ECO 2025 Day #1 Highlights –
Executive Highlights
- Hola from sunny Málaga, Spain, where the European Congress on Obesity (ECO) began Sunday at the indoor-outdoor Palacio de Ferias y Congresos de Málaga, or Malaga Trade Fair and Congress Palace. The excitement of the participants certainly felt appropriate for the venue’s title of palace!
- Dr. Louis Aronne (Weill Cornell) presented much-anticipated results from the SURMOUNT-5 trial, a head-to-head study of tirzepatide versus semaglutide for weight loss in people living with obesity, but not diabetes. Topline study results were announced in December 2024, and full results were simultaneously published today in the New England Journal of Medicine. As obesity rates climb worldwide and GLP-1 RA and dual GLP-1/GIP RAs continue to expand in popularity, there is a need for head-to-head studies comparing the safety and efficacy of market-leading drugs. This study was a phase 3b open-label, randomized, controlled trial. 751 adult participants received the maximum tolerated dose of tirzepatide (10 mg or 15 mg) or semaglutide maximum tolerated dose (1.7 mg or 2.4 mg) subcutaneously once weekly for 72 weeks in a 1:1 ratio. Participants had obesity, but not T2D. Over 72 weeks, tirzepatide, a dual GLP-1/GIP RA resulted in an average weight loss of 20.2%, 47% higher than the 13.7% average weight loss for semaglutide, a GLP-1 RA. In an unusual finding, weight reduction was about 6% lower among men than women in both treatment groups. Dr. Aronne said that further analysis of this difference is required, and that the higher proportion of men in this trial (35%) may account for the slightly lower weight reduction observed compared to previous works. The improvements to A1c, fasting serum glucose, and lipid levels demonstrated by this study also suggest relevance to outcomes for people with diabetes, and we hope to explore similar metrics in future work including diabetes.
See our preview here for more on what’s to come at ECO 2025!
Top Highlight
1. Weight reduction with tirzepatide 15 mg versus semaglutide 2.4 mg from the SURMOUNT-5 trial: 20.2% average versus 13.7% average respectively; amounts to 47% higher weight loss with tirzepatide
Dr. Louis Aronne (Weill Cornell) presented results from a new head-to-head study of tirzepatide versus semaglutide for weight loss in people living with obesity, but not diabetes. Topline study results were announced in December 2024, and full results were simultaneously published today in the New England Journal of Medicine. As obesity rates climb worldwide and GLP-1 RA and dual GLP-1/GIP RAs continue to expand in popularity, there is a need for head-to-head studies comparing the safety and efficacy of market-leading drugs.[1] This study was a phase 3b open-label, randomized, controlled trial. 751 adult participants received the maximum tolerated dose of tirzepatide (10 mg or 15 mg) or semaglutide maximum tolerated dose (1.7 mg or 2.4 mg) subcutaneously once weekly for 72 weeks in a 1:1 ratio. Participants had obesity, but not T2D. Over 72 weeks, tirzepatide, a dual GLP-1/GIP RA resulted in an average weight loss of 20.2%, 47% higher than the 13.7% average weight loss for semaglutide, a GLP-1 RA.
- All primary and key secondary endpoints were met during the study, with tirzepatide demonstrating a statistically significant improvement in weight loss over semaglutide. A higher proportion of all participants achieved all weight reduction targets with tirzepatide compared to semaglutide. Dr. Aronne described the 5.4 cm greater reduction in waist circumference due to tirzepatide as statistically and clinically significant, with a 5 cm difference previously demonstrated to have blood-pressure lowering effects, among other cardiometabolic benefits. He described waist circumference as a surrogate for abdominal visceral fat, which conveys stronger health risks compared to other types of body fat. Weight reduction was about 6% lower among men than women in both treatment groups, which Dr. Aronne described as unusual compared to previous studies of weight loss therapies and in other methods of weight loss. He said that further analysis of this difference is required, and that the higher proportion of men in this trial (35%) may account for the slightly lower weight reduction observed compared to previous works. Dr. Aronne said that coverage and reimbursement differences between the two drugs will likely continue to support the use of semaglutide in many cases, as well as variable treatment approvals for the two drugs. For example, semaglutide has been approved for cardiovascular risk reduction in the US, while a study evaluating tirzepatide for this metric remains ongoing. The improvements to A1c, fasting serum glucose, and lipid levels demonstrated by this study also suggest relevance to outcomes for people with diabetes, and we hope to explore similar metrics in future work including diabetes.
