JPM 2025 Full Report –
Executive Highlights
- The 43rd Annual JP Morgan Healthcare Conference wrapped up in mid-January at the Westin St. Francis Hotel in San Francisco. With what appeared to be sharply increased security compared to JPM 2024 and years earlier (we’ve been very lucky to have attended this conference for over two decades), the conference nevertheless progressed very seamlessly. We extend our appreciation to JPM for organizing an especially strong agenda, featuring standing-room only keynote addresses and fireside chats, along with many SRO addresses, in addition to engaging panel discussions, and extremely insightful presentations by industry leaders. A series for women investors in a new “Union Square” room was very popular, featuring very original commentary and discussions - the “sea of pink” was very striking!
- GLP-1 RAs dominated discussion throughout much of the week, as the diabetes and obesity fields continue to be even more dominant over time, moving to become one of the forefronts of the conference. Though the pace of innovation to much better address the diabetes and obesity epidemics continued to both expand and to receive more attention, we were surprised not to hear more about SGLT-2 inhibitors as well as GLP-1 agonists and GLP-1/GIP combinations.
- Of note, some of the days were simply packed with CEOs from the diabetes and obesity “worlds” – for example, on Monday, Vertex’s CEO, Dr. Reshma Kewalramani, opened the entire meeting, speaking in the Colonial Room, followed by Dexcom’s CEO Mr. Kevin Sayer. Both set a pattern for the rooms being absolutely packed during many sessions of the conference! Novartis’ CEO Dr. Vasant (Vas) Narasimhan spoke in the giant Grand Ballroom, and those interested in CV and metabolic health stayed put, as AstraZeneca’s and Sanofi’s top management followed (though we were hoping to hear something from Sanofi on the novel disease-modifying therapy Tzield, of course we know it is still a very small business compared to the typical ones discussed at this giant meeting). Bayer was next, where we were delighted to hear more about Kerendia momentum, then lunch, which featured General (Ret.) Mark Milley, who was the 20th Chair of the Joints Chiefs of Staff (2019-2023) and Ms. Rivka Friedman, Morgan Health’s Head of Innovation. In the afternoon, there was more to learn, with Lilly’s CEO Mr. Dave Ricks speaking, followed by Insulet’s CEO, Mr. Jim Hollingshead.
- See many of our top highlights below with eight conference themes across pipeline innovation in diabetes therapy and technology, clinical trial advancements, US public health policy, AI’s role in healthcare, the economic and geopolitical climate, security, and the riveting women’s health series that started this year. For a look at our on-the-ground view of the conference, see our Day #1, Day #2, and Day #3 coverage. We’ll be back with more on private companies and nonprofit organizations, some of which also had very valuable discussions.
Table of Contents []
-
Themes
- 1. Refined focus and priorities to advance promising candidates for diabetes, obesity, and liver health
- 2. Spotlight on upcoming clinical and regulatory approvals in 2025, with great interest in weight loss management
- 3. Commitment to improving AID algorithms and sensing capabilities to improve outcomes and reduce user burden
- 4. Artificial intelligence: From startups to regulators to national security
- 5. The potential evolution of the US public health policy under the Trump administration
- 6. A time of transition: Tumultuous geopolitical environment generates economic uncertainty
- 7. Women’s Health Series breaks new ground
- 8. Increased security presence following death of UnitedHealthcare CEO
-
Diabetes Therapy Highlights
- 1. Akero Health: Dr. Andrew Cheng highlights updates on MASH candidate, EFX; phase 2b data expected in February 2025, and phase 3 data in 1H26
- 2. Amgen: T2D and obesity candidate MariTide takes center stage, phase 2 readouts and phase 3 initiation in 2025; primary prevention trials for PCSK-9 inhibitor Repatha and olpasiran for Lp(a)
- 3. Amphastar: Increased projected 2025 sales of Baqsimi by $25 million, following promotional agreement with MannKind; GDUFA date for generic injectable GLP-1 RA set for 2Q25
- 4. AstraZeneca: Obesity program to drive growth beyond 2030; continued commitment to the development of small molecules
- 5. Bayer: Eylea HD as standard treatment for retinal disease; strong momentum of Kerendia (finerenone) in heart failure, with new prespecified analysis
- 6. Biomea: Refined pipeline focus on diabetes and obesity with icovamenib, a first-in-class menin inhibitor
- 7. Daiichi Sankyo: Nothing new from Dr. Sunao Manabe on pain
- 8. Ionis: Preparing for the launch of Tryngolza (olezarsen) as the first FDA-approved treatment for familial chylomicronemia syndrome
- 9. J&J: CEO Mr. Joaquin Duato discusses elements for success with a diversified portfolio, increasing attention for MedTech and Innovative Medicine
- 10. Lilly: Continued momentum with Mounjaro and Zepbound, advancing incretin treatments for diabetes, obesity, and beyond; upcoming “big assets” include oral GLP-1 orforglipron and “triple G” retatrutide
- 11. Madrigal: “Foundational MASH treatment” Rezdiffra sales total $180 million in the first nine months, with over 11,800 patients treated
- 12. Merck: Diversifying portfolio in diabetic macular edema; upcoming key readouts with PCSK9 and GLP-1 RA
- 13. Novartis: Innovation across four therapeutic areas, with significant progression of Entresto and Leqvio for heart failure and CVD
- 14. OPKO Health: Dr. Elias Zerhouni highlights the promising potential of dual GLP-1/glucagon RA in development for oral and injectable formulations
- 15. Pfizer: Dr. Albert Bourla expects phase 3 trial of once-daily oral GLP-1 in 2H25; confidence in capturing oral GLP-1 RA market
- 16. Regeneron: Dr. Leonard Schleifer highlights strategies to increase Eylea HD uptake; phase 2 data of trevogrumab (anti-myostatin antibody) with semaglutide expected in 2H25
- 16. Roche: Continued clinical progress in ophthalmology and cardiovascular-renal-metabolic portfolio; phase 1b trial of anti-TL1A therapy in MASH to initiate in 1Q25
- 17. Sanofi: Strides to become the world leader for immunology; regulatory decision of Tzield in China expected in 2H25
- 18. Terns Pharmaceuticals: High confidence in oral GLP-1 RA TERN-601, compared to competitors; phase 2 data expected in 2H25
- 19. Vertex: Suzetrigine to launch for acute pain following PDUFA date in January 2025; T1D phase 3 trial of VX-880 (now named zimislecel) to complete enrollment and initial dosing in 2025
- 20. Viking: first appearance at JPM highlights VK2735 (dual GLP-1/GIP RA) advancing to phase 3 obesity trials in 1H25, following end-of-phase 2 FDA meeting
- 21. Zealand: CEO Dr. Adam Steensberg highlights “strongest pipeline in obesity” with differentiated approach with amylin analog
-
Diabetes Technology Highlights
- 1. Dexcom: Preliminary full-year 2024 revenue of $4.03 billion (+11%) and 4Q24 revenue of $1.11 billion (+8%); global userbase grows 25% to 2.8-2.9 million; developmental multi-analyte sensors confirmed
- 2. embecta: Gaining financial stability, international GLP-1 opportunities, and revitalization
- 3. Insulet: Omnipod 5 launched in five additional European countries, with an additional five launches to follow this year; significant growth opportunity in highly underpenetrated T2D population
- 4. Medtronic: Focus on expanding MiniMed 780G indications for T2D, biosimilars, preschoolers, and pregnancy; goal to introduce Simplera in US by end of 2025
- 5. Teladoc: Collaboration with Amazon’s Health Benefits Connector to increase access to virtual cardiometabolic programs announced
- 6. Virta Health: GLP-1 RAs positioned as non-essential for metabolic success but still beneficial; over $100 million in revenue by end of 2024
-
Diabetes Big Picture Highlights
- 1. JPMorgan Chase CEO Mr. Jamie Dimon talks China, GLP-1 RAs, AI, the Trump administration, healthcare frustrations, and more in noontime conversation
- 2. The early innings of AI in healthcare: Panel discussion emphasizes the need for data cleaning and transparency to guide AI’s implementation
- 3. Inside the FDA: Panel discussion offers insight into approval processes, serving as an opportunity for connection between developers and regulators
- 5. Retired General Mark Milley tackles geopolitics and the military’s role in health innovation
- 9. ARPA-H: Catalyzing biomedical breakthroughs in women’s health through the Sprint for Women’s Health, NITRO, and POSEIDON
- 10. The US healthcare landscape: Former FDA Commissioner Dr. Scott Gottlieb speculates on the potential impact of Trump administration on biomedical innovation and public health
- 11. Fireside chat with HHS Secretary Xavier Becerra: Analyzing the Biden administration’s accomplishments, including MDPNP, ACA, and vaccines
- 12. AI in diagnostic testing: Panel demonstrates opportunities for innovation and equity, while calling for regulation and caution
- 13. Investment opportunities to drive innovation in emerging markets
Themes
1. Refined focus and priorities to advance promising candidates for diabetes, obesity, and liver health
Several companies outlined their refined focus and priorities to build a stronger pipeline for improving patient outcomes for diabetes, obesity, and liver health. Reflecting on all the positive results and approvals across the field, countless presentations spotlighted the strong momentum and high expectations for 2025. We particularly appreciated hearing updates on companies dedicating resources and improving their pipeline to advance impactful opportunities to the next stages of development.
- In one of the most packed sessions of the conference, Lilly’s CEO Mr. David Ricks highlighted significant milestones across 2024, with Mounjaro (tirzepatide for diabetes) and Zepbound (tirzepatide for obesity) showing strong growth in the guidance published Tuesday. While the guidance indicated lower-than-expected revenue, Lilly expects 2024 full-year worldwide revenue to be approximately $45 billion (+32%). In 4Q24, revenue for Mounjaro reached approximately $3.5 billion and $1.9 billion for Zepbound, compared to $3.1 billion in 3Q24 and $2.2 billion in 4Q23 for Mounjaro and compared to $1.2 billion in 3Q24 and $175 million in 4Q23 for Zepbound. To meet increasing demand, Lilly plans to bring additional manufacturing capacity and expects to produce at least 60% more scalable doses of incretins in 1H25 compared to 1H24. Especially with the FDA announcing the shortage resolution of tirzepatide in December 2024, we’re encouraged to see Lilly focus on continuing its robust growth.
- With Rezdiffra (resmetirom), the first and only FDA-approved treatment for MASH, CEO Mr. Bill Sibold highlighted Madrigal’s strong leadership and significant potential for continued success. According to Madrigal’s preliminary financials published on Monday, Rezdiffra revenue totaled $100-103 million in 4Q24 and $166-180 million in 2024, remarkable growth for only three quarters on the market. Furthermore, Mr. Sibold said that more than 11,800 patients are on Rezdiffra, also reflecting significant growth from over 6,800 patients in 3Q24 and just ~2,000 in 2Q24. Mr. Sibold also emphasized that Rezdiffra will be a foundational treatment for MASH, continuing to target 315,000 people diagnosed with F2 and F3. Madrigal plans to continue a strong launch of Rezdiffra in the US and awaits approval in the EU in 2025, planning for commercial launch in Germany and potentially expanding to other countries. Mr. Sibold also highlighted plans to build a pipeline to secure long-term growth, perhaps exploring the next best mechanism of action or combination treatments that enhance Rezdiffra’s efficacy.
- Refining the pipeline to increase focus on cardiometabolic health, Biomea CEO Mr. Thomas Butler reiterated the announcement on Monday that the company will become a diabetes and obesity treatment company based on promising results of recent clinical trials. Biomea plans to conclude its studies exploring icovamenib (potent and selective covalent menin inhibitor) in oncology and explore partnerships to further advance its oncology assets. Most recently, in December 2024, Biomea announced positive topline results of the phase 2 COVALENT-111 study of icovamenib in T2D. Biomea continues to advance icovamenib, aiming to address challenges with current standards of care with high discontinuation rates of GLP-1 RAs, SGLT-2 inhibitors, and DPP-4 inhibitors. In particular, icovamenib targets two specific populations: (i) people with severely-insulin deficient diabetes; and (ii) people on GLP-1 RA treatment.
2. Spotlight on upcoming clinical and regulatory approvals in 2025, with great interest in weight loss management
Companies celebrated achievements on pipelines from last year and discussed upcoming milestones for the new year. We were encouraged to see several regulatory approvals in 2024, including Madrigal’s Rezdiffra (resmetirom) for MASH in March 2024, Lilly’s Zepbound (tirzepatide) for obstructive sleep apnea in December 2024, and Bayer’s Eylea HD (aflibercept 8mg) and Roche’s Vabysmo (faricimab) prefilled syringes for eye diseases. Companies highlighted early performances of these products and strategies to capture a greater patient base by securing more formulary access, educating physicians, and establishing a strong supply chain.
- We were also excited about the upcoming trial readouts and regulatory milestones expected in 2025. We highlight several pipeline updates below:
- In T1D therapy and cures, Vertex plans to share phase 1/2 (n=17) results for VX-264, a cell-encapsulated device of stem cell-derived islets, in 2025. Sanofi expects a regulatory decision for Tzield (teplizumab) in China in 2H25.
- On weight loss management, Amgen plans to initiate its phase 3 program MARTIME between 1H25 and 2H24. Additional data from the ongoing phase 2 trial (expected to complete in early 2026) will be read out in 2H25, as well as phase 2 results in T2D. Zealand will launch a phase 2b ZUPREME-2 trial for petrelintide (amylin analog) for overweight or obesity in 1H25, and a phase 1b combination trial with petrelintide and GLP-1 RA in 2025. Regeneron’s phase 2 study of trevogrumab (anti-myostatin) and semaglutide combination therapy to minimize muscle loss during obesity treatment is expected in 2H25. AstraZeneca also expects a phase 2 data readout this year for an obesity program, although the candidate was not specified during the meeting. Terns Pharmaceutical’s phase 2 data of once-daily GLP-1 RA TERN-601 is expected in 2H25. Lastly, Amphastar’s generic injectable GLP-1 RA, AMP-018, awaits the FDA’s decision by the GDUFA date in 2Q25.
- In ophthalmology and liver health, Regeneron plans to launch pre-filled syringes of Eylea HD by mid-2025 in the US. Akero Health expects to share phase 2b data for efruxifermin (EFX) in February 2025 for MASH.
- For cardiovascular health, Amgen’s phase 3 VESALIUS-CV trial (n=12,301), investigating Repatha in adults at high cardiovascular risk and without prior myocardial infarction or stroke, is expected to complete in July 2025, and its data readout is expected in 2H25. Novartis’ data for the Lp(a)-HORIZON trial (n=8,323) assessing pelacarsen in major cardiovascular events in people with CVD is expected in May 2025.
- Companies expressed commitment to engage in lively discussions with the FDA to design trials, especially given the FDA’s draft guidance on developing weight-loss therapies and increasing diversity. For example, Viking CEO Dr. Brian Lian shared during Q&A that Viking had an end-of-phase 2 meeting with the FDA before these draft recommendations were published and knew where the agency stood on these topics. Additionally, a panel discussion by representatives from the FDA was fully dedicated to elucidating the agency’s approval processes and the Total Product Life Cycle (TPLC) Advisory Program (TAP) Pilot, which aims to streamline approval and increase communication between developers and regulators. The panel highlighted the differences in approval processes between medical devices and pharmaceuticals, the importance of building trust and relationships with the FDA.
3. Commitment to improving AID algorithms and sensing capabilities to improve outcomes and reduce user burden
Like therapy, innovation in diabetes technology continues at breakneck pace. Presentations from CGM and AID manufacturers underscored the industry’s commitment to developing more advanced algorithms, multi-analyte sensor probes, and smaller form factors to reduce user burden and further improve glycemic outcomes. Alongside this rapid innovation, the industry remains focused on expanding insurance coverage for diabetes technology, particularly in the T2D population, which has historically underused these devices.
- In CGM, Dexcom successfully submitted a 15-day Dexcom G7 to the FDA in 3Q24, which is expected to launch in 2H25. The company also has planned several software enhancements this year, including in the Dexcom Follow app. Development of the next-generation Dexcom G8 is also advancing with some individuals now wearing the sensor, which features more advanced electronics to accommodate multi-analyte sensors. CEO Mr. Kevin Sayer added that Dexcom is advancing several probes with extended lifespans – some of which can measure “two or three [analytes]” simultaneously.
- In AID, Medtronic’s CEO Mr. Geoff Martha briefly commented on the company’s next-generation AID pipeline, including a reveal of a potential form factor for an 800-Series pump and a patch pump. While details are scarce on these pumps, the company first unveiled the 800-Series pump at its Investor Day at ADA 2023 – a screenless, tethered pump that is roughly half the size of the 700-Series pump and will be fully controlled from a user’s smartphone. Insulet’s CEO Dr. Jim Hollingshead highlighted in his remarks that the company continues to advance development of next-generation AID algorithms, including SmartAdjust 2.0 and the fully closed-loop EVOLUTION algorithm.
- Beyond innovation, the technology industry continues to progress efforts to improve access to these life-changing technologies, particularly for T2D. Dexcom is committed to obtaining complete reimbursement for CGM for people with T2D on non-insulin therapy, which represents over 25 million people in the US alone. As of this month, Mr. Sayer announced that two of the three largest PBMs cover Dexcom CGM for anyone with diabetes, which will enable commercial coverage for over five million people with T2D on non-insulin therapy by the end of 2025. Several companies are expanding their sales forces to better reach the T2D population, including Dexcom and Insulet (after Omnipod 5’s FDA clearance for T2D in August 2024). Beyond T2D, Insulet remains focused on growing technology adoption among people with T1D, as the company will introduce Omnipod 5 into 10 international markets in 2025, with five launches already announced this week – Denmark, Finland, Italy, Norway, and Sweden. Launches in Australia, Belgium, Canada, Israel, and Switzerland are expected to follow later this year.
4. Artificial intelligence: From startups to regulators to national security
JPM 2025 highlighted AI’s impact on healthcare in multiple arenas, featuring multiple AI-focused panels with significant discussion across industry updates and broad-ranging keynotes. In his opening keynote, JPMorgan CEO Mr. Jamie Dimon reiterated the power of AI to accelerate significant breakthroughs in medicine, such as curing cancer and identifying disease-causing gene defects. He envisions AI eliminating a large portion of human medical error, citing a 2003 study showing 30,000 hospital deaths per year due to human error. AI seemed to weigh on the minds of presenters, as if speakers knew they must be at the forefront of AI innovation.
- JPM 2025 featured several riveting panels on the potential of AI to improve healthcare. A panel featuring Dr. Shiv Rao (CEO, Abridge), Mr. Hamid Tabatabaie (CEO, CodaMetrix), Ms. Seema Verma (EVP and General Manager, Oracle Health and Life Sciences), and Mr. Omri Yoffe (CEO, Vi) unanimously agreed that AI’s implementation in healthcare remains in its “early innings,” proposing several key considerations to guide ethical and effective use to support clinical decision making. In particular, they called for data cleaning and transparency to guide AI’s implementation in clinical settings. On Day #3, a separate panel analyzed AI’s role in diagnostic testing. Panelists deemed AI to be transformative, envisioning huge potential for rural medicine to connect more communities to academic medical centers. Despite the potential of AI, Mr. Troy Tazbaz (Director of Digital Health Center for Excellence, FDA) acknowledged the need for regulation and post-market monitoring at all hospitals, in a push for equity. Mr. Dimon similarly noted the need for regulation, likening AI to other dependable technologies, such as planes, cars, and pharmaceutical innovations, that can be abused but are safe and essential with proper government regulation.
