in obesity: At ESC2024, a post hoc analysis found that retatrutide improves lipoprotein and apolipoprotein levels in people with overweight or obesity. Specifically, retatrutide reduced all atherogenic apoB and apoC-III particles and improved lipoprotein particle profile, including reductions in total and large triglyceride-rich lipoprotein particles (by up to 75%), total (24%) and small lipoprotein particles (32%), and total (8%) and small high-density lipid particles (13%).
Phase 3 plans for retatrutide in obesity: Retatrutide is now in phase 3 investigation for obesity in the TRIUMPH program:
TRIUMPH-1 (est. completion May 2026), investigating people with obesity/overweight, including subsets of people with knee osteoarthritis or obstructive sleep apnea;
TRIUMPH-2 (est. completion May 2026), investigating people with obesity/overweight and T2D;
TRIUMPH-3 (est. completion February 2026), investigating people with obesity/overweight and established CVD;
TRIUMPH-4 (est. completion March 2026), investigating people with obesity/overweight and knee osteoarthritis; and
TRIUMPH-OUTCOMES (est. completion February 2029), investigating cardiovascular and renal outcomes in people with obesity/overweight and ASCVD and/or CKD.
Early responders to tirzepatide in SURMOUNT-1 and SURMOUNT-2 trials experience greater body weight and A1c reductions than non-early responders
Dr. Tina Thethi (AdventHealth) presented to a fully packed room a post hoc analysis of the SURMOUNT-1 (n=1,775) and SURMOUNT-2 (n=609) studies assessing the association between early weight reduction and body weight and A1c reductions. Participants in this pooled dataset were categorized as “early responders” or “non-early responders,” depending on their total body weight loss at Week 8 (≥5% and <5%, respectively).
On body weight loss,: Dr. Thethi said that early responders experienced a ~9% greater body weight reduction at Week 72 compared to non-early responders in both SURMOUNT-1 and SURMOUNT-2 studies (p<0.001). In SURMOUNT-1, weight reduction was ~23% and ~15%, respectively, and in SURMOUNT-2, weight reduction was 20% and ~11%, respectively. Significantly greater weight loss among early responders was seen beginning at Week four and extended through Week 72.
On dose-dependence,: Dr. Thethi said that, as expected, total body weight loss was dose dependent in both early and non-early responders in both SURMOUNT-1 and SURMOUNT-2. Interestingly, she noted that in the SURMOUNT-1 study there was no significant difference at Week 72 between early responders randomized to 10 mg and 15 mg tirzepatide (25.3% and 25.9% weight loss, respectively). Early responders in the study randomized to 5 mg tirzepatide, however, had a significantly lower ~19% reduction in body weight.
On A1c reduction,: Dr. Thethi said that early responders experienced a significantly greater A1c reduction compared to non-early responders. In SURMOUNT-1, early responders had a ~0.1% greater reduction in A1c (0.5% vs. ~0.4%, respectively), and in SURMOUNT-2, early responders had a ~0.4% greater reduction in A1c (~2.0% vs. ~2.5%, respectively). Furthermore, Dr. Thethi said that significantly more early responders achieved an A1c ≤5.7% (i.e., normoglycemia) compared to non-early responders among SURMOUNT-2 participants (, at ~70% and ~41% of participants, respectively).
Dr. Stephen Hursting on the potential role of tirzepatide in attenuating obesity-driven tumor progression – albeit with animal data
While we don’t typically cover pre-clinical data, we found UNC’s Dr. Stephen Hursting’s approach on (University of North Carolina at Chapel Hill) shared murine model data assessing the link between tirzepatide and reversal of obesity-driven cancers to be of interest. To start, Dr. Hursting explained said that in murine models, there is sufficient evidence from shared murine models that obesity promotes the development of certain cancers’ development and that, but calorie restriction can prevent the progression of these obesity-associated cancers’ progression. While he added that evidence is unclear whether intentional weight loss can reverse obesity’s pro-cancer effects, we are interested in finding more about this from researchers.