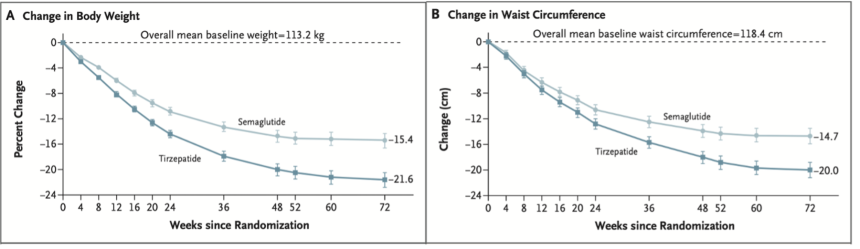
- Results. The mean percentage body weight change at week 72 was −20.2% with tirzepatide and -13.7% with semaglutide. This represents a 47% increase in weight loss using tirzepatide compared to semaglutide. The mean decrease in waist circumference was -18.4 cm with tirzepatide and -13.0 cm with semaglutide, and therefore 42% higher for tirzepatide. Participants taking tirzepatide were 30% more likely to achieve weight reduction targets of 10% body weight loss compared to semaglutide, 60% more likely to achieve 15%, 80% more likely to achieve 20%, and twice as likely to achieve 25% weight loss. In an exploratory endpoint, a total of 20% of participants taking tirzepatide had at least a 30% reduction in body weight compared with 7% taking semaglutide, representing a three-fold increase in likelihood. Tirzepatide was associated with greater improvements in cardiometabolic parameters including greater mean reduction in glycemia, fasting insulin, systolic and diastolic blood pressure, triglycerides, and VLDL cholesterol. It was associated with a greater mean increase in HDL cholesterol, and mean reductions in non-HDL and LDL cholesterol were similar in both groups. In both groups, greater magnitudes of weight reduction were associated with greater improvements to cardiometabolic parameters. In terms of treatment-emergent adverse events, 77% of participants treated with tirzepatide and 79% of those treated with semaglutide reported at least one adverse event during the treatment period, the most frequent of which were gastrointestinal. 6% of participants taking semaglutide discontinued treatment due to gastrointestinal events, as well as 3% of patients taking tirzepatide. Dr. Aronne emphasized these low discontinuation figures, especially in the context of popular understanding of gastrointestinal issues related to these treatments. He noted that the study included careful dosing titration, which may have minimized adverse events. 9% of participants taking tirzepatide reported injection site reactions, compared to 0.3% of patients taking semaglutide, although no participants discontinued treatment due to site reactions. No adverse events led to death.
- Study design. The phase 3b, multicenter, parallel-arm, open-label, randomized, controlled trial was conducted over 72 weeks at 32 sites in the United States and Puerto Rico. Inclusion criteria were an age of 18 years or older, a BMI of 30 or higher or a BMI of of 27 or higher and at least one obesity-related complication, including hypertension, dyslipidemia, obstructive sleep apnea, or cardiovascular disease. Participants also must have reported at least one unsuccessful diet-based effort for weight reduction. Exclusion criteria included a diagnosis of diabetes, previous or planned surgical treatment for obesity, treatment with weight loss medicines including GLP-1 RAs within 90 days before screening, or a change in body weight of over 5 kg within 90 days before screening. Participants (n=751) were randomized, in a 1:1 ratio, to subcutaneously receive 10 mg or 15 mg of tirzepatide, according to the maximum tolerated dose per individual, or the maximum tolerated dose of semaglutide, either 1.7 mg or 2.4 mg. The primary endpoint of the study was percent change in body weight from baseline to week 72 with tirzepatide compared to semaglutide. The key secondary endpoints were weight reduction of 10% or greater, 15%, 20%, and 25%, as well as change in waist circumference.
- Baseline characteristics. The mean age of participants was 45 years. 65% of participants were female, and 76% were White. 57% of participants had prediabetes. Mean body weight was 113 kg, 126 kg in men and 106 kg in women, with a mean BMI of 39 kg/m2. Mean waist circumference at baseline was 118 cm. The average duration of obesity was 16 years, and 50% had at least two obesity-related complications. Although patients with T2D were excluded from this study, the high proportion of participants with prediabetes in this study may have implications for future work including participants with diabetes.
--by Nour Khachemoune and Kelly Close
[1] In 2024, the global market for obesity hit $14.5 billion, doubling from the prior year, with $5.0 million in the last quarter of the year alone, driven by the two GLP-1 behemoths. Wegovy, Novo Nordisk’s semaglutide for obesity, led the overall obesity market share, capturing 58% of sales. Sales for Lilly’s Zepbound (tirzepatide for obesity) represented 38% of the market.