- Several pharmaceutical and biotechnology companies stated their intentions to leverage AI to assist with R&D and improve user experiences. Pfizer’s CEO Dr. Albert Bourla discussed how the company can improve R&D productivity and operational efficiency through AI, while Dexcom’s CEO Mr. Kevin Sayer highlighted the company’s launch of a generative AI to provide more personalized insights in the Stelo app through its use. Virta Health’s CEO Mr. Sami Inkinen also described how its digital cardiometabolic platform leverages AI and drives users to achieve their weight loss goals.
5. The potential evolution of the US public health policy under the Trump administration
JPM 2025 featured significant discussion on potential reform to US public health landscape under the incoming Trump administration. Panelists acknowledged the possibility for significant changes to US healthcare policy, including health insurance, PBMs, vaccinations, and more. Despite potential changes to healthcare policy in the coming years, panelists appeared optimistic that the pace of innovation in the healthcare industry would continue to accelerate. Specifically, former FDA Commissioner Dr. Scott Gottlieb said that the field has reached an “inflection point” where regenerative medicine seems inevitable. Several FDA representatives did not foresee significant impact on regulatory approval processes. Dr. Laura Gottschalk suggested the presidency and the Administrator of the CMS have limited influence, and she noted that the agency possesses staff that served under both the prior Trump and Biden administrations.
- Potential coverage changes for medical care and treatment were top of mind. Outgoing Secretary of the US Department of Health and Human Services (HHS) Xavier Becerra highlighted progress in the Biden administration to further expand health insurance coverage for millions of Americans, noting that over 300 million people now have health insurance. For instance, Sec. Becerra said that women on Medicaid can receive postpartum care for 365 days after birth, up from 60 days previously, to address the high rates of maternal morbidity and mortality. However, throughout the conference, stakeholders expressed concern regarding the incoming Trump administration’s intentions to reform healthcare policy. Several sessions discussed the feasibility of reform in several areas:
- Affordable Care Act (ACA): Addressing the incoming Trump administration’s threats to weaken or repeal the ACA, Sec. Becerra suggested this legislation cannot be undone easily due to the millions of Americans who depend on the ACA for health insurance. He said a record number of individuals, nearly 24 million, are receiving their health insurance through this legislation; therefore, if the ACA is targeted for reform, he expects substantial public opposition that would forestall significant repeals or adjustments to ACA coverage.
- Drug pricing: Due to the establishment of the Medicare Drug Price Negotiation Program (MDPNP) through the Inflation Reduction Act (IRA), drug pricing reform was of high interest throughout the conference. Dr. Gottlieb does expect Medicare reimbursement for GLP-1 RAs in obesity to be withdrawn because most of the elderly population will become eligible for these therapies as their indications further expand, stating CMS “saw the writing was on the wall” when proposing this policy. Beyond GLP-1 RAs, Dr. Gottlieb suggested the IRA could be reformed to remove orphan drug exemptions. He was skeptical that small molecule exemptions would be adjusted to establish equivalent price negotiation exemption for small molecules and biologics (i.e., increase small molecules nine-year exemption window to 13 years to match biologics’ exemption window) since the government will primarily seek to save money.
- Pharmacy benefit managers (PBMs): With a PBM Reform Act introduced in Congress, potential regulation of PBM was a focus. Dr. Gottlieb suggested this legislation could be incorporated into the reconciliation bill; however, he expressed concern it could but impede more productive long-term reform by antagonizing PBMs and increasing their resistance to further action.
- Interest in potential regulation of the food industry to encourage healthier product production and consumption was high. Dr. Gottlieb said the FDA strongly supports these efforts, proposing several changes to regulation on food, including: (i) more forceful disclosures on unhealthy food attributes; or (ii) permitting food manufacturers to make broader claims on healthy attributes and therefore directly compete on product healthiness.
- Given previous criticism of vaccines by individuals nominated to critical public health positions in the incoming administration, several sessions probed possible changes to vaccination policy. Sec. Becerra emphasized that the HHS operates on “science, not intuition or politics,” stating that the evidence supports the success of vaccines. Dr. Gottlieb also expressed concern that efforts to soften childhood vaccination requirements, for example in schools, could lead to significant decreases in vaccination rates, although he believes that President-elect Trump understood the importance of childhood vaccination in his briefings with him.
6. A time of transition: Tumultuous geopolitical environment generates economic uncertainty
Like JPM 2024, several keynote addresses acknowledged a challenging and tense geopolitical environment and speculated on the potential influence of the incoming Trump administration. Panelists expressed subtle concern over the long-term health of the economy in the current geopolitical climate, emphasizing this period as a time of transition globally with likely ramifications for the healthcare industry. In his keynote address, Mr. Jamie Dimon projected limited change at the inauguration of the new Trump administration, arguing that the US president does not affect the economy in the first year of their administration. However, he shared his worries about the economic future beyond 2025.
- Mr. Dimon and retired Gen. Mark Milley likened the current geopolitical climate to the state of the world before World War II. Gen. Milley pointed to the rise of nationalism, protectionism, populism, and tariffs as factors that may potentially disrupt peace. He advocated for diplomacy and for the US to negotiate with “adversaries” such as China. Mr. Dimon similarly urged US leaders to steadfastly prepare to address any unexpected geopolitical and economic crises.
- Given the evolving economic climate, cost considerations for GLP-1 RAs were a hot topic of discussion after the Biden administration proposed expanded Medicare and Medicaid coverage of anti-obesity medications in November 2024. When asked about the balance between GLP-1 RAs’ effectiveness and the costs of long-term use, Mr. Dimon strongly expressed his belief that use of GLP-1 RAs would present less of a cost over time than the consequences of diabetes and heart disease, urging the industry to move towards disease prevention when possible. (Of note, JPMorgan Chase covers employee costs of the drugs for both obesity and diabetes.)
7. Women’s Health Series breaks new ground
In what’s a first at the conference, JPM 2025 featured what it called a Women’s Health Series – a collection of very interesting panel discussions with focus on women in healthcare. While improving women’s health outcomes and catalyzing new innovations for diseases that predominately affect women are certainly major goals, part of the aim also appeared to be gathering women leaders together. Attendees were encouraged to wear pink in solidarity on the second day of the conference in recognition of this focus and it was striking to see upwards of 400 women in pink standing together in the middle of Union Square. Men who wore pink won praise from the women we heard speaking! In addition to ARPA-H, conversations centered on leadership in biopharma, the future of women’s healthcare, and the flow of capital to women’s health.
- ARPA-H: ARPA-H, a federal agency formed in 2022 that aims to achieve transformative, sustainable, and equitable health solutions, hosted an afternoon panel discussion devoted to discussing the agency’s efforts to catalyze biomedical breakthroughs to support women’s health. In 2024, ARPA-H initiated the Sprint for Women’s Health to fund and accelerate development of biomedical therapies and technologies to improve women’s health outcomes, providing $113 million in funding across 24 awards to support novel solutions for unmet needs in women’s health. As one example, this initiative provided $10 million in funding to support Daré Bioscience’s development of a novel treatment to clear persistent high-risk human papillomavirus (hrHPV) infection, which causes cervical cancer. Dr. Ross Uhrich (Program Manager, ARPA-H) also highlighted the efforts of the NITRO and POSEIDON initiatives to develop breakthrough therapies for osteoarthritis, which predominately affects women, and cancer screenings, respectively.
8. Increased security presence following death of UnitedHealthcare CEO
JPM 2025 was marked by a noticeably increased security presence compared to last year’s conference, stemming from the murder of UnitedHealthcare CEO Mr. Brian Thompson in December 2024. See Mr. James S. Hirsch’s reflections on the death as well as his musings on the US healthcare industry in the wake of this event. Significantly more San Francisco police officers were stationed both outside and inside venue than JPM 2024, fence barricades were erected around the perimeter of the entrance, and security personnel occasionally reminded exiting attendees to remove their name badges outside the venue. This heightened security seemed to reflect the uneasiness of many executives and attendees amidst contentious public sentiment on the healthcare industry ignited by the death of Mr. Thompson, which was underscored by peaceful demonstrations in Union Square on the opening day. We extend our gratitude to the police officers and security personnel for their efforts to secure the event and ensure smooth operations.
Diabetes Therapy Highlights
1. Akero Health: Dr. Andrew Cheng highlights updates on MASH candidate, EFX; phase 2b data expected in February 2025, and phase 3 data in 1H26
President and CEO Dr. Andrew Cheng provided promising updates on Akero Health’s lead MASH candidate, efruxifermin (EFX). See webcast here. As background, EFX is a bivalent fibroblast growth factor 21 (FGF21) analog that can regulate multiple metabolic pathways and cellular processes. With once-weekly dosing, EFX has the potential to treat MASH with or without cirrhosis. Celebrating Akero Health’s transition to phase 3 trials, Dr. Cheng highlighted advances in clinical trials and shared post-hoc analyses of phase 2 data. We’re excited to see Akero Health’s progress in MASH amid the evolving competitive landscape.

- In February 2025, Akero Health will share 96-week data of the phase 2b SYMMETRY study of EFX in patients with compensated cirrhosis (F4) from MASH. This is an exciting news, given limited treatment aside from liver transplantation for people with cirrhosis – this population has a significantly higher five-year mortality rate (~50%) than those without (less than 15%).
- As background, the primary readout of 36-week data was shared in October 2023, in which EFX demonstrated up to 10% placebo-corrected improvement in fibrosis without worsening of MASH. While these results did not meet statistical significance (n=0.279), Dr. Cheng noted that these were the best results seen yet in this clinically vulnerable population. EFX demonstrated the highest placebo-corrected improvement compared to competitors such as Novo Nordisk’s semaglutide (18%) and Gilead’s selonsertib (1%). Furthermore, one 69-year-old female participant recovered from F4 to F1 after nine months of treatment, in addition to a weight loss of ~2 kg (4 lbs), suggesting an antifibrotic activity of EFX.
- Dr. Cheng also highlighted results of the phase 2b HARMONY trial (n=128) announced in March 2024. The study demonstrated statistically significant fibrosis improvement of 75% in the EFX arm, compared to 24% on placebo in people with MASH but not cirrhosis at Week 96. Comparison to 24-week data, which demonstrated 41% improvement in fibrosis, suggests that longer dosing improves response. As well, 63% of initial non-responders responded to EFX.
- Finally, the phase 3 SYNCHRONY Real-World study (n=601), which evaluates safety and tolerability of EFX in patients with non-invasively diagnosed MASH or MASLD, completed patient enrollment in January 2024, and data is expected in 1H26. The other two trials of the SYNCHRONY program focus on efficacy. SYNCHRONY Histology evaluates the efficacy and safety of EFX in patients with biopsy-confirmed pre-cirrhotic MASH, with data including liver biopsy expected in 2027. The 260-week SYNCRONY Outcomes trial evaluates EFX on the time to first occurrence clinical events, in addition to histologic fibrosis improvement, in patients with non-invasively diagnosed MASH.
2. Amgen: T2D and obesity candidate MariTide takes center stage, phase 2 readouts and phase 3 initiation in 2025; primary prevention trials for PCSK-9 inhibitor Repatha and olpasiran for Lp(a)
Amgen’s CEO Mr. Bob Bradway highlighted upcoming pipeline milestones for the company in 2025, with particular emphasis on GIP antagonist/GLP-1 RA MariTide. Mr. Bradway began his presentation recognizing the devastation caused by the wildfires in Southern California, where Amgen is based, and said that the company is committed to rebuilding the community, starting with $10 million in support announced the day of his talk. We have been excited to see Amgen’s relatively large focus on obesity and obesity-related conditions, which falls under its “General Medicine” area – the other three areas are rare diseases, oncology, and inflammation. MariTide, PCKS9 inhibitor Repatha, and Lp(a)-lowering olpasiran were the main areas of focus of Mr. Bradway’s prepared remarks on the cardiometabolic treatment front. While Amgen is also developing half a dozen preclinical and early stage obesity candidates, including phase 1 AMG 513, Mr. Bradway did not discuss any specifics beyond sharing that the company is still in its early days on this front and that decisions on which molecules Amgen will promote will depend on the data.
- “We’ll be the first long-acting therapy in the treatment of obesity.” In response to JPM’s Mr. Chris Schott’s question about MariTide’s potential commercial hurdles and capabilities as the “third entrants into the market,” Mr. Bradway said he would instead position MariTide as the first in long-acting therapy for obesity. MariTide’s monthly (or even less frequent) dosing schedule, which he characterized as potentially “very attractive” to patients, was a key point in Mr. Bradway’s prepared remarks. He also highlighted positive phase 2 results reported in November 2024. Recall that MariTide conferred ~20% weight loss in people with obesity or overweight and ~17% weight loss in T2D at 52 weeks. Notably, Mr. Bradway pointed out that weight loss had not yet plateaued at 52 weeks, adding that Amgen will continue to share incremental data this year. In participants with T2D, MariTide conferred a 2.2% A1c reduction. Mr. Bradway said that over half of these participants achieved an A1c below 5.7%, indicating T2D remission.
- In 2025, Amgen plans to initiate its phase 3 program MARTIME between 1H25 and 2H24. Additional data from the ongoing phase 2 trial (expected to complete in early 2026) will be readout in 2H25 as will phase 2 results in T2D specifically. Looking ahead, Mr. Bradway also championed Amgen’s capability to meet potential supply demands, which it has been able to demonstrate across its portfolio in the past.
- On PCSK-9 inhibitor Repatha, we were excited to hear Amgen’s interest in a potential primary prevention indication. During the Q&A, Mr. Bradway emphasized the importance of lowering LDL levels to prevent heart attacks and strokes and that “the sooner, the better.” We imagine that a primary prevention indication will depend on results from the phase 3 VESALIUS-CV trial (n=12,301), which is investigating Repatha in adults at high cardiovascular risk and without prior myocardial infarction or stroke. The trial is expected to complete in July 2025, and its data readout is expected in 2H25. On Repatha broadly, Mr. Bradway emphasized the large need (>100 million patients) for effective treatment for heart disease. He said that Repatha’s remarkable growth (up 40% in 3Q24 with an expected 2024 annualization of over $2 billion) has been driven by its “enormous efficacy,” the unmet need for treatment, and access.
- On Lp(a)-lowering olpasiran, we were again delighted to hear Amgen’s interest in primary prevention, with plans to initiate a phase 3 study in 2H25. Mr. Bradway highlighted that olpasiran would be a first-in-class siRNA molecule targeting Lp(a), which affects ~20% of the population and is nonmodifiable with diet or exercise. Phase 2 results demonstrated that olpasiran can reduce Lp(a) concentration by over 95% in people with ASCVD, and its phase 3 Ocean(a)-Outcomes trial (n= 7,297) is ongoing and fully enrolled.
- On biosimilars, as with last year, Mr. Bradway highlighted the company’s global leadership in the area, sharing that he is proud of the fact that Amgen has a 100% success rate with the FDA approvals for biosimilars. He also highlighted that the company has been able to “supply every patient, every time with a biosimilar molecular when they come asking for one.” While there was no mention of Eylea biosimilar Pavblu, the company’s slides noted that it was launched last year.
3. Amphastar: Increased projected 2025 sales of Baqsimi by $25 million, following promotional agreement with MannKind; GDUFA date for generic injectable GLP-1 RA set for 2Q25
CEO Dr. Jack Zhang and CFO Mr. Bill Peters shared Amphastar’s business strategies and pipeline updates, focusing on nasal glucagon Baqsimi, insulin analogs, and a generic GLP-1 RA candidate (see webcast and presentation slides). Dr. Zhang introduced Amphastar’s dual growth strategy, which involves both internal pipeline development as well as strategic acquisitions of product facilities. As background, the company developed the first and only FDA-approved generic traditional glucagon injection kit in 2020 and acquired Baqsimi from Lilly in June 2023. Amphastar has seen significant growth over the past several years, with revenue totaling $191 million in 3Q24, up 5% from 3Q23. Looking forward, Amphastar plans to shift its pipeline from predominantly generic products (comprising 63% of the pipeline in 2021) to proprietary and biosimilar products (50% and 35%, respectively, by 2026). Alongside this shift, Amphastar will continue investing significantly (~21% of revenue) to R&D and diversifying products and revenue base. See the company’s diabetes portfolio below.

- Baqsimi (nasal glucagon) continues to be Amphastar’s biggest branded product with growing sales, following its spin-out from Lilly in June 2023. As the only FDA-approved non-injectable glucagon, Baqsimi offers a user-friendly, ready-to-use solution for treating severe hypoglycemia, especially for caregivers, friends, and school staff who may be unfamiliar with injectable products during emergencies. Despite ADA recommendations, however, Mr. Peters noted that only 10% of people on insulin use glucagon, indicating a significant opportunity for much greater market expansion. To raise greater awareness, Amphastar has signed a promotional agreement with MannKind, a manufacturer of inhaled insulin Afrezza, in 3Q24. Amphastar has raised the peak sales projection for Baqsimi to $250-275 million, up from $225-250 million, to reflect the partnership with MannKind that would double the salesforce. The company also announced a 3% increase in the unit price this year – while no price increase would be better for patients and the system, we understand the price increase is important to offset price increases in raw materials and other increased expenses. Excitingly, Mr. Peters also shared that Baqsimi has expanded its international footprint.
- Amphastar awaits the FDA’s decision on the generic injectable GLP-1 RA, AMP-018, as the GDUFA (generic drug user fee amendments) is set for 2Q25. If approved, Amphastar expects AMP-018 to have revenue potential of ~$1.1 billion, according to IQVIA. That said, during Q&A, Dr. Zhang acknowledged that the company will face significant competition in the GLP-1 RA landscape.
- Amphastar’s interchangeable insulin pipeline aims to cover the full spectrum of insulin, including insulin aspart AMP-004 (Novolog), recombinant human insulin AMP-005, and insulin degludec AMP-025 (Tresiba). Mr. Peters updated that Amphastar resubmitted the Biologics License Application (BLA) for AMP-004 in 4Q24, following multiple FDA meetings to confirm submission requirements. He added that AMP-005 is manufactured at the company’s facility in France, while the API production for AMP-025 is based in China, reflecting on the company’s global manufacturing capabilities.
4. AstraZeneca: Obesity program to drive growth beyond 2030; continued commitment to the development of small molecules
CFO Ms. Aradhana Sarin delivered a comprehensive overview of AstraZeneca’s financial growth and late-stage pipeline, spanning oncology, biopharmaceuticals, and rare diseases (see webcast and presentation slides). AstraZeneca saw robust growth in 2024, totaling $39 billion for nine months (up 19% from 2023) and marking double-digit growth across all therapeutic areas and in the US, Europe, and emerging markets by geography. Of note, cardiovascular, renal, and metabolism (CVRM) sales totaled $9.4 billion for nine months of 2024, contributing 24% of total sales. Ms. Sarin boasted AstraZeneca’s broad pipeline, including 91 ongoing late-stage trials, each with peak year revenue greater than $1 billion. Continuing this momentum, AstraZeneca aims to deliver $80 billion in total revenue and launch at least 20 new molecular entities by 2030. The company is also preparing for growth beyond 2030 by investing in areas such as weight management, antibody conjugates, and cell therapy. During Q&A, however, Ms. Sarin added that AstraZeneca expects to face a headwind from Farxiga value-based purchasing 11 VBP 11).