Dr. Hursting explained that in several obesity-driven cancer murine models, tirzepatide reversed or restored tumor mass to control size. Regarding In female mice with mammary tumors in female mice and diet-induced obesity, Dr. Hursting said that following inducement of mammary tumor growth, mice showed that with three weeks of tirzepatide treatment, these mice had demonstrated restoration of tumor size to that of the control mice (mice fed low calorie diets). (However, Dr. Hursting said that mice in the calorie restriction intervention also had a notable decrease reduction in tumor mass.) Following tirzepatide treatment, endometrial tumor masses in female mice and colon tumor masses in male mice, tirzepatide also were reduced tumor mass significantly more than control.
Notably, Dr. Hursting said that the size of this effect on colon tumor mass was matched in pair-fed mice (i.e., mice fed the same amount of food as the experimental group). He said this indicates that much of the effect of tirzepatide treatment on tumor cell mass reduction is due to its effect on appetite suppression and weight reduction, rather than due to a potential direct effect on the tumor cells. Dr. Hursting said that high dimensional flow cytometry has shown that tirzepatide has no effect on viability of cancer cells in colon or mammary tumor models, but he has seen early evidence that it could be involved in greater CD8+ T cell activation.
Drs. Philip Schauer and Beverly Tchang square off in lively debate on metabolic surgery vs. anti-obesity medications
Drs. Philip Schauer (Pennington Biomedical Research Center) and Beverly Tchang (Weill Cornell) captivated the audience with an engaging debate on metabolic surgery vs. pharmacotherapy for managing obesity. While having a debate on this topic may imply insinuate that there is a divide between the two approaches, Drs. Schauer and Tchang both noted that in practice, there is significant alignment and cooperation between surgeons and endocrinologists.
Close followers of this topic may remember that Dr. Schauer has debated this issue topic in two other conferences this year – at ENDO 2024 against University of Michigan’s Dr. Amy Rothberg and at the Rachmiel Levine-Arthur Riggs Diabetes Research Symposium 2024 against Dr. Donna Ryan, also from Pennington Biomedical. In contrast to those conferences, which had few or no surgeons in the audience, ObesityWeek had a significantly greater showing of surgeons – surgeons they made up about a third of the audience (based on a voluntary hand raise). We find this interesting to note as audience members predicted Dr. Schauer would win the debate (based on a poll), and the post-debate poll deemed Dr. Tchang as the “winner.” In contrast, at ENDO 2024, audience polling deemed metabolic surgery the overall winner (there was no poll at the other conference).
Drs. Philip Schauer (Pennington Biomedical Research Center) and Beverly Tchang (Weill Cornell) captivated the audience with an engaging and friendly debate on metabolic surgery vs. pharmacotherapy for managing obesity. While having a debate on this topic may insinuate that there is a divide between the two approaches, Drs. Schauer and Tchang both noted that in practice, there is significant alignment and cooperation between surgeons and endocrinologists.
Close followers of this topic may remember that Dr. Schauer has debated this topic in two other conferences this year – at ENDO 2024 against University of Michigan’s Dr. Amy Rothberg and at the Rachmiel Levine-Arthur Riggs Diabetes Research Symposium 2024 against Dr. Donna Ryan, also from Pennington Biomedical. In contrast those conferences, which had few or no surgeons in the audience, ObesityWeek , expectedly, had a significantly greater showing of surgeons – surgeons made up about a third of the audience (based on a voluntary hand raise). We find this interesting to note as audience members predicted Dr. Schauer would win the debate (based on a poll), and the post-debate poll deemed Dr. Tchang as the “winner.” In contrast, at ENDO 2024, audience polling deemed metabolic surgery the overall winner (there was no poll at the other conference).
Both speakers presented convincing arguments. Dr. Schauer delivered a data-heavy presentation highlighting the durability and efficacy of metabolic surgery, demonstrated by long-term data from the Swedish Obese Subjects study, for example. While pharmacotherapy has not had long-term data beyond four years (i.e., SELECT), Dr. Tchang, who , we note, really commanded the stage by delivering her presentation away from the podium, pointed out that the promising data available so far suggests that pharmacotherapy will be effective in the even longer-term as well.