- AstraZeneca made significant progress on obesity programs, including: (i) AZD5004 (oral GLP-1 RA); (ii) AZD6234 (long-acting amylin); and (iii) AZD9550 (GLP-1/glucagon RA). These candidates are expected to drive growth beyond 2030. As we heard from ObesityWeek®, these obesity candidates have moved into phase 2 trials, following positive phase 1 results. While Ms. Sarin did not clarify the candidate, there will be a phase 2 data readout for the obesity program in 2025. Based on the following completion dates (listed in 3Q24), we anticipate select trials of the following will have data readout this year: (i) VISTA trial (n=304) for AZD5004 in obesity/overweight, plus one comorbidity (expected to complete in December 2025); or (ii) CONTEMPO trial (n=88) for safety of AZD9550 in obesity/overweight with or without T2D (expected to complete in April 2025).
- Baxdrostat (aldosterone synthase inhibitor) is among the seven candidates that will have first phase 3 results in 2025. Approximately 45 million people in the US, Japan, and Europe have difficulty treating hypertension, despite standards of care. In phase 2b BrigHTN trial (n=275), baxdrostat conferred a 20% reduction in systolic blood pressure (SBP) at Week 12, compared to a 9% reduction with placebo. Phase 3 BaxHTN trial (n=720), investigating the drug’s effect on SBP at Week 12, has completed enrollment in 4Q24 and is expected to complete in 2H25. Data from the phase 3 Bax24 trial (n=212), investigating baxdrostat on ambulatory blood pressure is expected in 2H25. Ms. Sarin also briefly discussed that BaxDuo-Arctic trial (n=2500) assesses combination therapy of baxdrostat and SGLT-2 inhibitor Farxiga (dapagliflozin) and is expected to complete in December 2027.
- When asked about what modalities AstraZeneca will focus on, given the US Inflation Reduction Act 2022,[1] Ms. Sarin expressed continued commitment toward small molecules, which have the highest value and biggest opportunities, such as AZD5004 or oral PCSK9.
5. Bayer: Eylea HD as standard treatment for retinal disease; strong momentum of Kerendia (finerenone) in heart failure, with new prespecified analysis
In the afternoon, President Mr. Stefan Oelrich presented updates on Bayer’s promising therapies, including ones of high interest to us, Kerendia (finerenone) and Eylea (aflibercept).
- Eylea remains a leader in the retinal market and sustains its strong market position. Mr. Oelrich positioned Eylea as the number one anti-VEGF treatment since 2016 and called Eylea HD (aflibercept 8 mg) the new standard for retinal diseases. Last year, around the time of JPM 2024, Eylea HD received approval for neovascular age-related macular degeneration (nAMD) and diabetic macular edema (DME) in Europe and Japan. As a reminder, Bayer exclusively markets Eylea outside the US, while Regeneron is responsible for US sales. Regeneron received FDA approval for Eylea HD for nAMD, DME, and diabetic retinopathy (DR) in August 2023.
- As a differentiating factor among emerging competitors and biosimilars in the field, Mr. Oelrich emphasized that Eylea HD is the only drug approved for extended treatment intervals (up to five months) for nAMD and DME. During Q&A, Mr. Oelrich said that Eylea HD has received positive responses from HCPs, especially given larger dosing intervals, and therefore feels “bullish” about future progression. Additionally, he noted that Eylea HD pre-filled syringe (PFS) is now indicated and available for nAMD and DME, approved in September 2024. In the US, Regeneron shared in 3Q24 that it would launch the PFS by mid-2025.
- Non-steroidal MRA Kerendia (finerenone) has strong momentum across chronic kidney disease and heart failure. While Kerendia initially showed a slower uptake, Mr. Oelrich highlighted the strong growth of Kerendia in CKD and T2D, particularly in the US and China. We think the progression will go just one direction, up! Kerendia’s revenue totaled $125 million in 3Q24, up 91% from 3Q23 and up 10% sequentially, and has shown impressive growth since its launch in August 2021. In 2025 and beyond, Mr. Oelrich expects further uptake, particularly accelerating the CKD/T2D indication and seeking broader coverage for the whole CKD spectrum.
- Mr. Oelrich referred to the groundbreaking results of the FINEARTS-HF (n=6,001) trial presented at ESC 2024, in which finerenone demonstrated a 16% relative risk reduction of the primary composite outcome of total heart failure outcomes and cardiovascular death over 32 months among patients with HFmrEF or HFpEF. Application for a heart failure indication has been submitted in the US and China. In 3Q24, Bayer announced that the company is preparing to launch Kerendia for heart failure in 2026.
- The Lancet published a prespecified analysis of the FINEARTS-HF trial of Kerendia in new-onset diabetes in people with HFmrEF or HFpEF. During a median duration of a follow-up of 31 months, 7.2% of participants in the finerenone group and 9.1% of the placebo group developed new-onset diabetes. Finerenone significantly reduced the hazard ratio of new-onset diabetes by 24%. These results were similar in sensitivity analyses, in which new-onset diabetes was expanded to include initiation of SGLT-2 inhibitor treatment with diabetes as an indication, restricted to A1c measurements, and limited to new initiation of glucose-lowering treatments. These results highlight the efficacy of finerenone in reducing the hazard of new-onset diabetes, representing a potential benefit of treatment in this population.
6. Biomea: Refined pipeline focus on diabetes and obesity with icovamenib, a first-in-class menin inhibitor
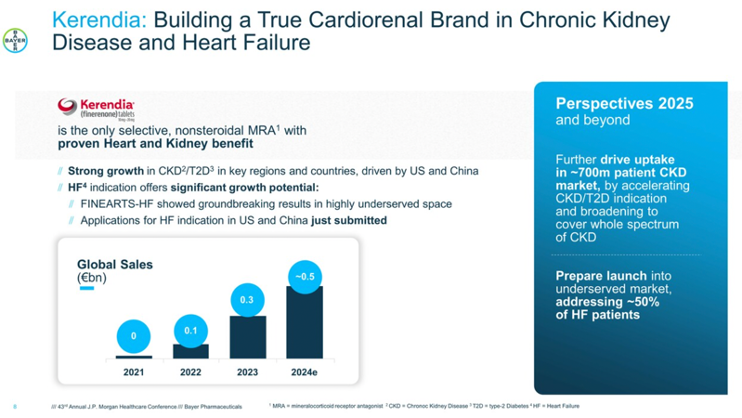
CEO Mr. Thomas Butler highlighted several updates and expectations for Biomea’s icovamenib (BMF-219), a potent and selective covalent menin inhibitor. On Monday, Biomea announced that the company will become a diabetes and obesity treatment company based on promising results of recent clinical trials. Dedicating internal resources to diabetes and obesity, Biomea plans to conclude its studies exploring icovamenib in oncology and explore partnerships to further advance its oncology assets. Most recently, in December 2024, Biomea announced positive topline results of the phase 2 COVALENT-111 study of icovamenib in T2D – full 52-week results are expected in 2H25. Among patients who completed dosing and had suboptimal diabetes management at baseline with one or more treatments, icovamenib showed an A1c reduction of 0.36%, a reduction that would be considered meaningful regardless of the baseline A1c (and more meaningful the lower the A1c). In the study analysis with T2D phenotypes, icovamenib showed further A1c reduction in people with severely insulin-deficient diabetes (SIDD) and those treated with GLP-1 RAs. Additionally, the prespecified SIDD subgroup performed as well as other FDA-approved treatments for T2D (e.g., Ozempic, Mounjaro, Jardiance, Januvia). Biomea plans to discuss results with the FDA in 1H25 and advance icovamenib into the phase 2/3 COVALENT-311 trial in people with SIDD. Icovamenib is also being studied in people with T1D in the open-label portion of the COVALENT-112 trial, which is expected to complete in 2H25.
- Aims to address challenges with current standards of care. Mr Sibold quoted data in his talk asserting that up to 50-75% of people on GLP-1 receptor agonists discontinue them in 12 months, that nearly 70% discontinue SGLT-2 inhibitors at 18 months, and nearly 75% discontinue DPP-4 inhibitors at 18 months. Factors that contribute to discontinuation include side effects, aversion to injection, inability to meet glycemic targets, cost, and affordability. Mr. Butler said icovamenib has the potential to address these challenges, as it provides durable treatment impact on beta cell function and incretin effect.
- Icovamenib to focus on two key patient segments. First, the development of icovamenib targets people with SIDD, which represents 18% of the T2D population according to Biomea Fusion. Mr. Butler explained that the SIDD population has the lowest insulin production of all adults with T2D, representing the highest unmet need with high all-cause mortality and worst CV outcomes. Icovamenib also targets patients on GLP-1 RA treatment, including those who struggle to meet glycemic targets with GLP-1 RAs alone.
- Significant potential for combination treatment. Mr. Butler shared that icovamenib has demonstrated enhanced responsiveness of human donors to Lilly’s tirzepatide and Novo Nordisk’s semaglutide. Icovamenib also enhanced the responsiveness of human donor islets to Lilly’s orforglipron and BMF-650, Biomea’s oral small molecule GLP-1 RA that is in preclinical development. Given these results that were also discussed at WCIRDC 2024, Biomea plans to meet with the FDA to discuss a phase 2 trial of icovamenib and GLP-1 RA combination in T2D.
7. Daiichi Sankyo: Nothing new from Dr. Sunao Manabe on pain
Daiichi Sankyo’s President and CEO Dr. Sunao Manabe provided updates on the company’s oncology portfolio, focusing on its proprietary antibody-drug conjugate (ADC) platform. Of note, the company’s breast cancer drug Enhertu won the 2024 Galien USA Award – which Dr. Manabe compared to the Nobel prize in biomedical innovations – for best biotechnology product.
- Neuropathic pain agent Tarlige was not discussed, nor were DPP-4 inhibitor Tenelia or Canalia, a combination DPP-4 and SGLT2 inhibitor. Tarlige remains part of the company’s “profit growth for current business and products” strategy in the five-year business plan – it was first approved in April 2019 for peripheral neuropathic pain and received an indication expansion in March 2022 to include central neuropathic pain. In May 2023, Daiichi Sankyo launched a disintegrating tablet formulation that dissolves on the tongue, particularly beneficial for people for whom swallowing tablets is difficult.
8. Ionis: Preparing for the launch of Tryngolza (olezarsen) as the first FDA-approved treatment for familial chylomicronemia syndrome
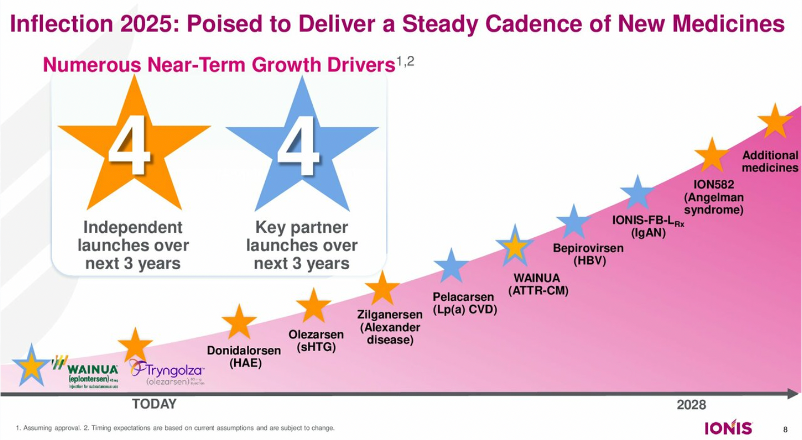
CEO Dr. Brett Monia presented Ionis’ progress, especially with the recent approval of Tryngolza (olezarsen) for familial chylomicronemia syndrome (FCS) – the company’s first independent product launch. Dr. Monia emphasized that Ionis is pioneering the field of oligonucleotide therapeutics with a rich history of discovering and developing RNA-targeted treatments. When he delivered his first presentation at JPM 2020 as the newly appointed CEO, Dr. Monia outlined four key objectives for the company: (i) prioritize and advance a wholly owned pipeline; (ii) build commercial capabilities; (iii) expand and diversify technology; and (iv) independently bring treatment to patients. Over the past few years, under Dr. Monia’s leadership, Ionis has, he said, advanced 10 treatments into mid- and late-stage development, established an innovative and scalable commercial organization, and delivered positive clinical results and product launches. Over the next three years, Ionis expects four other independent launches and four key partner launches.
- 2025 will include several key readouts. Ionis’ pipeline also includes pelacarsen [Lp(a)-lowering therapy] in the phase 3 HORIZON program (n=8,323), which is expected to complete in May 2025[2].
- Ionis plans to expand therapeutic opportunities. Looking forward, Ionis plans to expand research and investment in established areas, including cardiovascular disease. Ionis also hopes to expand toward new fields, including pulmonary and renal health.
9. J&J: CEO Mr. Joaquin Duato discusses elements for success with a diversified portfolio, increasing attention for MedTech and Innovative Medicine
This morning, CEO Mr. Joaquin Duato reinforced J&J’s progress for both MedTech and Innovative Medicine, outlining key updates on its ever-expanding diversified portfolio. Looking at long-term growth, Mr. Duato explained that J&J has two elements for success: (i) clear purpose with direction; and (ii) model that prioritizes diversified healthcare. Mr. Duato said that J&J continually works toward spanning capabilities to address needs, and looking forward, he mentioned several exciting opportunities to expand growth. Notably, Mr. Duato expressed confidence in meeting the projected 5-7% growth, especially in Innovative Medicine, a portfolio across therapeutic areas of oncology, immunology, neuroscience, cardiology, and ophthalmology. Given the intersection between diabetes and heart, brain, and eye health, we’re curious how J&J could leverage its existing pipeline for broader outcomes.
- J&J strengthens focus on neuroscience. Just today, J&J acquired Intra-Cellular Therapies for $14.6 billion to expand the development and production of treatments for central nervous system disorders. With this partnership, J&J adds Intra-Cellular Therapies’ Caplyta (lumateperone), a once-daily oral treatment for adults with schizophrenia, as well as depressive episodes associated with bipolar disorders. The acquisition also includes ITI-1284, a promising phase 2 candidate for generalized anxiety disorder and Alzheimer’s disease-related psychosis and agitation. Mr. Duato emphasized that this partnership helps strengthen J&J’s current focus areas.
- MedTech continues to strive toward strong growth. Reinforcing his remarks from last year, Mr. Duato said that J&J has made significant progress in robotics and digitals to advance the MedTech sector. In fact, J&J has met its goals with MedTech to grow in the top market and become more competitive, with 50% of the markets growing more than 5%. Mr. Duato partly attributed this growth to J&J’s acquisition of medical device company Abiomed, which was announced in November 2022 and completed in December 2022. J&J is particularly awaiting approval of the Impella heart pump, which finished clinical trials in 2023, for broader patient populations (e.g., people at risk for heart attack).
10. Lilly: Continued momentum with Mounjaro and Zepbound, advancing incretin treatments for diabetes, obesity, and beyond; upcoming “big assets” include oral GLP-1 orforglipron and “triple G” retatrutide
In an afternoon fireside chat, CEO Mr. David Ricks called 2024 a highly impressive year, with Mounjaro (tirzepatide for diabetes) and Zepbound (tirzepatide for obesity) showing robust growth, and he expressed confidence for this trend to continue (see webcast and presentation). Mr. Ricks discussed Lilly’s updates on the 2024 and 2025 revenue guidance published that morning. Lilly expects 2024 full-year worldwide revenue to be approximately $45 billion (+32%). In 4Q24, Lilly expects revenue of approximately $13.5 billion (+45%), including $3.5 billion for Mounjaro and $1.9 billion for Zepbound. Mr. Ricks acknowledged that the expected 2024 revenue is $400 million (about 3%) below the guidance range announced in 3Q24. Mr. Ricks said that while the US incretin market grew 45% compared to 4Q23, Lilly’s previous guidance had anticipated even faster growth. While 2024 revenue was lower than expected, Mr. Ricks highlighted several milestone achievements, including readouts of tirzepatide in obstructive sleep apnea (OSA) and diabetes prevention, as well as the shortage resolution announced by the FDA in December 2024. With Lilly’s 4Q24 earnings on February 6, 2025, we’re looking forward to hearing more detailed updates.
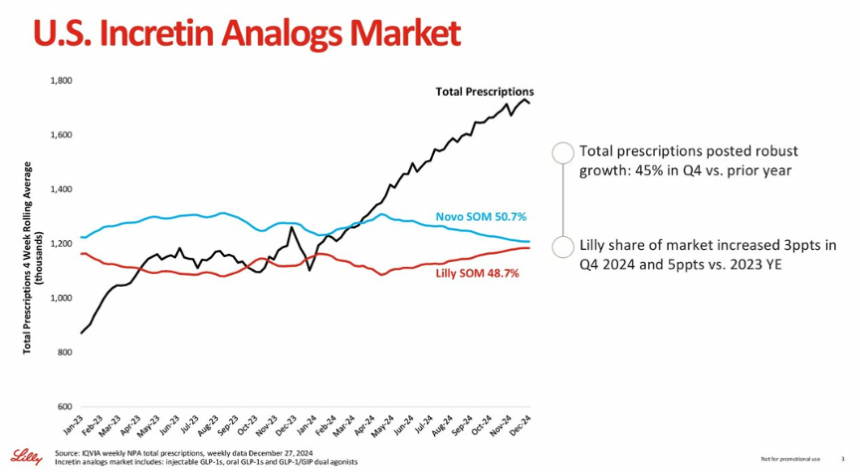
- Lilly expects to produce at least 60% more scalable doses of incretins in 1H25 compared to 1H24. Given robust sales of Mounjaro and Zepbound, Lilly plans to bring additional manufacturing capacity. Indeed, last year’s JPM 2024 was held when Zepbound had only been on the market for about four weeks, and since then, Zepbound has marked significant penetration in the obesity market. In particular, Zepbound demonstrated strong underlying prescription growth with broad commercial formulary access through employer opt-ins. As of October 1, 2024, Zepbound achieved around 87% of commercial insurance coverage in the US, and 50% of employers have opted into anti-obesity medicine coverage.
- Tirzepatide has promising potential to “unlock” new categories again and again. Beyond diabetes and obesity, Mr. Ricks said tirzepatide has shown significant efficacy in clinical trials for expanded indications. Most recently, in December 2024, the FDA approved Zepbound for moderate-to-severe obstructive sleep apnea and obesity. This approval reflected results on phase 3 SURMOUNT-OSA trial, in which tirzepatide conferred 20% weight reduction and prevented 25 breathing interruptions per hour of sleep. Following this approval, the CMS announced last week that Medicare will cover Zepbound for this expanded indication. Aside from OSA, in the phase 3 SURMOUNT-1 trial, tirzepatide conferred a 94% reduction in the risk of developing T2D in adults with prediabetes and overweight or obesity.
- Upcoming readouts include results for Lilly’s potential “big assets,” including orforglipron (oral GLP-1 RA) and retatrutide (GLP-1/GIP/glucagon triple RA). On orforglipron, Mr. Ricks said meaningful data could significantly impact people who prefer oral formulations. Given that Lilly plans to use different assets that already exist, he imagines scaling to be “multiples” above injectables. With successful results, Lilly plans to first launch orforglipron in the US and progress with “no limit,” as it will be “easy to make and easy to access.” On retatrutide, Mr. Ricks imagines the additional component of a glucagon RA will help those who need to achieve more weight loss and address comorbidities. Aside from orforglipron and retatrutide, Mr. Ricks highlighted Lilly’s expansive portfolio of nine other weight loss candidates.
- Remaining questions involve the duration of treatment, adherence, and maintenance. Mr. Ricks said that people on Mounjaro and Zepbound not only experience clinical outcomes fast but also “feel better quickly.” He said he imagines these treatments to be a “long-duration” therapy and might be even longer than other chronic treatments. Furthermore, Mr. Ricks addressed the importance of weight maintenance, especially among people who successfully achieve weight loss targets. On adherence, Mr. Ricks said it would be important to understand tolerability, as some people can’t tolerate even low doses of GLP-1 RAs.
- Increasing coverage and accessibility remains an important value. In November 2024, the Biden administration proposed a new health policy to expand Medicaid and Medicare coverage of obesity medications. Mr. Ricks said it’s unclear how the incoming Trump administration would implement and finalize this goal with competing priorities. Nevertheless, Mr. Ricks expressed encouragement that the administration has been receptive to conversations and discussions, especially in comparison to the past.
- It was interesting to hear toward the end of the discussion the areas on which Lilly reinforced that it will be particularly focused:
- Increasing transparency on pricing and distribution;
- Addressing the Inflation Reduction Act (IRA); and
- Expanding obesity coverage.
Focusing on the latter, Mr. Ricks said that while diet and exercise are crucial for prevention, these interventions are ineffective treatments for most – and, of course, we know that all can benefit from better heart, kidney, and liver health stemming from GLP-1 and multi-agonists. Mr. Ricks said that Lilly hopes to work with the new US administration to increase broad population targets, changing the trajectory of healthcare and continually impacting patient outcomes.
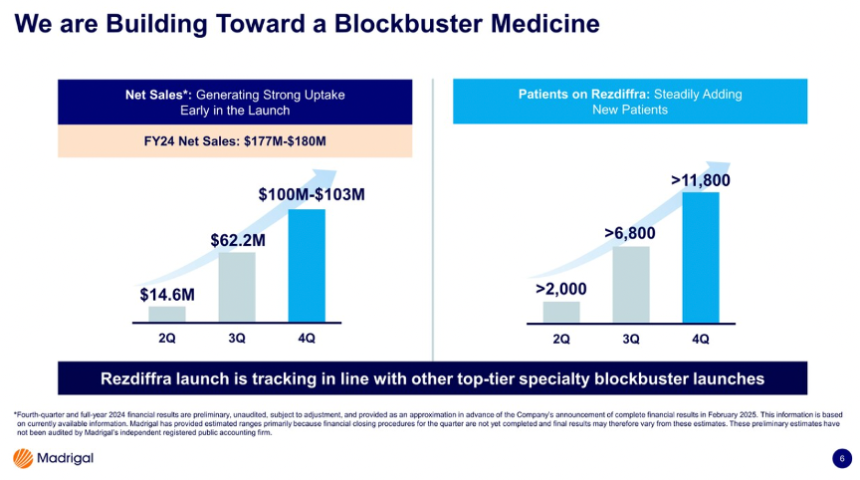
11. Madrigal: “Foundational MASH treatment” Rezdiffra sales total $180 million in the first nine months, with over 11,800 patients treated
CEO Mr. Bill Sibold highlighted Madrigal’s Rezdiffra (resmetirom), the first FDA-approved treatment for MASH, as a highly competitive and successful therapy for continual growth. Overall, this was one of the strongest presentations we saw at JP Morgan, and we look forward to seeing the company expand. Compared to where the company stood at JPM 2024, Madrigal has achieved multiple milestones in just one year.
- In March 2024, Rezdiffra received FDA approval for people with MASH with moderate to advanced liver fibrosis (F2 and F3), based on results from the phase 3 MAESTRO-NASH trial (n=966). Mr. Sibold emphasized that this approval has helped Rezdiffra secure the position as the “foundational” MASH treatment. According to Madrigal’s preliminary financial published on Monday, Rezdiffra totaled $100-103 million in 4Q24 and $166-180 million in 2024 – of note, the full-year result only includes nine months of Rezdiffra’s launch. Previously, Rezdiffra sales totaled $62.2 million in 3Q24 and $14.6 million in 2Q24.
- Furthermore, Mr. Sibold shared that more than 11,800 patients are on Rezdiffra, a significant growth from over 6,800 patients in 3Q24. Madrigal ended 2024 with $931 million in cash, which is expected to support continual launch in the US and upcoming commercialization in the EU.
- Mr. Sibold emphasized that Rezdiffra’s substantial growth reflects the critical need for MASH treatments. He called MASH a serious liver disease, which is expected to become the leading cause of liver transplants in the US. In fact, MASH represents the leading cause of liver-related death and liver transplants. Mr. Sibold shared alarmingly data that once people pass F2 and F3, they have a 10 to 17-fold higher chance of liver mortality. While MASH was previously seen as a “graveyard of drug development,” Mr. Sibold shared excitement that Rezdiffra has demonstrated the potential to address this disease with high unmet needs. We will know more about how broad the Rezdiffra application is when data emerges for patients with stage 4 fibrosis in several years (see more below).
- Rezdiffra’s foundational MASH treatment is attractive from multiple perspectives. Mr. Sibold highlighted Rezdiffra’s differentiated profile as a thyroid hormone receptor-β agonist that directly targets the liver. With this unique mechanism of action, Rezdiffra targets the underlying causes of MASH. Additionally, Rezdiffra provides highly effective treatment, as it improves liver stiffness in 91% of patients at three years. Finally, as a once-daily oral pill, Rezdiffra offers patients ease of administration, along with a well-tolerable profile.
- The market opportunity is very large, regardless of competitors entering the landscape. Mr. Sibold emphasized that given Rezdiffra is the first and only MASH treatment, and that it provides an attractive market of 315,000 people diagnosed with F2 and F3 who are seeing a specialist that Madrigal could call on. Mr. Sibold estimated that Rezdiffra has treated <4% of the target population to date, and therefore, there’s plenty of market opportunity ahead.
- During Q&A, Mr. Sibold expressed confidence in Madrigal’s success and leadership position, even with (some may say especially with) the potential approval of GLP-1 RAs in MASH. While we aren’t sure of the market that they will target, we imagine it will include those with stage 1 fibrosis. In fact, Mr. Sibold said GLP-1 RAs entering this landscape will help expand the market and further increase awareness, screening, diagnosis, and treatment. Furthermore, he noted high discontinuation rates of GLP-1 RAs pose a challenge compared to Rezdiffra, which has high tolerability. Mr. Sibold also pointed to opportunities for combination therapies, as 25% of patients of Rezdiffra are on GLP-1 RAs, and 50% have been previously exposed to GLP-1 RAs.
- F4 indication has the potential to double Rezdiffra’s opportunity. Mr. Sibold reminded that the ongoing MAESTRO-NASH-OUTCOMES trial (n=845) of Rezdiffra in patients with cirrhosis (F4) has completed enrollment in October 2024, as shared in 3Q24. This trial is of great interest, as positive outcomes could lead to expanded indications for Rezdiffra, potentially doubling its target patient population. This trial was initiated in August 2022 and is expected to complete in January 2027. Alarmingly, Mr. Sibold said that people with F4 have a 42-fold increased risk of liver mortality.
- During Q&A, Mr. Sibold addressed a question regarding the potential of Rezdiffra in delaying or preventing MASH. Given the efficacy of Rezdiffra in F2 and F3, Mr. Sibold said that this is the population Madrigal is focused on. He said, “That’s where the benefit lies … where the values are with the highest unmet need.” He suggested that perhaps GLP-1 RAs could be indicated for prevention.
- Madrigal awaits Rezdiffra’s approval and commercial launch in the EU in 2025, country by country. As emphasized in 3Q24, Madrigal plans to commercialize Rezdiffra in the EU pending EMA regulatory approval, expected in mid-2025. In the discussion, Mr. Sibold announced plans to start commercialization in the EU one country at a time, starting with Germany. Overall, Mr. Sibold expressed high optimism, as Rezdiffra has already been recommended as a first-line treatment for MASH in the joint EASL-EASD-EASO clinical practice guidelines for MASH, presented at EASL 2024. With 125 trial sites across the EU, Mr. Sibold said several countries are familiar with Rezdiffra and have expressed significant interest.
- Mr. Sibold hopes to build a pipeline to secure long-term growth. He emphasized the high efficacy and quality of Rezdiffra, explaining that now, the company aims to build a pipeline over the next several years to fortify Madrigal’s leadership market position in MASH. When asked about the specifics of a potential pipeline during Q&A, Mr. Sibold suggested exploring the next best mechanism of action or combination treatments that enhance Rezdiffra’s efficacy. In the meantime, of course, Madrigal plans to continue positioning Rezdiffra as a foundational treatment. We imagine this will continue to be quite effective given the major needs in the field and no competition now.
- Diagnosing and screening MASH as an emerging field. As the field is moving away from using liver biopsies to diagnose MASH (at least later stage MASH, meaning stage 2 or stage 3), Mr. Sibold expects several developments in this arena. He said, for example, that more interest has emerged in non-invasive tests (NITs), especially with the approval of Rezdiffra. He said it would be important to figure out the best sequence and combination of NITs to improve diagnoses. Additionally, he referred to the FIB-4 score, which the ADA called the “most cost-effective” strategy for identifying people at risk for MASH in the 2025 Standards of Care.
12. Merck: Diversifying portfolio in diabetic macular edema; upcoming key readouts with PCSK9 and GLP-1 RA
Reflecting on the past three years since his appointment as CEO, Mr. Robert Davis said Merck has delivered impressive “breadth and depth” to the field beyond oncology. Alongside consistent revenue growth, Merck has expanded its portfolio to increase focus on other therapeutic areas – compared to nine assets in 2021, Merck had 26 candidates in 2024. With a growing pipeline, Mr. Davis affirmed Merck’s ability to continually grow and impact patient outcomes into the next decade and beyond.
- As a testament to a diversifying portfolio, Mr. Davis recalled Merck’s acquisition of London-based EyeBio in May 2024. Following this acquisition, Merck announced the initiation of the phase 2b/3 BRUNELLO trial (960), evaluating Restoret (MK-3000) for diabetic macular edema (DME) in September 2024. The BRUNELLO trial will enroll people with T1D or T2D and DME, and it is expected to complete in December 2027. With Restoret, Merck aims to address current shortcomings with DME with anti-VEGF injections (Eylea or Vabysmo).
- On key readouts, Mr. Davis noted several upcoming results in the cardiometabolic portfolio that will be shared in 2025. Oral PCSK9 enlicitide decanoate (previously called MK-0616) is in a phase 2b trial that is expected to complete in December 2025; this timeline is a bit delayed from the projected date of October 2025. Additionally, GLP-1/glucagon RA, MK-6024 (efinopegdutide) is in multiple phase 3 trials with hypercholesterolemia: (i) CORALreef Lipids (n=2,760), expected to complete in August 2025; (ii) CORALreef HeFH (n=270), expected to complete in April 2025; and (iii) CORALreef Outcomes (n=14,550), expected to complete in November 2029.
- While Mr. Davis did not share in today’s presentation, in December 2024, Merck announced a partnership with Hansoh Pharma for HS-10535, an investigational preclinical oral small molecule GLP-1 RA. This announcement reinforces Merck’s expanding portfolio into the obesity arena, in which Dr. Davis shared significant interest in recent earnings calls (1Q24, 2Q24, 3Q24).
13. Novartis: Innovation across four therapeutic areas, with significant progression of Entresto and Leqvio for heart failure and CVD
CEO Dr. Vas Narasimhan highlighted Novartis’ innovation across four therapeutic areas: (i) cardiovascular-renal-metabolic disease; (ii) immunology; (iii) neuroscience; and (iv) oncology. Dr. Narasimhan affirmed that Novartis continues to progress significantly, combining core therapeutic areas and technology platforms in four priority regions, including the US, China, Germany, and Japan. Over the past few years, Novartis has streamlined the pipeline by nearly 40% (155 projects in 3Q21 compared to 94 in 3Q24). With a more focused R&D, Dr. Narasimhan said the company has increased research resources by nearly 50% per project and 5% of developmental spending. Novartis also continues to leverage technology platforms to enhance the scalability of the pipeline and integrate a diverse, broad application.
- Novartis has eight in-market brands in the US with up to $8 billion peak potential. These products include Entresto (sacubitril/valsartan), an angiotensin receptor-neprilysin inhibitor (ARNI), for heart failure, which totaled $7.5 billion in annualized 3Q24 sales. Dr. Narasimhan emphasized that peak sales will reach more than $7 billion, assuming Novartis files for a loss of exclusivity (LOE) for Entresto in the US in mid-2025. Previously, in 3Q24, US sales grew by 25%, with total prescriptions (TRx) increasing by 20%, around 45,000 new-to-brand prescriptions (NBRx), and around 500,000 TRx per month. Another product that Dr. Narasimhan highlighted included Leqvio (inclisiran), an siRNA PCSK-9 inhibitor, which totaled $800 million in annualized 3Q24 sales, with a potential to reach more than $4 billion in peak sales.
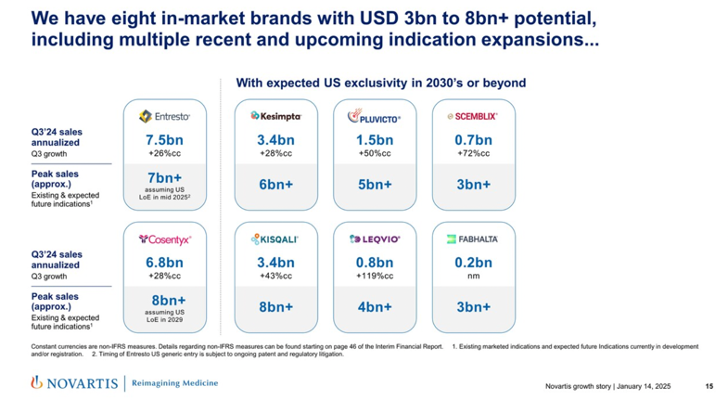
- Novartis continues to build a strong pipeline across cardio-renal-metabolic health to address high needs. In this sector, Novartis focuses on select disease areas, including heart failure and hypertension, atherosclerosis, arrhythmia, and acute kidney injury (AKI). Dr. Narasimhan shared that Entresto and Leqvio contribute to this therapeutic area as anchor assets, and he expressed excitement about advanced platforms like siRNA.
- Novartis has more than 15 submission-enabling readouts in the next two years. Dr. Narasimhan particularly showed excitement about readouts for Leqvio in primary prevention (VICTORION-1-PREVENT, expected April 2029) and secondary prevention (ORION-4 and VICTORION-2-PREVENT, expected in 2026 and 2027, respectively). Additionally, data for the Lp(a)-HORIZON trial (n=8,323) will highlight pelacarsen in major cardiovascular events in people with CVD (expected in May 2025).
14. OPKO Health: Dr. Elias Zerhouni highlights the promising potential of dual GLP-1/glucagon RA in development for oral and injectable formulations
President and Vice Chairman Dr. Elias Zerhouni shared updates on OPKO Health’s business and pipeline development across oncology, immunology, and metabolic diseases (see webcast and presentation slides). Reflecting on 2024, Dr. Zerhouni highlighted the successes of the ModeX proprietary technologies, acquired in May 2022, in advancing several candidates to the clinical stage for oncology and viral diseases. He also celebrated the growing revenue from OPKO Health’s two products, Ngenla (human growth hormone for pediatrics) and Rayaldee (calcifediol for stage 3 or 4 chronic kidney disease and vitamin D insufficiency). He also highlighted the growth of BioReference Health’s laboratories, which has positioned OKPO Health on a stronger financial footing.
- We were particularly excited to hear updates on the company’s incretin-based candidates: (i) GLP-2 RAs; and (ii) oxyntomodulin (OXM) analog, a dual GLP-1/glucagon RA for diabetes, obesity, and MASH. OPKO Health is developing these candidates in both injectable and oral tablet formulations – the latter in collaboration with Entera Bio since September 2023. Dr. Zerhouni noted that GLP-1 receptor mono-agonists are no longer “sufficient” for increasing metabolism and addressing comorbidities, expressing confidence in the potential of dual GLP-1/glucagon RA for obesity and advanced liver diseases (F2, F3, and F4). Dr. Zerhouni emphasized that glucagon RAs act on the liver and complement the limitations of GLP-1 RAs, and the preclinical data are promising. While not mentioned in today’s presentation, preclinical data of oral OXM in rodents and pigs are also favorable, as announced in September 2024. Dr. Zerhouni expects these candidates to advance to clinical trials by late 2025 or early 2026.
15. Pfizer: Dr. Albert Bourla expects phase 3 trial of once-daily oral GLP-1 in 2H25; confidence in capturing oral GLP-1 RA market
In a fireside chat, CEO Dr. Albert Bourla shared Pfizer’s vision for 2025 amid new administrations, product launches, and ongoing clinical trials. See webcast and presentation slides. Dr. Bourla began by reflecting on the successes of 2024. Pfizer secured 13 approvals across generic disorders, vaccines, and cancer treatments; achieved a $4 billion cost reduction; and restructured R&D, including leadership changes and organizational improvements, meeting five key priorities discussed last year at JPM 2024. Looking ahead with the new year, Dr. Bourla highlighted new values: (i) improve R&D productivity and operational efficiency through technologies like AI; (ii) maintain leadership in vaccines, cardiovascular disease, and migraine; and (iii) optimize capital allocation. Dr. Bourla also shared anticipation that the upcoming Trump administration will cause “radical changes” and believes that opportunities will outweigh the risks. Pfizer will engage proactively with the new administration to influence policy to improve innovation and access to medicine, given the intensifying competition with global players in biosciences like China. While much of the fireside chat focused on R&D and commercial updates for COVID-19 and cancer treatments, Dr. Bourla presented insights on Pfizer’s once-daily, oral GLP-1 RA, danuglipron.
- Phase 3 trial of once-daily danuglipron for overweight or obesity may begin in 2H25, if Pfizer can replicate results from discontinued phase 2b study of twice-daily formulation of danuglipron. As a reminder, Pfizer initially developed twice-daily danuglipron and planned to advance into phase 3 trials. However, due to safety and tolerability concerns, Pfizer discontinued the twice-weekly formulation in December 2023 and shifted its focus to the once-daily formulation. A dose optimization study of once-daily danuglipron was completed in 3Q24, and management aims to use data from phase 1 pharmacokinetic results, phase 2b data from the discontinued twice-daily formulation of danuglipron, and the recently completed dose-optimization study to select dosage for phase 3 trial – these results encompass over a total of 1,600 patients.
- On phase 3 study design, Dr. Bourla said Pfizer is considering various approaches, from full outcome studies to trials more focused on weight loss and will make decisions based on what will optimize results.
- While Dr. Bourla was cautious, Dr. Bourla expressed confidence that danuglipron, if successful, may become the second oral GLP-1 RA to reach the market. Of note, Novo Nordisk’s Rybelsus (semaglutide) is the only FDA-approved oral GLP-1 RA in the market, and Lilly’s orforglipron is currently in phase 3. Dr. Bourla added the significance of the growth potential for danuglipron as an oral formulation.
- On a broader metabolic portfolio, Pfizer is expanding its expertise in obesity with the acquisition of multiple candidates over the past year. The company is focusing on oral GLP-1 RAs rather than the injectable, as it views the latter as less viable from a business development perspective at this stage. Pfizer is exploring other mechanisms in both oral and injectable metabolic therapies, however, as the company has the capacity to develop and sell them.