We highlight below some key points from the debate:
Patient preferences. Dr. Tchang pointed out that patients do not want surgery, backing her argument with the graph below of Google search trends for the words “weight loss surgery,” “Mounjaro,” and “Ozempic.” We imagine this may be related to patient perceptions around the invasiveness or safety of surgical procedures.
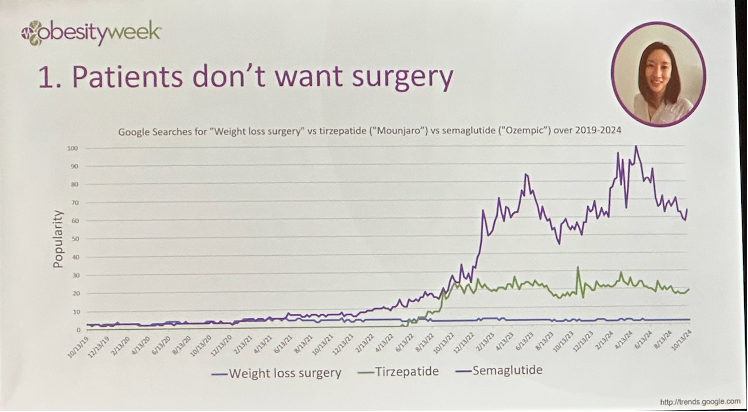
Efficacy. While clinical trials have shown that newer incretin therapies can confer weight loss close to metabolic surgery-levels, Dr. Schauer highlighted a recent real-world study from the Cleveland Clinic that showed only modest weight loss (5.1%) with semaglutide at one year. Additionally, 60% of patients stopped treatment within one year. He also pointed out that 27% of participants in the SELECT trial stopped treatment, suggesting indicating that tolerability may be a significant issue, which it certainly is for some. In contrast, he characterized metabolic surgery ais, effectively, a one-and-done treatment. This makes sense though the “bar” to be open to surgery is higher in our view.
Weight loss mechanism. Dr. Tchang pointed out that pharmacotherapy can target the mechanisms driving obesity, while with metabolic surgery, it is less clear mechanistically how metabolic surgery treats obesity beyond weight loss. Indeed, we have seen, even in the recently- presented SURMOUNT-1 three-year outcomes study at Obesity Week 2024, that pharmacotherapy has weight-loss independent benefits on cardio-renal-metabolic diseases.
Population health. Dr. Tchang highlighted that pharmacotherapy is ideal for population-level implementation given its scalability, pointing out that metabolic surgeons cannot likely perform more than ten surgeries a day – and even 10 sounds like a lot! She also pointed out that pharmacotherapy may one day be used for primary prevention of obesity, which is not possible with metabolic surgery. We certainly are interested in combination therapy using both.
Cost-effectiveness. Both speakers agreed that, currently, metabolic surgery is currently more cost effective than GLP-1 RAs, though with time that differential certainly shifts. Dr. Schauer noted that one-time cost associated with metabolic surgery would cost patients around $1,000-$1,500 per year if annualized over several years, compared to $11,000-$14,000 per year for GLP-1 RAs.
OASIS 4 full results: Oral semaglutide 25 mg confers nearly 14% weight loss in people with obesity; 71% reverted from prediabetes to normoglycemia
Dr. Timothy Garvey (University of Alabama at Birmingham) presented full OASIS 4 results (n=307) for high dose once-daily oral semaglutide (25 mg) in people with obesity or overweight without diabetes. As background, OASIS 1 evaluated the safety and efficacy of an even higher dose of oral semaglutide (50 mg), and results presented at ADA 2023 showed a body weight reduction of 15.1% vs 2.4% in placebo. OASIS 4 was designed to evaluate a lower dose of oral semaglutide (25 mg). While oral semaglutide requires more frequent administration than once-weekly injectable semaglutide, it serves as an additional option for patients who, for example, have a fear of injections or may be concerned about injection site reactions. Oral semaglutide is also being studied in people with obesity and overweight in East Asia (OASIS 2) and China (OASIS 3).