16. Regeneron: Dr. Leonard Schleifer highlights strategies to increase Eylea HD uptake; phase 2 data of trevogrumab (anti-myostatin antibody) with semaglutide expected in 2H25
In this session, CEO Dr. Leonard Schleifer and CSO Dr. George Yancopoulos provided company updates, primarily focusing on its clinical pipeline (see webcast and presentation slides). Reflecting on the company’s history, Dr. Schleifer attributed Regeneron’s success to its differentiated technologies, scientific expertise, and, most important, its robust pipeline of clinical candidates. Dedicated efforts have led to breakthroughs like Eylea (aflibercept 2 mg), and both Drs. Schleifer and Yancopoulos were eager to share candidates of the current clinical pipelines – including in cancer, COPD, obesity, and allergy – which have significant potential for future breakthroughs.
- On obesity and metabolic health, Dr. Yancopoulos pointed out the suboptimal quality of weight loss with current GLP-1 RAs. Studies have shown that 30% of weight loss is from muscle, and regained weight from GLP-1 RA discontinuation is entirely fat. Cycling on and off GLP-1 RAs could seriously damage the human body. Regeneron’s trevogrumab (anti-myostatin) addresses this challenge by minimizing muscle loss or even increasing muscle mass when used in combination with semaglutide. A phase 2 study investigating this combination therapy, with and without garetosmab (anti activin A), is currently ongoing. As announced in the 3Q24 call, the study is fully enrolled, and data is expected in 2H25.
- Management also expressed interest in developing GLP-1 RA-independent mechanisms, such as ones focused on muscle growth and increasing muscle metabolism to overcome obesity.
- In ophthalmology, Eylea and Eylea HD (aflibercept 8 mg) continue to lead the anti-VEGF class, generating $6 billion in full-year revenue in 2024 (up 1% from 2023), with Eylea HD contributing 20% of the total. To further drive the uptake of Eylea HD, Regeneron completed regulatory submission for a pre-filled syringe, with a US launch anticipated by mid-2025. The company also plans to submit a supplemental biologics license application (sBLA) in early 2025 for retinal vein occlusion (RVO), which accounted for 17% of Eylea sales in 2024, and seeks to maximize dosing flexibility through extended dosing intervals up to 24 weeks for wet age-related macular degeneration (wAMD) and diabetic macular edema (DME) and every-four-week dosing for wAMD, DME, and diabetic retinopathy (DR). Management also highlighted goals to establish Eylea HD as the new standard of care.
- On overall R&D, Regeneron has launched the Regeneron Genetic Medicines (RGM) program, a therapeutic platform that allows customized analysis of genetic data to pursue targeted treatments aiming to improve patient outcomes. Regeneron shares this platform with Alnylam and Intellia.
16. Roche: Continued clinical progress in ophthalmology and cardiovascular-renal-metabolic portfolio; phase 1b trial of anti-TL1A therapy in MASH to initiate in 1Q25
Ms. Teresa Graham (CEO, Roche Pharmaceuticals) provided updates on clinical development in Roche’s five priority therapeutic areas: (i) oncology/hematology; (ii) immunology; (iii) neurology; (iv) ophthalmology; and (v) cardiovascular, renal, and metabolic health (CVRM). While we had not expected to not hear directly from Roche’s CEO, Dr Thomas Schinecker, this crowd certainly appreciated hearing from the very popular head of Roche Pharma. It was valuable for those new to Roche (and a good reminder for all) when Ms. Graham stressed that Roche employs a “one asset team approach” that examines how to maximize each molecule’s value across therapeutic areas, citing the company’s incretin candidates as examples of assets with potential efficacy for other therapeutic areas in addition to CVRM, such as neurology. Ms. Graham also described Roche’s approach to resource allocation in R&D development that has enabled the company to streamline its pipeline: “The Bar.” Ms. Graham said “The Bar” – which considers an asset’s addressable unmet needs, potential efficacy, differentiated characteristics, and possible economic and patient value – is applied to every asset in the pipeline. Through this approach, Ms. Graham said Roche culled about 30% of its pipeline, freeing resources to accelerate development of more promising candidates.
- Ophthalmology: Ms. Graham highlighted continued progress in its ophthalmology portfolio across several assets.
- Susvimo: Roche’s surgically-placed eye implant Susvimo (ranibizumab) awaits regulatory approval in diabetic macular edema (DME) and diabetic retinopathy (DR) in the US after filing in April 2024. Ms. Graham said regulatory approval is expected this year. Roche is also conducting a 36-week phase 3 trial for Susvimo for neovascular or “wet” age-related macular degeneration (nAMD), which is expected to complete at the end of 2026. Ms. Graham said EU submission of Susvimo for nAMD is expected in 2025. As of 3Q24, DME submission will follow in the EU in 2026.
- Vabysmo: Roche obtained EMA approval for the Vabysmo (faricimab) 6.0 mg single-dose prefilled syringe (PFS) in December 2024 for nAMD, DME, and retinal vein occlusion (RVO). This followed FDA approval for Vabysmo PFS in July 2024 for these conditions. Ms. Graham said that Vabysmo, which remains one of the company’s top growth drivers, will continue to drive significant revenue generation for Roche in 2025, particularly as uptake of the PFS increases. She said that Roche is very confident Vabysmo will become standard of care.
- Vamikibart: Roche’s intravitreally injected IL-6 targeting therapy vamikibart continues to progress in clinical trials for uveitic macular edema (UME) and DME. Roche’s phase 2 program for DME, which includes the BARDENAS and ALLUVIUM trials, remains ongoing. Ms. Graham did not disclose when results from these studies may be expected. Data is expected from Roche’s phase 3 program in UME, which includes the SANDCAT and MEERKAT trials, in 2025. While most patients respond to anti-VEGF therapy, Ms. Graham emphasized that some individuals do not; thus, vamikibart could serve as a valuable alternative for this unmet population.
- Cardiovascular, renal, and metabolic health: Ms. Graham highlighted several upcoming clinical trial milestones in its CVRM portfolio.
- Hypertension: Ms. Graham said a phase 3 CVOT of zilebesiran, an angiotensinogen inhibitor, for hypertension will be initiated later this year. The trial design will be informed by results from the phase 2 KARDIA-3 trial (n=390) in people at high risk for cardiovascular disease with CKD. Ms. Graham characterized this therapy as possessing “best-in-disease potential.”
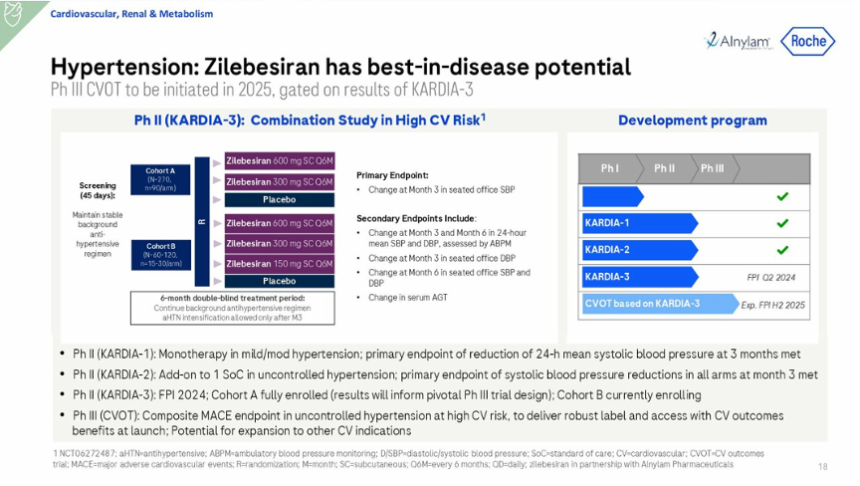
-
- Obesity: Ms. Graham briefly surveyed Roche’s obesity pipeline and upcoming milestones. Phase 1 data of CT-996 in T2D is expected sometime this year after positive phase 1 results in obesity without T2D were presented at EASD 2024. Phase 2 data of CT-868 in T1D and obesity is expected in 2H25. Ms. Graham said Roche continues to explore combination therapy approaches for its incretin pipeline, including with the myostatin antibody GYM 329 or PYY analog CT-173. A phase 1 trial of CT-173 in obesity will be initiated this year.
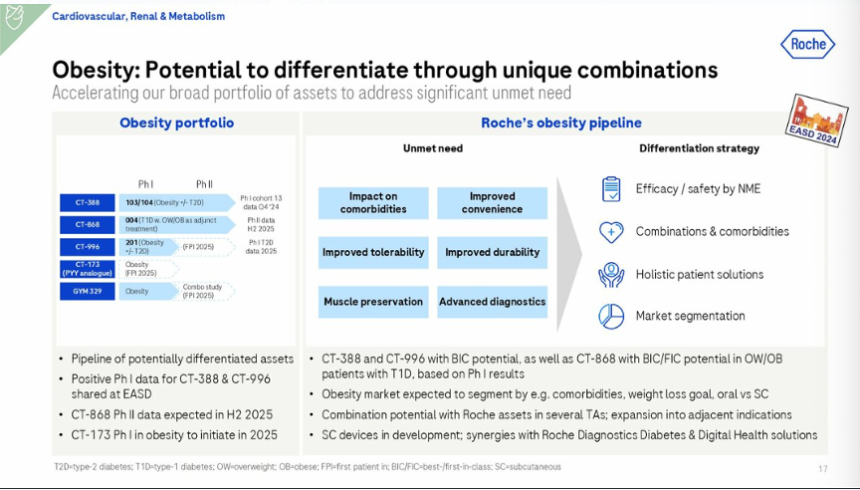
- MASH: Roche announced that it will initiate a phase 1b trial of RG6631, an anti-TL1A antibody, in MASH in 1Q25. The protein TL1A activates the DR3 receptor, which stimulates downstream inflammatory and fibrotic mechanisms. Due to previous research implicating TL1A in the pathogenesis of fibrotic conditions, TL1A inhibition could be applicable for slowing MASH pathogenesis. Ms. Graham did not provide significant detail on the trial design, and we’ll look forward to Roche’s 4Q24 earnings report on January 30 when we hope more details are shared.

17. Sanofi: Strides to become the world leader for immunology; regulatory decision of Tzield in China expected in 2H25
In a packed room, CEO Mr. Paul Hudson, EVP of R&D Dr. Houfman Ashrafian, and CFO Mr. François-Xavier Roger discussed Sanofi’s business and pipeline updates - see webcast and presentation slides here. The company delivered strong growth in sales (+11%) in 2024, with nine newly launched drugs generating €2.9 billion ($3 billion). Reinforcing statements from JPM 2024, management expressed ambition to become the world’s leading immunology company. Mr. Hudson particularly celebrated the outstanding performance and increasing global uptake of Beyfortus (nirsevimab-alip), a monoclonal antibody launched in 2023 that prevents respiratory syncytial virus (RSV) infection in newborns. This drug has saved countless infants in early adopter countries like Spain and France, which saw over 90% reduction in hospital visits for newborns. Management also shared progress in setting a lower price (at a premium vaccine price rather than the price of a small molecule drug) to prioritize universal access. Mr. Hudson said, “We’re in healthcare… All infants, that was our goal.” Other discussion topics included pipeline updates on other candidates, including non-mRNA vaccines, eczema, asthma, COPD, and hemophilia. Dr. Ashrafian was particularly excited about the launch of Dupixent (depilumab) for COPD in 2024, which expands to the long list of indications, including atopic dermatitis, asthma, and chronic rhinosinusitis with nasal polyps.
- While Mr. Hudson did not discuss diabetes or Tzield in today’s presentation, we look forward to the regulatory decision for Tzield (teplizumab) in China, which is expected in 2H25. As of Sanofi’s 3Q24 update, 373 people have been infused with Tzield since its approval in November 2022, and quarterly sales totaled $17 million (+67% YoY; +36% Q/Q). Sanofi is currently discussing with the FDA and EMA about expanding Tzield’s indication, following results of the PROTECT (n=328) trial, which showed that children and adolescents with recent onset stage 3 T1D treated with Tzield have preserved beta-cell function. We’ll await any updates on the first disease-modifying therapy this year.
- On overall business strategies, Sanofi’s management expressed continued commitment to R&D, focusing on disciplined lifecycle management of clinical candidates (across development, commercialization, and geneticization) and first-in-class and best-in-class innovations. Management also expects that the US Inflation Reduction Act (IRA) will have little impact on Sanofi due to minimal exposure to IRA-related changes through 2030.
18. Terns Pharmaceuticals: High confidence in oral GLP-1 RA TERN-601, compared to competitors; phase 2 data expected in 2H25
CEO Ms. Amy Burroughs delivered promising updates on its pipeline in metabolic diseases (see webcast, which also included progress in oncology). In particular, Ms. Burroughs highlighted Terns’ commitment to develop potential best-in-class molecules in blockbuster indications. The company is in a strong financial position with ~$373 million in cash and runway into 2028, supporting several clinical developments.
- Ms. Burroughs highlighted positive phase 1 results (n=36) of oral, once-daily GLP-1 RA TERN-601. Announced in September 2024, TERN-601 demonstrated statistically significant, dose-dependent weight loss of up to 5.5% over 28 days with a favorable safety and tolerability profile. Ms. Burroughs emphasized that the phase 1 trial implemented the fastest titration scheme compared to other oral GLP-1 RA studies, yet TERN-601 was very well tolerated with no severe adverse events or discontinuation. She believes this profile provides a significant competitive edge against injectable GLP-1 RAs on the market, which has a high discontinuation rate. “Physicians and patients are looking for a drug that patients [can] stay on for the rest of their life,” she said, noting that tolerability may be a more crucial factor than smaller differences in efficacy. She added that the weight loss curves do not show any signs of plateaus, suggesting potential to achieve greater weight loss in longer trials. Terns will initiate a 12-week part A of the phase 2 trial in early 2Q25 to optimize dose titration, with data expected in 2H25. Part B of the phase 2 trial will be conducted for a longer duration and will incorporate learnings from Part A. See below for Terns’ comparison of TERN-601 to other oral GLP-1 RAs in development.
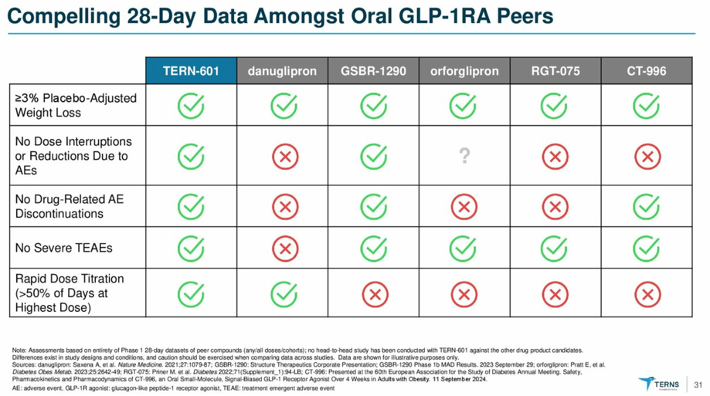
- Terns’ pipeline also includes: (i) TERN-501, a THR-β agonist; and (ii) TERN-800, a GIP RA. At JPM 2024, Ms. Burroughs announced plans to pause TERN-501 in MASH to explore its potential in combination therapy for other metabolic conditions. While phase 2a study in MASH demonstrated safety and dose-dependent reductions in liver fat content at Week 12, the company made this decision, given the cost to pursue an indication in MASH which requires paired biopsy studies. Preclinical data suggested TERN-501’s potential to enhance weight loss effects in combination therapies, as well. We are curious whether TERN-800 began IND-enabling studies for overweight and obesity in 2024; this was the goal according to the company’s JPM 2024 presentation.
19. Vertex: Suzetrigine to launch for acute pain following PDUFA date in January 2025; T1D phase 3 trial of VX-880 (now named zimislecel) to complete enrollment and initial dosing in 2025
In a packed room, CEO Dr. Reshma Kewalramani delivered a comprehensive presentation with business and pipeline updates in several disease areas (webcast, presentation slides). Dr. Kewalramani highlighted Vertex’s broad focus on diseases with clear causal human biology and validated targets, which have led to successful R&D and approvals in cystic fibrosis and sickle cell disease. Beyond these successful areas, Dr. Kewalramani reaffirmed Vertex’s focus on pain and T1D programs that continue to make significant strides with new data. Vertex will continue diversifying the pipeline with additional disease areas and geographic regions to maximize growth and address unmet needs.
- On pain, NaV1.8 pain signal inhibitor suzetrigine (VX-548) is ready to be launched for moderate-to-severe acute pain, following its PDUFA date on January 30, 2025. Dr. Kewalramani emphasized that this potential approval could address significant needs among 80 million people in the US currently suffering from acute pain. Among this population, 40 million are prescribed opioids, contributing to 85,000 new cases of opioid use disorder (OUD) each year and $10-20 billion annual costs for OUD management. Suzetrigine, which conferred a 3.4-point (or 50%) reduction in the numeric pain rating scale (NPRS) score in phase 3 programs, has the potential to fill the gap between weaker, non-addictive anti-inflammatory treatments and stronger opioids that are associated with the development of OUD. In preparation for the launch, Vertex has engaged with commercial and governmental payors to ensure formulary inclusion and limit prior authorizations. Vertex has also engaged with integrated delivery networks (large healthcare networks for hospitals and clinics) and retail pharmacy chains to broaden the national distribution of suzetrigine. During Q&A, management clarified that pricing, which remains undisclosed at the moment, will aim to balance equitable access and clinical value. Pricing will be important as suzetrigine will set the foundation for future candidates in pain management, which will involve improved NaV1.8 or NaV1.7 inhibitor molecules or combination therapy.
- On peripheral neuropathic pain, as announced in 3Q24, phase 3 enrollment is ongoing following positive phase 2 (n=192) results. Vertex plans to advance suzetrigine for painful lumbosacral radiculopathy (LSR) to phase 3, following phase 2 (n=218) results announced in December 2024. As a reminder, suzetrigine met the primary endpoint conferring a 2.02-point (or 32%) reduction in NPRS, although the placebo group experienced a similar (1.98-point reduction). As management emphasized during a conference call following these results, Vertex is satisfied with the safety and efficacy results and will design a more innovative phase 3 trial to manage the placebo effect.
- On T1D, Vertex announced zimislecel as the new name for VX-880, a stem cell-derived islet with standard immunosuppression in people with T1D with hypoglycemic awareness and severe hypoglycemic events. As announced in 3Q24, the VX-880 program has now advanced to phase 1/2/3 (n=50), following impressive results of the ongoing phase 1/2 study (n=12) announced at EASD 2024. Results showed that 11 of the 12 participants had a reduction or elimination of exogenous insulin use and maintained A1c <7.0% with TIR >70%. Importantly, Dr. Kewalramani announced plans to complete enrollment and initial dosing by this year and file for approval one year after this cohort achieves insulin independence.
- Vertex focuses on an innovative approach for developing a scalable and one-time T1D cure. This approach includes VX-264, a cell-encapsulated device, with phase 1/2 (n=17) results expected in 2025. Vertex is also developing zimislecel with alternative immunosuppression and stem cell-derived islets with hypoimmune gene editing in the preclinical stage.
20. Viking: first appearance at JPM highlights VK2735 (dual GLP-1/GIP RA) advancing to phase 3 obesity trials in 1H25, following end-of-phase 2 FDA meeting
Making a strong first appearance at JPM, CEO Dr. Brian Lian highlighted multiple clinical programs in metabolic and endocrine diseases. Viking’s pipeline includes impressive candidates progressing with impressive data.
- Subcutaneous VK2735 (dual GLP-1/GIP RA) for obesity. Calling this the “flagship program,” Dr. Lian shared that Viking has recently completed an end-of-phase 2 meeting in December 2024 and plans to initiate two phase 3 studies in 1H25: (i) in people with obesity; and (ii) in people with T2D and obesity. In February 2024, results from the phase 2 VENTURE study (n=176) showed that all four doses of VK2735 (2.5 mg, 5 mg, 10 mg, 15 mg) conferred statistically significant reductions in body weight compared to placebo at 13 weeks, with no sign of weight loss plateauing. The highest dose of 15 mg conferred 13% placebo-adjusted weight loss at 13 weeks. In the trial, VK2735 also met key secondary endpoints, with up to 88% of participants experiencing ≥10% weight loss at 13 weeks. The phase 2 trial also included a subset of participants in a pharmacokinetic assessment. At Week 16 (four weeks from the last dose), 94% of weight loss was maintained, and 83% was maintained at Week 19. Dr. Lian said these results suggest the feasibility of a monthly dosing regimen of VK2735.
- During Q&A, Dr. Lian discussed approaches to designing phase 3 trials for obesity treatments. Just last week, the FDA published a draft guidance on developing weight-loss therapies, establishing a new efficacy benchmark and providing recommendations for trials. In particular, the FDA recommends phase 3 trials with a minimum of 5% weight loss sustained for one year, with statistically significant differences from the control group, required to demonstrate efficacy. The FDA also calls for diverse populations, including people with comorbidities such as cardiovascular disease, chronic kidney disease, and liver disease. Given that Viking had an end-of-phase 2 meeting with the FDA before these draft recommendations were published, Dr. Lian knew where the agency was standing on these topics and therefore, the guidance does not impact Viking’s design for a phase 3 trial.
- Oral VK2735 (dual GLP-1/GIP RA) for obesity. Expanding promising findings of subcutaneous VK2735, Viking is pursuing oral VK2735. At ObesityWeek® 2024, phase 1 (n=92) results showed a dose-dependent reduction in body weight across all VK2735 dosing cohorts (ranging from 2.5 mg to 40 mg). Placebo-adjusted weight loss was as high as 6.8% (in the 100 mg dose group). At 28 days, weight loss did not plateau, suggesting that greater weight reduction may be possible. Weight loss maintenance was also observed up to four weeks after treatment cessation. The phase 1 trial also included an exploratory maintenance cohort to evaluate the effects of transiting from a higher to lower exposure regimen (i.e., 80 mg daily to 80 mg every other day). Despite a 50% dose reduction over the final two weeks, progressive weight loss was maintained. Dr. Lian said this reduction suggests that low maintenance dose may retain and even extend body weight reduction.
- Following these results, Viking initiated a phase 2 VENTURE oral dosing (15 mg to 120 mg) trial last week. This 13-week study in people with obesity will include two-week titration blocks applied at doses ≥60 mg.
- In 3Q24, management emphasized oral VK2735 as maintenance therapy following successful treatment with subcutaneous VK2735. Viking has also said this oral formulation could be a potential “lead-in” to an injectable, particularly in a primary care setting.
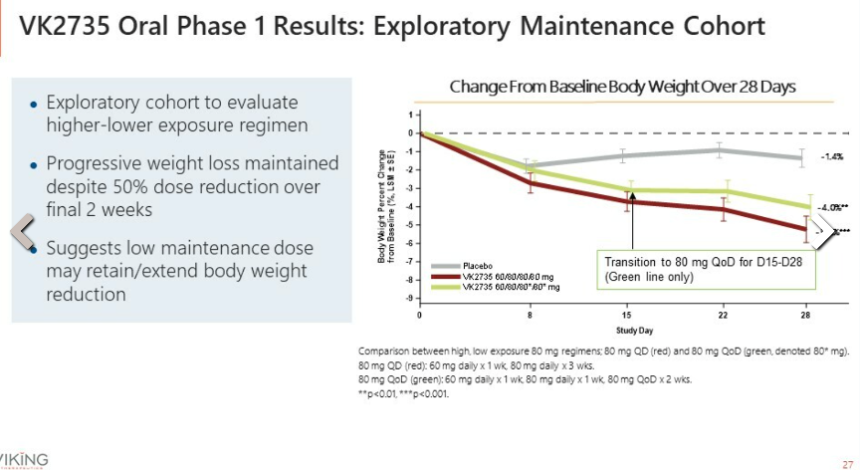
- VK2809 (small molecule agonist of the thyroid hormone receptor) in MASH. In June 2024, Viking announced positive 52-week histologic results from the phase 2b VOYAGE trial of VK2809 in MASH. Across VK2809 treatment groups, 69% achieved MASH resolution vs. 29% for placebo, and 51% achieved ≥one-stage improvement in fibrosis vs. 34% for placebo. Furthermore, participants had statistically significant improvement in plasma markers (LDL-C, triglycerides, apolipoprotein B, lipoprotein (a), and apolipoprotein C-III) compared to placebo.
21. Zealand: CEO Dr. Adam Steensberg highlights “strongest pipeline in obesity” with differentiated approach with amylin analog
CEO Dr. Adam Steensberg emphasized Zealand’s goal to be a key player in the obesity market and expressed confidence in meeting this goal based on the company’s long-standing experience with peptides for over 25 years. Dr. Steensberg said that Zealand has “the strongest pipeline in obesity” with a differentiated approach. Zealand is heavily invested in the obesity arena, as the obesity pandemic represents one of the greatest healthcare challenges in the field. 50% of adults globally are expected to be overweight or obese by 2030, and more than 5 million deaths are attributed to overweight and obesity every single year. Dr. Steensberg noted that the field is in the “early days” in the evolution of the market, as only 2% of eligible patients in the US receive prescriptions for weight loss therapies, and obesity is associated with >220 complications comorbidities (e.g., CVD, liver disease, T2D, kidney disease, neuroinflammation, and cancer). Given the prevalence of significant unmet needs and the challenges of GLP-1 RAs in obesity, Dr. Steensberg reinforced Zealand’s focus on advancing differentiated obesity programs in phase 2 and 3 trials. He said it’s not an “Olympic” for weight loss treatments but a movement toward improving tolerability and affecting co-morbidities.
- Petrelintide (amylin analog) in obesity. Dr. Steensberg said petrelintide offers a differentiated profile with high-quality weight loss and the potential for preserving lean mass, serving as an alternative to GLP-1 RAs. Petrelintide also reduces food intake via a non-incretin mechanism that increases satiety and restores leptin sensitivity. Most important, petrelintide significantly improves tolerability with GI-adverse events with both lower frequency and severity of adverse events. Petrelintide has consistently shown best-in-class data with weight loss of up to 8.6% in a 16-week phase 1b multiple-ascending dose (MAD) trial (n=48) – full results were announced in 3Q24. Following positive results, the phase 2b ZUPREME-1 trial (n=480) for overweight or obesity without T2D was initiated in December 2024. Dr. Steensberg said enrollment has been going so well that it’s beating the company’s expectations. The phase 2b ZUPREME-2 trial in people with overweight or obesity with T2D will launch in 1H25, and a phase 1b combination trial with petrelintide and GLP-1 RA is expected in 2025.
- While GLP-1 RAs are effective with 15-20% weight loss, Dr. Steensberg highlighted the following several challenges with GI side effects (i.e., constipation, nausea, vomiting, and diarrhea). He also highlighted low treatment adherence, with up to 30% of people with obesity discontinuing treatment within one month and up to 60-70% of people discontinuing treatment within 12 months.
- During Q&A, Dr. Steensberg shared his thoughts on Novo Nordisk’s CagriSema (fixed dose combination of cagrilintide 2.4 mg and semaglutide 2.4 mg) in obesity. CagriSema met the trial’s primary endpoint in the phase 3 REDEFINE 1 trial (n=3,417) announced in December 2024, demonstrating statistically significant and superior weight loss. At Week 68, CagriSema conferred a superior weight loss of 20.4%, compared to a reduction of 11.5% with cagrilintide 2.4 mg, 14.9% with semaglutide 2.4 mg, and 3.0% with placebo. Dr. Steensberg said these results demonstrate promising results for the amylin class, and given that petrelintide has almost 6x greater bioavailability, he imagines a differentiated potential.
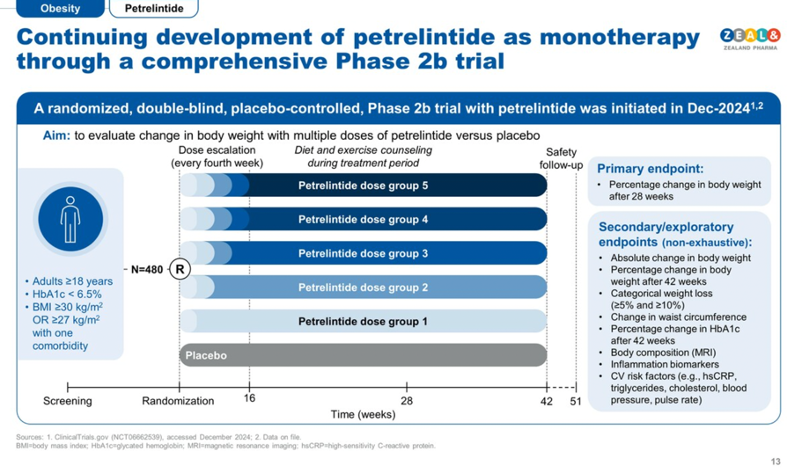
- Dapiglutide (dual GLP-1/GLP-2 RA) in obesity. In September 2024, Zealand announced positive topline results of dapiglutide from a 13-week phase 1b (n=54) multiple ascending dose (MAD) trial. In Part 1 of this trial, dapiglutide demonstrated a mean placebo-adjusted body weight reduction of up to 8.3%. Furthermore, dapiglutide doses up to 13 mg were considered safe and well-tolerated with GI adverse events consistent with the profile reported with other incretin-based therapies. Full results are expected in 2025. Furthermore, as announced in 3Q24, Zealand is conducting Part 2 of the phase 1b trial with higher doses of dapiglutide up to 26 mg over 28 weeks of treatment. Topline results are expected in 1H25. Additionally, Zealand plans to progress dapiglutide into a phase 2b trial in 1H25.
- Survodutide for obesity and MASH. Dr. Steensberg said survodutide has the potential for 20-25% weight loss, improved glycemic management, and MASH treatment. In obesity, survodutide is in the phase 3 SYNCHRONIZE program, following phase 2 results, in which survodutide demonstrated 19% weight loss in people with obesity and overweight. In MASH, Dr. Steensberg said survodutide has shown the best-in-class potential. Indeed, phase 2b trial (n=295) results showed that up to 65% of participants achieved biopsy-proven improvement in fibrosis without worsening MASH with survodutide, compared to 26% receiving placebo. Following positive results, survodutide received FDA Breakthrough Therapy Designation in October 2024 for MASH and has advanced to two global phase 3 trials in MASH: (i) 52-week LIVERAGE (n=1,800); and (ii) LIVERAGE-Cirrhosis (n=1,590).
Diabetes Technology Highlights
1. Dexcom: Preliminary full-year 2024 revenue of $4.03 billion (+11%) and 4Q24 revenue of $1.11 billion (+8%); global userbase grows 25% to 2.8-2.9 million; developmental multi-analyte sensors confirmed
Before CEO Mr. Kevin Sayer’s presentation at JPM, Dexcom announced a total preliminary, unaudited full-year 2024 revenue of $4.03 billion, up 11% from 2023. Revenue was slightly above the midpoint of Dexcom’s full-year 2024 guidance for $4.0-$4.05 billion, reiterated in 3Q24. Dexcom also shared preliminary, unaudited revenue for 4Q24 of at least $1.11 billion, up 8% from 4Q23 and 12% sequentially. 4Q24 US revenue is expected to be $803 million, up 4% from 4Q23 and 14% sequentially, while international revenue is expected to be $310 million, up 17% from 4Q23 and 6% sequentially. Mr. Sayer also announced that Dexcom’s global CGM userbase is now 2.8-2.9 million people globally, up 25% from 2.3 million users at the end of 2023.
- Acknowledging several challenges in 2024, including sales force expansion and DME share loss, Mr. Sayer was pleased with Dexcom’s progress in overcoming these circumstances. After some turbulence in the initial sales force expansion and restructuring in 2Q24, Dexcom’s US sales force personnel increased by over 40% in 2024, and Mr. Sayer said the sales force is now performing how the company originally envisioned. Specifically, Dexcom obtained over 50,000 new prescribers in 2024, and prescriptions per physician returned to levels consistent with 2Q24. Additionally, Dexcom achieved record new customer starts in the US in both 3Q24 and 4Q24. Moreover, Dexcom continued to rebuild its relationships with DME suppliers following market share loss in 2Q24. DME share continued to remain stable in 4Q24 after stabilizing late in 3Q24.
- Given meaningful progress in mitigating these challenges, Mr. Sayer expressed confidence in Dexcom’s financial outlook in 2025. Dexcom issued guidance for a total full-year 2025 revenue of $4.6 billion, which would represent a growth of 14% compared to 2024. Dexcom also anticipates a non-GAAP gross margin of 64-65% and a non-GAAP operating margin of 21% in 2025. Mr. Sayer said revenue growth will continue to be driven by expansion in all of Dexcom’s core US markets. Mr. Sayer highlighted that there are over 4.5 million people in the US on insulin therapy with reimbursement for CGM who are not on CGM, stating that this population is larger than the overall US population on CGM currently (see below). He presented data suggesting that the US T1D and T2D intensive insulin therapy populations are about 60% and 50-55% penetrated, respectively. Penetration is even lower among people with T2D on basal-only insulin therapy: about 20-25%.
- Mr. Sayer emphasized that the significant growth opportunity within the T2D population necessitated significant sales force expansion. Therefore, he said that Dexcom is “fully committed” to obtaining complete reimbursement for CGM for people with T2D on non-insulin therapy, which represents over 25 million people in the US alone.
- To build the case for broader T2D coverage, Mr. Sayer detailed four key pillars to meet this reimbursement: (i) establish CGM as a clinical standard in guidelines, such as the ADA’s Standards of Care; (ii) generate real-world evidence; (iii) establish CGM’s cost savings potential for the healthcare system; and (iv) obtain supportive clinical evidence in a randomized clinical trial.
- To advance these pillars, Mr. Sayer highlighted strengthened recommendations in the 2025 Standards of Care for HCPs to consider CGM use in non-insulin T2D. Moreover, Dexcom is conducting a randomized controlled trial in non-insulin T2D, including those on GLP-1 RAs – Mr. Sayer said this trial will complete enrollment in 2025. He said six-month results from this trial would not be expected until late 2025 at the earliest and are more likely to be announced in 2026.
- Given the growing evidence supporting CGM’s benefits in non-insulin T2D, reimbursement for this population is already expanding. As of this month, Mr. Sayer announced that two of the three largest PBMs cover Dexcom CGM for anyone with diabetes. As a result, by the end of 2025, Dexcom will obtain commercial coverage for over five million people with T2D on non-insulin therapy. Mr. Sayer emphasized that reimbursement drives high utilization among users, presenting data demonstrating that reimbursed Dexcom CGM patients use CGM regularly (see below).
- Mr. Sayer characterized Stelo’s adoption as key to “broadening impact” and noted that user numbers have now reached over 140,000 since its launch in late August. This indicates an increase of about 70,000 users from 3Q24 alone. Moreover, Mr. Sayer said Stelo generated about $22 million in revenue through the end of 2024. A majority of users have opted for a subscription model, and broad demand was indicated from people with T2D not on insulin, prediabetes, and general wellness customers.
- Mr. Sayer also highlighted continued updates in Stelo’s distribution model. Stelo is expected to become available through Amazon and Life Time, a chain of health clubs in the US and Canada. This will supplement existing distribution models through Stelo.com, direct sales to physicians, and DME cross-selling. We appreciate the increased access options for patients, considering a continued lack of availability in traditional pharmacies, while recognizing the potential environmental and logistic impacts that stem from most Amazon partnerships as well.
- To further evolve Stelo’s software, Mr. Sayer expressed Dexcom’s commitment to integrating other health data sources into Stelo, including nutrition and activity data. He highlighted Dexcom’s new partnership with ŌURA specifically to integrate product data. The Oura Ring tracks more than 20 biometrics, including heart rate, blood oxygen levels, body temperature, sleep patterns, and activity.
- Mr. Sayer echoed widespread industry interest in generative AI, broadly promising to provide personalized insights in the Stelo app through its use. Dexcom aims to integrate certain Stelo features into the Dexcom G-Series experience, and vice versa – we’re curious to hear more details on planned software updates in the future.
- Internationally, Dexcom intends to expand directly in one to two new markets per year in line with historical trends, following direct expansion to Japan and Saudi Arabia in 2024. Mr. Sayer highlighted Dexcom’s 17% international growth rate in 2024, and an additional four million in covered lives internationally for CGM in the past three years.
- To conclude, Mr. Sayer discussed the company’s future objectives, including priorities within its pipeline. After the FDA submission of a 15-day Dexcom G7 in 3Q24, Mr. Sayer said that Dexcom expects to launch this product in 2H25; however, upon launch, he said that the sensor will not be fully connected to all of Dexcom’s pump partners. On software, Mr. Sayer said that Dexcom has several updates planned, including enhancements to the Dexcom Follow app. Additionally, Dexcom will continue to generate more clinical publications supporting CGM’s benefits for a diverse set of populations, including non-insulin T2D, gestational diabetes, health and wellness, and related comorbidities (such as CKD progression).
- Mr. Sayer noted that some individuals are now wearing Dexcom G8, the company’s next-generation G-Series sensor in development. He said this features more advanced electronics to accommodate multi-analyte sensors. He added that Dexcom is advancing several probes with extended lifespans – some of which can measure “two or three [analytes]” simultaneously.
2. embecta: Gaining financial stability, international GLP-1 opportunities, and revitalization
To open Day #3, CEO Dr. Dev Kurdikar contextualized the company’s progress in achieving its strategic objectives with its challenging origin. As a reminder, the company was spun off from Becton, Dickinson and Company (BD) in April 2022, transitioning the entirety of BD’s Diabetes Care business into an independent entity.
- Dr. Kurdikar identified the three main goals that the company has prioritized since its formation and embecta’s initiatives to achieve them:
- Strengthening the core business, through overcoming inflationary pressures and other challenges, leading to national contracts with the US Department of Veterans Affairs;
- Separating from BD and “standing up,” with 98% of global revenue now under embecta’s systems and processes, including transferring a Suzhou, China, facility from BD to embecta; and
- Investing in growth, receiving FDA clearance for the now-discontinued insulin patch pump and introducing a new small-pack pen needle in Germany aimed at customers using GLP-1 RAs.
- Dr. Kurdikar addressed the rationale for the company’s recent announcement that it would not pursue a patch pump for T2D patients. He said the continued investment that would have been required to launch the patch pump within an AID system in partnership with Tidepool would have severely impacted the company’s ability to pay down debt. A market check to determine how to monetize the pump after receiving FDA clearance also did not identify major viable options. This prompted the decision to restructure the company’s focus, in hopes of leveraging capital most effectively and putting funding to work in the base business.
- This program discontinuation will generate expected pre-tax cash charges between $25 and $30 million and additional pre-tax non-cash charges between $10-$15 million. This is expected to generate cost savings of $60-$65 million, allowing the company to focus on debt repayment, with $1.6 billion debt principal outstanding.