Trial design. Participants were randomized 2:1 to oral semaglutide 25 mg or placebo, in addition to lifestyle intervention (-500 kcal/day deficit and 150 min/week physical activity). Participants had obesity (BMI ≥30 kg/m2) or overweight (BMI ≥27 kg/m2) with at least one weight-related comorbidity (e.g., hypertension, dyslipidemia, obstructive sleep apnea, CVD). Dose escalation took place over 12 weeks, followed by a 52-week maintenance phase, for a total trial duration of 64 weeks. The co-primary endpoints were percent change in body weight and the proportion of participants achieving ≥5% weight loss. Secondary endpoints included change in IWQOL-Lite-CT Physical Function score, cardiometabolic risk factors, and glycemic status. On treatment completion, 82% of semaglutide-treated participants (n=167/205) and 75% of placebo-treated participants (n=76/102) completed the study. Trial sites were located in Canada, Germany, Poland, and the US.
Baseline characteristics. Mean age was 48 years old, BMI was 38 kg/m2, and A1c was 5.7% (47% of participants had prediabetes). On sex and race, 79% were female, and the majority (92%) of participants were White. Black, Asian, and “Other” participants made up 7%, 1%, and 1% of the trial population, respectively.
Results. As shown below, oral semaglutide 25 mg significantly decreased body weight by 13.6% from a baseline of 105.9 kg (233 lbs) vs. 2.2% in placebo. Categorically, 79% of participants achieved ≥5% weight loss vs. 31% in placebo. Notably, half of participants in the semaglutide achieved ≥15% weight loss vs. 6% in placebo, and 30% achieved ≥20% weight loss vs. 3% in placebo.
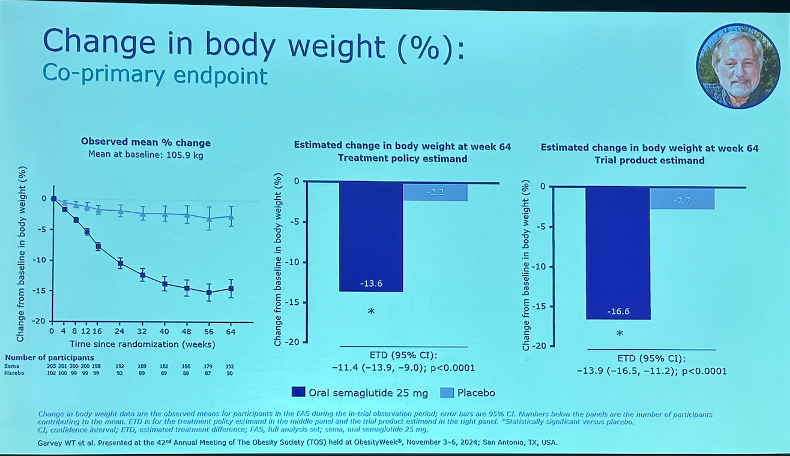
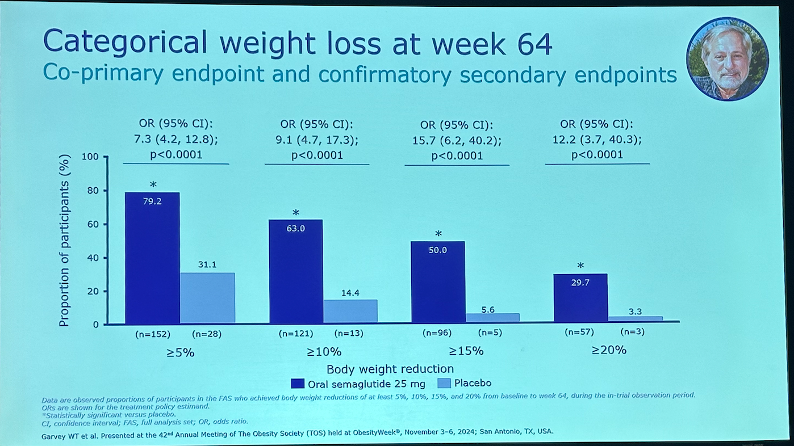
Secondary endpoints. IWQOL-Lite-CT Physical Function score[5] significantly increased by 16.2 points vs. 8.4 points in placebo. An increase of ≥14.6 points is considered clinically meaningful – 55% of semaglutide-treated participants met this threshold vs. 35% in placebo (p=0.0022). Waist circumference significantly decreased by 12.2 cm from a baseline of 113.9 cm vs. 3 cm in placebo. C-reactive protein, a biomarker for inflammation, also decreased significantly by 46% vs. 4% in placebo from a baseline of 3.7 mg/L. Decreases in systolic and diastolic blood pressure were greater in the semaglutide group but not statistically significant. Fasting plasma glucose significantly decreased by 6.6 mg/dL vs. 0.4 mg/dL in placebo from a baseline of 95.5 mg/dL, and A1c also significantly decreased by 0.29 percentage points vs. 0.06 in placebo (baseline of 5.7%). At Week 64, 71% of participants who had prediabetes at baseline had reverted to normoglycemia, compared to 33% in placebo.