- CFO Mr. Jake Elguicze discussed possibilities for company expansion, such as opportunities for involvement in the GLP-1 RA space. embecta sees potential for a movement towards co-packaging its multi-use pen needles with generic GLP-1 RAs and away from single-use auto injectors. Mr. Elguicze said the company can leverage its existing strengths and their position as world leaders in manufacturing pen needles, syringes, and safety devices to address this opportunity as GLP-1 RA adoption accelerates. Dr. Kurdikar cited the introduction of small-pack pen needles for GLP-1 RA users in Germany last year as one example of embecta’s emerging efforts to seize this opportunity.
- Dr. Kurdikar downplayed investor concerns that GLP-1 RAs could delay intensification to insulin therapy, which could possibly impact embecta’s business, stating it remains to be seen whether a delay would be more dramatic than one caused by other therapies – it doesn’t seem dissimilar to that of SGLT-2 inhibitors. Ultimately, we believe that in GLP-1 RA therapies’ benefits on other organ systems, such as the heart and kidneys, will extend patient lifetimes and therefore the lifespan of therapies such as insulin and associated medical supplies like syringes and pen needles, etc.
- Dr. Kurdikar emphasized a need to strengthen the company’s core business model, alongside the continuously strong performance of embecta’s major offerings: pen needles and safety products. As part of these efforts, embecta will initiate a “brand transition plan” this year focused on packaging, with the goal of modernization and revitalization.
- The packaging’s new, modern look is based upon “significant research in multiple countries.” The color scheme and product remain the same, as well as the placement of the needle length (four millimeters in an example shown). Dr. Kurdikar described the complex processes involved in the redesign, which involved the entire manufacturing process. The new branding will be first introduced in the US and Canada in the second half of fiscal year 2025, followed by Europe and the rest of the world – benchmarked for the next two years.
3. Insulet: Omnipod 5 launched in five additional European countries, with an additional five launches to follow this year; significant growth opportunity in highly underpenetrated T2D population
Insulet’s CEO Dr. Jim Hollingshead discussed the company’s efforts to accelerate growth of Omnipod 5 in the US and internationally. He said Omnipod 5 remains the market leader in AID, stating that Omnipod 5 led the field in US new customer starts (as of 3Q24) and overall AID prescriptions in the US in 2023. Moreover, he cited dQ&A data that found Omnipod was the most frequently chosen insulin pump in the previous year among new pump users in Europe. While he did not provide an updated global userbase (which will be announced in Insulet’s upcoming 4Q24 financial report), Dr. Hollingshead said that Insulet continues to achieve strong user growth in the US and internationally – at the end of 2023, Insulet reported a total global userbase of over 425,000, including 250,000 on Omnipod 5. Dr. Hollingshead attributed Insulet’s strong growth to four pillars essential to its business: (i) Omnipod 5’s unique patch pump form factor; (ii) access (including availability in 44,000 US pharmacies with Medicaid Part D coverage); (iii) optimized manufacturing; and (iv) data capabilities through Omnipod 5’s cloud connectivity, enabling easier product improvement.
- Dr. Hollingshead described the significant market opportunity for Omnipod 5 in both T1D and T2D with a total addressable market of 14 million people with diabetes. While Insulet has observed strong adoption among the T1D population, Dr. Hollingshead highlighted that this population remains significantly underpenetrated with only 40% of people with T1D in the US and 20% of people with T1D in Insulet’s international markets on an insulin pump. He said Insulet will continue to grow this market, noting that over 85% of US new customer starts in 3Q24 were previously on MDI, which remains Insulet’s target population. Since Omnipod 5’s launch in the US in 3Q22, the percentage of new user starts from MDI relative to competitive conversions has continued to increase – for comparison, this split was 65%/35% in 4Q22 and 80%/20% in 4Q23. Dr. Hollingshead also expects most new European customer starts to also come from MDI, largely due to the lack of the pharmacy benefit like in the US. Thus, he emphasized that Insulet still possesses a large growth opportunity in further expanding the AID market in T1D both in the US and internationally.
- Dr. Hollingshead especially expressed excitement for Insulet’s opportunity to develop the insulin pump market for T2D after Omnipod 5’s FDA clearance for this indication in August 2024, based on positive results from the SECURE-T2D trial. He highlighted that only 5% of people with T2D on intensive insulin therapy in the US have adopted an insulin pump, noting that part of this stems from Omnipod DASH and (previously) off-label Omnipod 5 adoption. Moreover, the US T2D basal-only insulin therapy and international T2D populations remain unpenetrated. Given Insulet is the first AID system to receive FDA clearance for T2D, Dr. Hollingshead emphasized that the company could quickly develop this market and grow its installed base, highlighting a notable increase in T2D prescriptions for Omnipod 5 already in September 2024.
- To support its efforts in growing the T2D market, Insulet is expanding its sales force to better access high-prescribing primary care practices and supplement its existing relationships with endocrinologists. In 3Q24, Dr. Hollingshead suggested this force expansion will enable Insulet to reach over 40% of the 2.5 million people on intensive insulin therapy in the US during 2025, up from roughly 30% currently. Dr. Hollingshead also said that Insulet will leverage direct-to-consumer efforts to reach more of this population because over half of Insulet’s direct-to-consumer leads historically have been people with T2D. With Omnipod 5’s FDA clearance for T2D, he said Insulet can now direct these individuals to Omnipod 5 instead of Omnipod DASH, boosting efficiency in its patient capture.
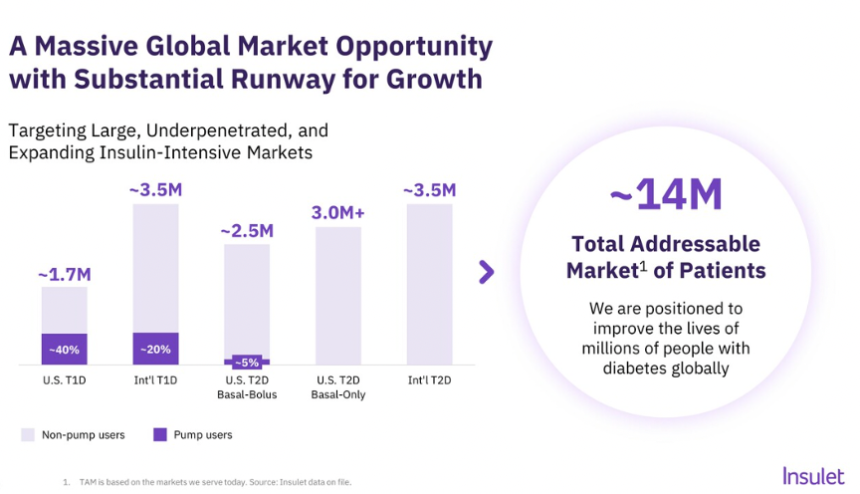
- Insulet is committed to accelerating Omnipod 5 launches in additional international markets to further grow its installed base, including 10 international markets this year. Earlier this week, Insulet announced that Omnipod 5 has launched with Dexcom G6 and FreeStyle Libre 2 Plus compatibility in Denmark, Finland, Italy, Norway, and Sweden. Launches in Australia, Belgium, Canada, Israel, and Switzerland are expected later this year. Insulet also intends to expand CGM compatibility in its international markets in 1H25. The company will expand FreeStyle Libre 2 Plus integration, presumably to Germany and France, and begin introducing Dexcom G7 integration after a full launch in the US in June 2024. As Omnipod 5 enters new markets, Dr. Hollingshead expects rapid shifts in its new customer mix between Omnipod 5 and Omnipod DASH as Omnipod 5 will drive new customer starts. Nevertheless, unlike the US where Omnipod 5 now comprises over 90% of new starts, he expects that Omnipod DASH will persist for some time in international markets due to its established installed base.
4. Medtronic: Focus on expanding MiniMed 780G indications for T2D, biosimilars, preschoolers, and pregnancy; goal to introduce Simplera in US by end of 2025
Medtronic’s CEO Mr. Geoff Martha expressed excitement about the growth trajectory of its diabetes franchise. Mr. Martha highlighted that the business has grown above Medtronic’s corporate average (about 4-5%) for six consecutive quarters, and he said Medtronic does not expect this trend to stop soon. He celebrated the turnaround in this business, emphasizing that Medtronic is developing a “comprehensive ecosystem” of diabetes technology to meet the various needs of people with diabetes. Thus, Mr. Martha expressed the company’s intention to be the leader in the smart insulin dosing space through this portfolio approach.
- Mr. Martha emphasized that CGM alone is insufficient for people with diabetes, stating that smart insulin dosing technology is expected to dominate diabetes care by the end of this decade. Among people with T1D or T2D on intensive insulin therapy[3], Mr. Martha presented data indicating that only about 14% of patients use a smart dosing system (i.e., AID or a Smart MDI system, such as InPen) with over half on standalone CGM. By the end of this decade, Mr. Martha suggested that about 50-60% of people on intensive insulin therapy could use a smart dosing system, with growth driven primarily by the conversion of standalone CGM users to these systems. He highlighted that the diabetes technology market (including AID, Smart MDI, and standalone CGM) is over $16 billion and growing in the double digits – of which about $6.5 billion of this market is attributed to AID and Smart MDI solely, indicating a substantial market opportunity.
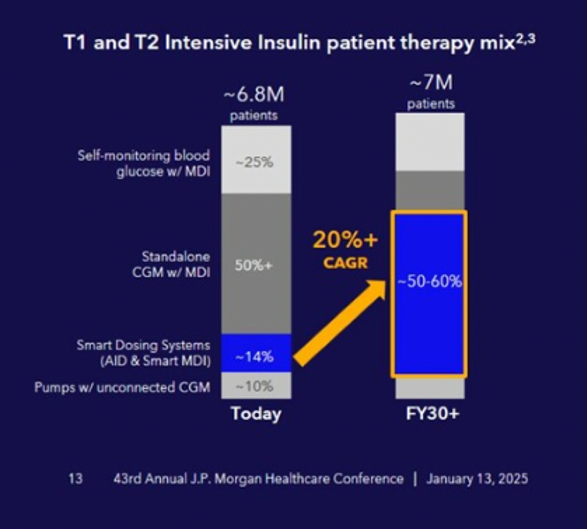
- Ms. Dallara expressed confidence in the strength of Medtronic’s portfolio, given the growing recognition of AID as the standard of care for insulin therapy. In his prepared remarks, Mr. Martha highlighted that the ADA strengthened its recommendations in the 2025 Standards of Care to consider AID at the time of diagnosis in T1D. As recognition of AID’s benefits grows, Ms. Dallara said significant opportunity to increase MiniMed 780G’s userbase remains not only among AID-naïve individuals, but also among Medtronic’s existing installed base. Specifically, she said only 60% of Medtronic’s global installed base uses MiniMed 780G.
- Mr. Martha highlighted that Medtronic has overhauled its CGM portfolio to address patient feedback on previous generations. This includes the launch of Simplera in Europe, which is half the size of the previous Guardian 4 sensor and offers a simplified insertion process. During Q&A, Diabetes EVP and President Ms. Que Dallara said that Simplera is performing “incredibly well” in Europe following its launch in 2024. Medtronic received FDA clearance for an updated InPen app to integrate with Simplera in 3Q24, facilitating a limited launch of Simplera with InPen in the US. Medtronic has also launched Simplera Sync with MiniMed 780G in at least 16 countries (as of 3Q24). Simplera Sync continues to await FDA clearance after submission in 1Q24.
- Mr. Martha also highlighted Medtronic’s partnership with Abbott on a FreeStyle Libre-based CGM that will exclusively integrate with Medtronic’s insulin delivery portfolio. During Q&A, Ms. Dallara said that this partnership will provide significant access to Abbott’s installed base, including over one million people with T1D and over two million people with T2D. Since the announcement of the partnership in August 2024, details have been scant on a potential launch time, so we’re curious to learn more about this offering in the future.
- Expanding MiniMed 780G’s indications also remains a priority for Medtronic. Medtronic intends to obtain additional indications for use: (i) T2D; (ii) pregnancy; (iii) preschoolers (i.e., ages 2+ years); and (iv) ultra-rapid acting insulin. Mr. Martha highlighted T2D, in particular, as a potentially meaningful growth driver for the system. As of 3Q24, Medtronic will file FDA submissions for expanded MiniMed 780G indications in T2D, pregnancy, and ultra-rapid acting insulin analogs in the first half of calendar year 2025. In the EU, Medtronic already filed CE Mark applications for T2D, pregnancy, and preschoolers in 3Q24, which await approval.
- Mr. Martha also briefly commented on Medtronic’s next-generation AID pipeline, including a reveal of a potential form factor for an 800-Series pump and a patch pump (see below). No details were shared regarding the development status of these pumps or their feature set, but Mr. Martha said these pumps will be integrated with Medtronic’s next-generation sensors. As a reminder, at JPM 2024, Medtronic announced that it had resumed internal development of a patch pump following the termination of its acquisition of EOFlow in December 2023. Regarding the 800-Series pump, commentary on its development has been minimal. The company first unveiled the 800-Series pump at its Investor Day at ADA 2023. Last we heard, the pump is roughly half the size of the 700-Series pump and will be a tethered pump (i.e., requiring an infusion set and a holder). The screenless pump will be fully controlled from a user’s smartphone.

5. Teladoc: Collaboration with Amazon’s Health Benefits Connector to increase access to virtual cardiometabolic programs announced
Teladoc’s CEO Mr. Chuck Divita briefly highlighted the company’s newly announced partnership with Amazon, describing it as an “exciting avenue of access” for its chronic condition management programs. Through Amazon’s Health Benefits Connector – a platform that matches customers with digital health companies that facilitate management of mental health, prediabetes, diabetes, and high blood pressure – eligible adults can now discover and enroll in Teladoc’s chronic care programs for diabetes, hypertension, prediabetes, and weight management. CFO Ms. Mala Murthy explained that Amazon users searching for related chronic care programs, products, or devices will also be shown Teladoc’s platform options. Interested individuals can check their eligibility and, if eligible, apply for Teladoc’s cardiometabolic programs on the website. Ms. Murthy noted that other platforms are already part of Amazon’s Health Benefits Connector and tempered expectations for an immediate revenue impact from the partnership. Mr. Divita expressed optimism about the collaboration to provide an opportunity to grow its chronic care userbase.
6. Virta Health: GLP-1 RAs positioned as non-essential for metabolic success but still beneficial; over $100 million in revenue by end of 2024
Virta Health’s CEO Mr. Sami Inkinen spoke to a full room on the 32nd floor of the Westin St. Francis, with a dramatic view overlooking San Francisco. To start, he explained that Virta’s digital health model is composed of three main parts: (i) personalized nutrition; (ii) clinician support; and (iii) technology and AI. Acknowledging the breadth of these three parts, he described Virta’s strength as the ability to focus on and tackle all three parts equally.
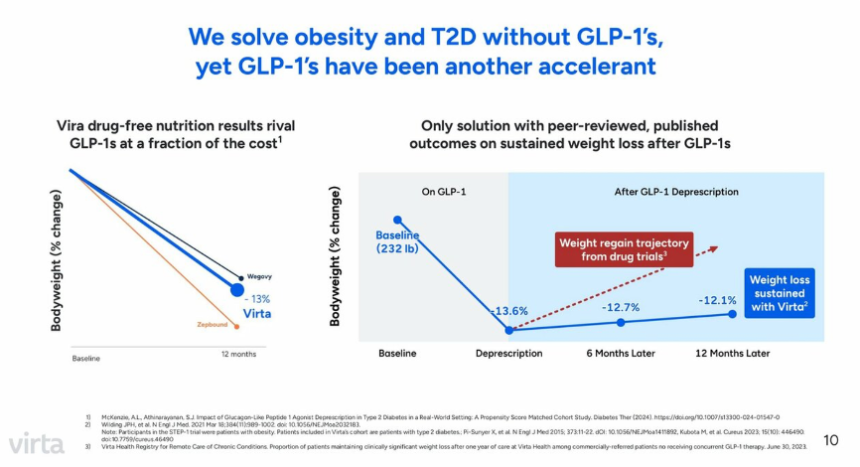
- Virta Health’s focus is on treatment modalities unrelated to GLP-1 RAs, in a notable difference from the vast majority of the diabetes and obesity field. Mr. Inkinen compared Virta’s 13% average percent body weight change after 12 months to Wegovy and Zepbound, and said it falls between average weight loss associated with the respective GLP-1 RAs (see below). He also characterized adherence to Virta Health’s platform as twice that of GLP-1 RAs, citing 83% adherence for the company’s main program compared to what he quoted as adherence of about 40% for GLP-1 RA users. The platform has also shown benefits for patients maintaining weight loss after discontinuing GLP-1 RAs, with 12% weight loss maintained 12 months after discontinuation.
- Mr. Inkinen also shared promising financial updates, starting with revenue expectations of $100 million for 2024 with profitability to follow in the next 12 months. He cited $7,200 as the economic savings per member per year, which contributes to the company’s extensive partnerships with over 530 large employers, health plans, government organizations, and tribal nations. When founded in 2017, Virta Health set the goal of reversing T2D in 100 million patients by 2025, since expanding their goal to reverse diabetes and obesity in one billion patients. The company covers over 12 million patients in the US as of this month.
Diabetes Big Picture Highlights
1. JPMorgan Chase CEO Mr. Jamie Dimon talks China, GLP-1 RAs, AI, the Trump administration, healthcare frustrations, and more in noontime conversation
Mr. Jamie Dimon (CEO, JPMorgan Chase) took part in a compelling conversation in the meeting with Ms. Lisa Gill, Head of JP Morgan’s Healthcare Services team. In about 50 minutes, Mr. Dimon shared his broad outlook on both American and global futures, touching on a wide variety of topics and soaring from high optimism to quiet concern. In his direct, often colloquial speech, Mr. Dimon presented multiple thoughts on China, GLP-1 RAs, AI, the incoming Trump administration, frustrations of the average American citizen, and much more. The first keynote of the conference left the audience rapt and reflective, and we anticipate similar invigorating talks throughout the week.
- Mr. Dimon opened with his broad outlook for this year, expecting limited change at the inauguration of the new Trump administration. He argued that the US president does not affect the economy in the first year of his administration yet shared his worries for the future beyond 2025.
- He compared the current geopolitical situation to the state of the world before World War II, describing world leaders as having “no idea what is about to happen.” He characterized China as being prepared for all possibilities related to tariffs and global influence, and urged US leaders to rise to the occasion and match this level of preparation. He also repeatedly emphasized China’s adversarial status to the US throughout the conversation.
- Ms. Gill asked Mr. Dimon about the balance between their GLP-1 RAs’ effectiveness and the costs of long-term use for companies such as JPMorgan Chase, which covers employee costs of the drugs for both obesity and diabetes. Mr. Dimon expressed his strong belief that use of GLP-1 RAs would present less of a cost over time than the consequences of diabetes and heart disease, urging the industry to move towards disease prevention when possible.
- AI remained an important topic of discussion, echoing last year’s keynote address by Mr. Dimon. He reiterated the power of AI to accelerate significant breakthroughs in medicine, such as curing cancer and identifying disease-causing gene defects.
- Mr. Dimon also reflected on the true challenge of using AI effectively, spotlighting the difficulty, even within one company, of collecting data across different databases in a usable form and leveraging the insights.