Safety. The safety profile of oral semaglutide 25 mg was consistent with that of semaglutide and other GLP-1 RAs. About three-fourths (74%) of participants in the semaglutide-treated group experienced a GI-related adverse event – primarily nausea, vomiting, and constipation – compared to 42% in placebo.
Comparing OASIS 4, OASIS 1, and STEP 1. As shown below, weight loss in OASIS 4 was comparable to OASIS 1 (oral semaglutide 50 mg) and STEP 1 (injectable semaglutide 2.4 mg). The safety profile was also similar across all three trials, though Dr. Garvey highlighted that mean duration of nausea was shorter in OASIS 1 – 13 days for semaglutide 25 mg vs. 19 days for semaglutide 50 mg.. See our table below for further comparison of the three trials.
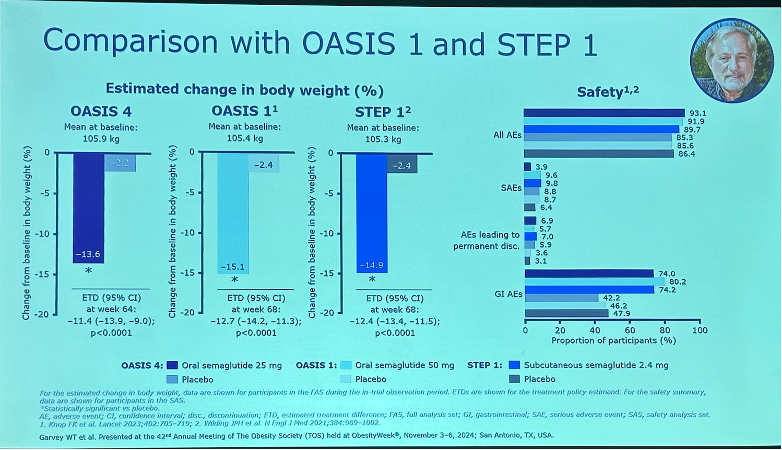
OASIS 4 (oral semaglutide 25 mg) Week 64 | OASIS 1 (oral semaglutide 50 mg) Week 68 | STEP 1 (injectable semaglutide 2.4 mg) Week 68 | |
| Body weight reduction* | 11.4% | 12.7% | 12.4% |
| Proportion achieving ≥5% weight loss | 79% | 85% | 86% |
| Proportion achieving ≥10% weight loss | 63% | 69% | 69% |
| Proportion achieving ≥15% weight loss | 50% | 54% | 51% |
| Proportion achieving ≥20% weight loss | 30% | 34% | 32% |
| Decrease in waist circumference* | 9.5 cm | 10.0 cm | 9.4 cm |
| Decrease in A1c* | 0.23 percentage points | 0.3 percentage points | 0.29 percentage points |
| *Values shown are the estimated treatment difference (ETD) in the treatment policy estimand. | |||
Novel GLP-1 RA GZR18 confers over 17% weight loss in phase 2b trial in Chinese people with obesity or overweight
Dr. Linong Ji (Peking University People’s Hospital, China) presented phase 2b study results of GLP-1 RA GZR18 in overweight or obesity. GZR18 is being developed by China-based Gan & Lee, and results from its phase 1b study showed that weight loss with once-weekly and bi-weekly administration of GZR18 was 17.8% and 12.8%, respectively, with 35 weeks of treatment. Dr. Ji said that the phase 2b results presented today indicate promise for an upcoming phase 3 study of the novel GLP-1 RA.
Study design. Participants (n=340) were randomized to one of six groups: (i) GZR18 12 mg bi-weekly (n=52); (ii) GZR18 18 mg bi-weekly (n=53); (iii) GZR18 24 mg bi-weekly (n=52); (iv) GZR18 48 mg bi-weekly (n=64); (v) GZR18 24 mg once-weekly (n=53); or (vi) placebo (n=66). Participants were on treatment for 30 weeks. The study was conducted among Chinese adults with a BMI ≥28 kg/m2 or BMI of ≥24 kg/m2 with ≥one obesity-related comorbidity.
Baseline characteristics. Mean age was 33 years old in both the bi-weekly GZR18 48 mg and once-weekly GZR18 24 mg cohorts, and 35% and 23% of participants were female, respectively. Baseline weight and BMI were 95.6 kg (209 210.8 lbs) and 33 kg/m2, respectively. Baseline A1c was 5.5% in the bi-weekly GZR18 48 mg cohort and 5.7% in the once-weekly GZR18 24 mg cohort.
Results at 176 30 weeks. At 30 weeks, once-weekly GZR18 24 mg and bi-weekly GZR18 48 mg conferred significant weight loss of 1817.8% and 17.3%, respectively, vs. 1.0% in placebo. These correspond to 16.7 kg (36.8 lbs) from a baseline of 94.5 95.0kg (208.3209.4 lbs) and 16.5 kg (36.4 lbs) from a baseline of 94.996.4 kg (209.2212.5 lbs), respectively. Impressively, in the highest dose group (GZR18 48 mg) 9897.8% of participants lost ≥5% body weight, 82.2% lost ≥2010%, 60% lost ≥15%, and 3837.8% lost ≥20%.
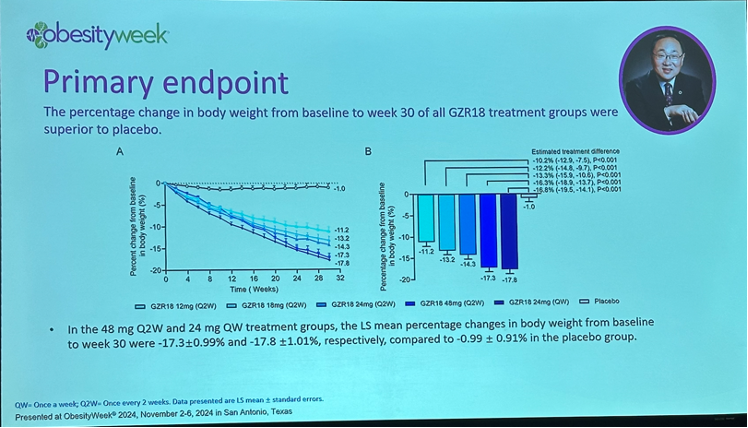
Changes in glycemia. At 30 weeks, bi-weekly GZR18 48 mg and once-weekly GZR18 24 mg both conferred a ~0.5% decrease in A1c from a baseline of 5.5% and 5.7%, respectively. Participants on placebo experienced a ~0.1% increase in A1c from baseline. Bi-weekly GZR18 48 mg also conferred a 0.4 mmol/L (7 mg/dL) reduction in fasting blood glucose at 30 weeks.
Changes in waist and hip circumference. At 30 weeks, bi-weekly GZR18 48 mg conferred a 14.2 cm (5.6 in) reduction in waist circumference from a baseline of 105.5 cm (41.5 in). Once-weekly GZR18 24 mg conferred a 13.7 cm (5.4 in) reduction in waist circumference from a baseline of 106.3 cm (41.9 in).
Changes in blood pressure and lipids. Reductions in systolic and diastolic blood pressure were superior to placebo and not specifically dose dependent, although bi-weekly GZR18 48 mg conferred the greatest reductions of 10 mmHg and 8 mmHg, respectively. Bi-weekly GZR18 48 mg also significantly reduced triglycerides by 4433.3%.
Safety. 16% (n=54) of participants discontinued the study, a slight majority of which was attributed to adverse events. The most reported adverse events were GI reactions, occurring in 9190.6% of participants receiving bi-weekly GZR18 48 mg and 83.0% of participants receiving one-weekly GZR18 24 mg. A moderate number of participants also reported injection site reaction, occurring in 44% of those receiving bi-weekly GZR18 48 mg and 51% of those receiving one-weekly GZR18 24 mg.
Consensus achieved on the need to drop “anti” from “anti-obesity medications” due to its associated stigma
In an early morning joint symposium titled, “What's in a Name? Controversy Surrounding What to Call Obesity Medication,” Participants panelists from The Obesity Society, European Association for the Study of Obesity, European Coalition for People living with Obesity, and Obesity Canada discussed in this impassioned panel discussion reached consensus on the need to change the term “anti-obesity medications.” The panel featured representatives from various organization, as well as firm agreement from the Korean Society for the Study of Obesity, represented by Dr. Cheol-Young Park. All panelists and audience members emphasized that the term “anti” has stigmatizing implications, that impacting both patients with obesity and broader public health outreach.
Dr. Marc-Andre Cornier of The Obesity Society suggested using “obesity medications,” but said that a clearer definition of obesity as a disease is needed. He opposed “weight management medication,” arguing it incorrectly implies that the disease treated is weight itself.
Dr. Jennifer Baker, representing the European Association for the Study of Obesity, raised concerns about cultural appropriateness, pointing out that in many languages the term “anti” is not even used.
Ms. Vicki Mooney of the European Coalition for People living with Obesity emphasized that “anti” terminology negatively affects patients’ self-worth, instead proposing the term “obesity management medications.”
Dr. Stuart Flint (University of Leeds) recommended “medications for obesity,” explaining that current terminology often evokes feelings of “disgust and hatred” among the general population.
Dr. Sanjeev Sockalingam, representing Obesity Canada, noted that the field does not label treatments as “anti-obesity behavioral interventions” or “anti-obesity surgery,” therefore medications should not be uniquely subjected to this stigmatizing language. As a psychiatrist, he said that there are lessons not to repeat from the history of psychiatric medications. He said that the frequent use of “anti” in psychiatric medication has neither reduced stigma around mental health conditions nor clarified the diverse conditions treated by such medications.
--by Jeremy Alkire, Elaine Young, Monica Oxenreiter, and Kelly Close
[1] EE66.0: obesity due to excess calories; EE66.1: severe obesity due to excess calories; EE66.09: other obesity due to excess calories.
[2] Z codes are part of the ICD-10 coding system and capture additional health-related factors. In obesity diagnoses, Z codes enhance specificity by providing more detailed BMI ranges, which help with precise documentation and tailored treatment planning.
[3] The nine countries are: Argentina, Brazil, Mexico, China, Japan, Taiwan, India, Russian Federation, and the US.
[4] The five-component kidney endpoints are (i) death from kidney causes; (ii) initiation of chronic kidney replacement therapy; (iii) onset of persistent eGFR <15 mL/min/1.73 min2; (iv) persistent ≥50% reduction in eGFR; and (v) onset of persistent macroalbuminuria.
[5] The IWQOL-Lite-CT Physical Function score measures the ability to perform activities such as bending over, standing comfortably, and walking quickly.