- Addressing potential dangers of AI, he likened it to other dependable technologies such as planes, cars, and pharmaceutical innovations that can be abused, but are safe and essential with proper government regulation. He envisions AI eliminating a large portion of human medical error, citing a 2003 study showing 30,000 hospital deaths per year due to human error.
- Taking a question on advice for healthcare companies in light of the murder of UnitedHealthcare CEO Mr. Brian Thompson, Mr. Dimon denounced the shooting. He also acknowledged significant frustration present among American populations with health insurance companies and beyond. He noted the impossibility for insurance companies to cover every procedure and treatment, leading to natural discontent, and called upon all businesses to look at how to serve clients fairly and ethically.
2. The early innings of AI in healthcare: Panel discussion emphasizes the need for data cleaning and transparency to guide AI’s implementation
The opening day of JPM 2025 concluded with an engaging panel discussion on the role of AI in healthcare. Panelists included Dr. Shiv Rao (CEO, Abridge), Mr. Hamid Tabatabaie (CEO, CodaMetrix), Ms. Seema Verma (EVP and General Manager, Oracle Health and Life Sciences), and Mr. Omri Yoffe (CEO, Vi). Ms. Verma said healthcare has reached an inflection point where better solutions are required, emphasizing that healthcare is becoming increasingly expensive with many healthcare providers exhibiting symptoms of burnout. She said AI has tremendous potential to address some of the current limitations in care delivery. In particular, she said that AI may be especially beneficial for automating repetitive or mundane tasks; however, she noted that AI’s implementation for clinical decision support raises significantly more challenges for regulators and policymakers. Panelists unanimously agreed that AI’s implementation in healthcare remains in its “early innings,” proposing several key considerations to guide ethical and effective use to support clinical decision making.
- Panelists acknowledged that there are significant questions regarding the interface between humans and AI models. In particular, circumstances in which humans should retain authority within automated processes should be identified. Mr. Tabatabaie cited examples from radiology, which has significant experience with automated data interpretation due to shortages in specialists. He said that the implementation of AI in the radiology field has indicated the need for a “human in the loop,” stating that data often contains noise and contaminants that limits the efficacy of an AI model and requires human interpretation. Mr. Tabatabaie said that healthcare data requires significant cleaning, which is essential for the effective implementation of an AI model.
- Panelists also argued that AI models should be transparent to engender trust among policymakers, regulators, clinicians, and patients. Dr. Rao argued that “trust is the only currency in healthcare,” stating that ensuring models are transparent is essential. He said end users should be able to audit the output to facilitate more trust in implementing the models. Mr. Yoffe agreed, advising model developers to be transparent in the exact benefits and return on investment that a model will provide. He said that appropriately structuring and indexing underlying data is essential to a model’s integrity in producing reliable, interpretable outcomes. In his words, providing transparency into the model’s functions should therefore cultivate more trust in its outcomes.
3. Inside the FDA: Panel discussion offers insight into approval processes, serving as an opportunity for connection between developers and regulators
We attended a popular panel discussion with representatives from the FDA focused on the FDA’s Total Product Life Cycle (TPLC) Advisory Program (TAP) Pilot – an initiative that began in 2023 with the goal of streamlining approval and increasing communication between developers and regulators. Panelists included Dr. Douglas Kelly (Center for Devices and Radiological Health Deputy Center Director, FDA), Mr. Kai Kadoich (TAP Advisor), Dr. Laura Gottschalk (TAP Advisor), and Dr. Ross Segan (Director of the Office of Product Evaluation and Quality, FDA). In a rare conversation to hear from FDA, panelists offered a fascinating opportunity to glean some insight into the FDA’s regulatory processes for medical technology.
- Moderator Mr. Robbie Marcus (JP Morgan) refered to the FDA as a “black box” to outsiders, asking Dr. Kelly to elaborate on how the TAP program can reduce confusion in the approval process. Dr. Kelly contrasted medical devices and pharmaceuticals, stating that medical device developers must appeal to the Centers for Medicare & Medicaid Services (CMS), payors, and advocates in conjunction with the FDA approval process. He said he emphasizes communication between developers and regulators in his role as Deputy Center Director, showing companies the coordination required between regulatory agencies. Drawing laughs from the crowd, he likened the FDA approval process to dating: companies may meet with the FDA and not immediately have “chemistry,” leading to a guarded approach instead of an open dialogue. Dr. Kelly said this may cause confusion and delay the approval process. He advocated for companies to explain the background and goals behind their devices to build trust and cultivate an ongoing relationship with the FDA. When asked about how the TAP program can improve the lengthy approval process for developers, he advocated for increased communication and feedback, so developers may better understand the level of evidence needed for approval before companies.
- An audience member rebutted by sharing his experiences in receiving delayed responses to requests for meetings with the FDA, asking how TAP’s ambitions could improve scheduling processes. Dr. Kelly described TAP’s goal of frequent, informal meetings between regulators and developers. Mr. Kadoich asserted that teams within TAP are quite responsive to companies, and Dr. Segan noted that in his four months at the FDA, he has encountered innumerable mission-oriented people who have been working with very limited bandwidth since the COVID-19 pandemic – certainly, we can imagine this would be questioned by anyone. On a positive note, the audience member was urged to talk further with the panel after the session. We are always impressed by regulatory resources of FDA and believe we likely don’t have a good sense of the pressures and delays.
- Regarding coordination between multiple federal agencies, the panelists discussed strategies for communication with CMS, acknowledging more collaboration between FDA and CMS to offer approval and reimbursement simultaneously would simplify processes. Dr. Kelly spoke about limited staffing at CMS and reassured the crowd that he has attempted to improve outreach – for instance, assigning TAP members to work for one week at CMS to better understand their processes.
- Addressing the incoming Trump administration, panelists expected limited future impact on the approval process, noting that many of the panelists had served under both the prior Trump and Biden administrations. Dr. Gottschalk characterized the approval process as very focused, with limited influence from the presidency and the Administrator of the CMS. No one mentioned the much-admired current head of FDA, Dr. Robert Califf, or what changes were likely in store from incoming FDA head Dr. Mark Makery.
5. Retired General Mark Milley tackles geopolitics and the military’s role in health innovation
Tuesday’s keynote address was delivered by retired General Mark Milley, former chairman of the Joint Chiefs of Staff under Presidents Trump and Biden. Since retiring in September 2023, Gen. Milley has joined JPMorgan Chase as a senior advisor, responsible, in his own words, for “evaluating risk around the world.” He characterized the current global geopolitical risk as definitively increasing, delving into a detailed analysis of foreign affairs in each region. For the politically minded, this talk was a thrilling opportunity to contextualize healthcare with geopolitical context.
- Echoing JPMorgan Chase CEO Mr. Jamie Dimon’s remarks in yesterday’s noontime discussion, Gen. Milley compared the current political state of affairs to World War I and II. In a history overview spanning the Treaty of Westphalia in 1648 to the state of China’s military in 1980, Gen. Milley showed how previous cycles of power inform his current outlook on the world.
- Gen. Milley pointed to the rise of nationalism, protectionism, populism, and tariffs as factors that may potentially disrupt peaceful norms of the past one hundred years. He urged diplomacy and for the US to negotiate with “adversaries” such as China.
- He spoke of technological optimization, arguing that Germany’s success in World War II was due to innovation using the same military technologies that all major powers shared. With the invention of the Blitzkrieg, Germany was able to overpower larger armies and occupy large swathes of Europe at the time. Gen. Milley drew parallels to the age of AI and robotics, such as self-driving cars. He said future technological success will rely on innovation, and wants the US to “get there first,” as opposed to other world powers.
- Gen. Milley surveyed the geopolitical climate, including analyzing China’s rivalry with the US, describing the war in Ukraine as a stalemate, and discussing complexities across the Middle East. He described each situation along a spectrum from competition to confrontation to conflict, with the goal of avoiding full-blown military conflict and bloodshed. He concluded, “Isn’t that cheerful? … I’m a very positive person. Very positive things will get bad,” eliciting the most laughter of the session.
- Gen. Milley discussed the military’s role in driving health innovation. He compared the size of an organization such as Doctors Without Borders to the sheer magnitude of the US military, which presents many opportunities for expedited development in healthcare.
- Gen. Milley highlighted the US military’s involvement in Operation Warp Speed during the COVID-19 pandemic. He also cited treatments for yellow fever, malaria, and modern anesthetics that were partially developed by the military.
- In a fascinating statistic, he estimated survival rate in combat is around 90% when surgery is performed within one hour, even with very serious injuries. He envisions huge potential for telemedicine to be used to increase medical access on the battlefield and in remote areas, with a surgeon guiding trained medics on how to perform life-saving operations from a distance.
- During Q&A, an audience member shared a deeply personal connection regarding mental health challenges among veterans. Gen. Milley agreed, calling for destigmatization and for all of us to look out for each other and recognize signs of mental health challenges in our loved ones.
9. ARPA-H: Catalyzing biomedical breakthroughs in women’s health through the Sprint for Women’s Health, NITRO, and POSEIDON
As part of JPM 2025’s new and extremely valuable Women’s Health Series, ARPA-H hosted an afternoon panel discussion devoted to discussing the agency’s efforts to catalyze biomedical breakthroughs to support women’s health. We will be back with more on these sessions. As background, ARPA-H is a federal agency formed in 2022 as part of the U.S. Department of Health and Human Services (HHS). Aiming to achieve transformative, sustainable, and equitable health solutions, ARPA-H is tasked to build capabilities (or platforms) through investments and partnerships. For more on ARPA-H’s involvement with diabetes and obesity specifically, see our interview with ARPA-H’s Dr. Paul Sheehan and former ADA CSMO Dr. Bob Gabbay in August 2024. Inaugural ARPA-H Director Dr. Renee Wegrzyn positioned the agency as both disease- and technology-agnostic, seeking to derisk new technologies and empower them to be financially sustainable. Dr. Jenica Patterson (Portfolio Lead, ARPA-H) and Dr. Ross Uhrich (Program Manager, ARPA-H) discussed several ongoing projects to accelerate innovative solutions in women’s health.
- In 2024, ARPA-H initiated the Sprint for Women’s Health to fund and accelerate development of biomedical therapies and technologies to improve women’s health outcomes. Dr. Patterson said the initiative received 1,700 abstract submissions from 34 countries within a 45-day period, providing $113 million in funding across 24 awards to support novel solutions for unmet needs in women’s health. She highlighted that ARPA-H presents a unique opportunity to reach non-traditional partners that typically do not collaborate with the federal government, noting that over half of submissions originated from small businesses composed of under 10 people.
- As an example, this initiative provided $10 million in funding to support Daré Bioscience’s development of a novel treatment to clear persistent high-risk human papillomavirus (hrHPV) infection, which causes cervical cancer. Ms. Sabrina Johnson (President and CEO, Daré Bioscience) said funding to support this treatment, called DARE-HPV, was crucial because investors are wary of funding projects without a defined regulatory approval path. ARPA-H can thus furnish necessary support for novel treatment approaches that may experience difficulties raising capital.
- Dr. Uhrich discussed how the NITRO and POSEIDON initiatives approach innovation. NITRO aims to catalyze development of regenerative tissue therapies for osteoarthritis, which predominately affects women, while POSEIDON seeks to develop an over-the-counter, breath- or urine-based screening for over 30 cancers at stage 1, including breast cancer. Speaking specifically on the NITRO program, he said innovation resides within three areas: (i) technology; (ii) access; and (iii) incentives. On access, he noted that clinical trials in the NITRO program must reflect the exact demographic distribution of the disease in the US, and the program stipulates a pricing clause mandating a 75% discount relative to the standard of care.
10. The US healthcare landscape: Former FDA Commissioner Dr. Scott Gottlieb speculates on the potential impact of Trump administration on biomedical innovation and public health
In JPM 2025’s final keynote session, JPMorgan’s Mr. Christopher Schott interviewed former FDA Commissioner Dr. Scott Gottlieb on the future of the US healthcare landscape under the incoming Trump administration. Dr. Gottlieb most recently served as the 23rd Commissioner of the FDA in the first Trump administration from May 2017 until April 2019. Dr. Gottlieb drew from his extensive public health and policymaking expertise across the public and private sectors to speculate on potential healthcare reform that could be spearheaded by the second Trump administration, including on GLP-1 RAs and compounding, PBMs and drug pricing, and the food industry. Reflecting on the current state of the healthcare industry, Dr. Gottlieb expressed optimism regarding the pace of innovation, stating that the field has reached an “inflection point” where regenerative medicine, especially with cell- and gene-based therapies, is beginning to seem palpable.
- Addressing uncertainty regarding the incoming administration’s stance on access and reimbursement for GLP-1 RAs, Dr. Gottlieb expressed minimal concern about possible restrictions on access. He does not expect Medicare reimbursement for GLP-1 RAs in obesity to be withdrawn because most of the elderly population will become eligible for these therapies as their indications further expand. For instance, positive results from ongoing clinical trials investigating GLP-1 RAs’ efficacy in dementia and early Alzheimer’s disease, such as the phase 3 EVOKE/EVOKE+ program of semaglutide, would open an even broader market, so Dr. Gottlieb does not foresee this policy to change.
- Dr. Gottlieb emphasized that GLP-1 RA compounding is currently one of the most contentious areas in policymaking. When the FDA declared that the shortage of Lilly’s tirzepatide had resolved (announced in December 2024), Dr. Gottlieb said he urged the agency to pursue legal action against compounding pharmacies due to safety concerns that could result in “tragedies.” Upon announcement of the shortage resolution, the FDA announced that pharmacies would stop making compounded versions of tirzepatide within 90 days. Dr. Gottlieb said that he received significant pushback from policymakers and politicians, primarily conservatives, who believed that the FDA was too aggressive against compounding pharmacies. However, he argued that if compounding was permitted to become more widespread, serious adverse events could occur, which would subsequently attract severe scrutiny to the FDA for failing to crackdown on compounding pharmacies and protect public health.
- Speculating on potential healthcare reform, Dr. Gottlieb expects any significant policy changes to be incorporated into a reconciliation bill rather than standalone legislation. He suggested that reforms to the Inflation Reduction Act may be implemented, particularly regarding drug pricing. As one example, he suggested policymakers could remove orphan drug exemptions. However, he was skeptical that policymakers would also alter the small molecule exemption (i.e., small molecules are exempt from Medicare price negotiations for nine years after FDA approval compared to 13 years for biologics), which pharmaceutical companies have criticized as an impediment to innovation in small molecules. Dr. Gottlieb described establishing an equivalent 13-year small molecule exemption as a “stretch” since the federal government will primarily seek to save money, rather than spend.
- Dr. Gottlieb also suggested that PBM reform could be incorporated into the reconciliation bill. However, he expressed his disagreement with this bill, expressing his view that it is “not very impactful.” Specifically, he said that it may simply appease short-term momentum for healthcare reform, but it may impede long-term reform by antagonizing PBMs and increasing their resistance to further action. Acknowledging the need for PBM reform, he argued that the pharmaceutical industry committed a strategic mistake by “demonizing” the PBM industry, resulting in policymakers developing distrust of both pharmaceutical manufacturers and PBMs and perceiving price controls as the potential solution.
- Dr. Gottlieb supported efforts to reform regulation of the food industry; however, he suggested that current momentum and rhetoric has not yet detailed specific regulatory approaches. He proposed several changes to regulation on food, such as requiring more forceful disclosures on unhealthy food attributes or permitting food manufacturers to make broader claims on healthy attributes and therefore directly compete on product healthiness. He said the existing FDA staff strongly supports increased regulation of the food industry; however, he suggested the agency has historically not had the resources to effectively combat this issue.
11. Fireside chat with HHS Secretary Xavier Becerra: Analyzing the Biden administration’s accomplishments, including MDPNP, ACA, and vaccines
Ms. Ipsita Smolinski (Capitol Street) moderated a fascinating fireside chat with the outgoing Secretary of the US Department of Health and Human Services (HHS) Xavier Becerra. Sec. Becerra reflected on the department’s accomplishments during the Biden administration to improve drug affordability, expand healthcare access, and accelerate biomedical innovation. He likened his journey leading the department to a spinning ride at an amusement park. Due to the Department’s efforts, he said that the US is a “far healthier and stronger country” than it was just four years ago.
- Sec. Becerra celebrated the establishment of the Medicare Drug Price Negotiation Program (MDPNP) through the Inflation Reduction Act. As a reminder, the MDPNP requires that the Centers for Medicare and Medicaid Services (CMS) negotiate prices with drug manufacturers for brand-name drugs without generic or biosimilar competition that are covered by Medicare Parts B or D – a monumental shift in US policy since the federal government previously could not directly negotiate drug prices. For the first 10 drugs incorporated in the MDPNP (which includes AstraZeneca’s Farxiga, Merck’s Januvia, BI/Lilly’s Jardiance, and Novo Nordisk’s NovoLog and Fiasp), the federal government negotiated significant list price discounts, ranging from 38% to 79%. Once these prices become effective in 2026, Sec. Becerra said that these discounts will save Medicare Part D beneficiaries $1.5 billion, which is nearly double what the Congressional Budget Office initially projected would have been saved. If these prices were in effect in 2023, an estimated $6 billion in net covered prescription drug costs would have been saved, translating to 22% lower net aggregate spending.
- Sec. Becerra described the US as the “pocketbook” for drug sales, stating the MDPNP revealed that the US is often paying more than it should compared to other countries. He said the MDPNP will have tremendous benefits for drug affordability and reduce cost burden for patients.
- On health insurance, Sec. Becerra highlighted several meaningful policy changes to expand coverage for Americans, noting that over 300 million Americans now have health insurance. He partly attributed this significant growth in the covered population to the Affordable Care Act (ACA), highlighting that a record number of individuals, nearly 24 million, are receiving their health insurance through this legislation – about double from those covered during the previous Trump administration. Addressing the incoming Trump administration’s threats to weaken or repeal the ACA, Sec. Becerra suggested this legislation cannot be undone easily due to the millions of Americans who depend on the ACA for health insurance.
- On women’s health, Sec. Becerra emphasized that the US suffers from the highest rates of maternal morbidity and mortality among high-income countries. He said that mortality typically occurs within three to four months of birth; thus, HHS sought to expand the duration of coverage for postpartum care. Now, women on Medicaid can receive postpartum care for 365 days after birth, up significantly from 60 days previously.
- On medical devices, Sec. Becerra said that innovation is dictated by the speed of the FDA. He acknowledged that bottlenecks at the FDA can slow the pace of technologies entering the market. He attributed capacity constraints at the FDA to its budgeting structure, which is based on a Congressional allocation that was set based on historical data. Consequently, as the medical device industry rapidly advances, the FDA’s regulation cannot effectively maintain pace with innovation. Sec. Becerra weighed in on this, saying, “The FDA can only move as fast as the speed of the money it receives.”
- Sec. Becerra also mentioned the rapid development of safe and effective COVID-19 vaccines as a transformative public health accomplishment, stating over 700 million COVID-19 vaccinations have been administered to Americans. He said the US must remain ahead of other potential pandemic threats, such as avian flu or mpox, suggesting that the COVID-19 vaccination effort may serve as a foundation to inform responses to future public health emergencies. When asked about the nomination of Mr. Robert F. Kennedy Jr. as the next HHS Secretary and his repeated criticism of vaccines, Sec. Becerra emphasized that the HHS operates on “science, not intuition or politics,” stating that the evidence supports the success of vaccines.
12. AI in diagnostic testing: Panel demonstrates opportunities for innovation and equity, while calling for regulation and caution
The Union Square room featured yet another standing-room only attendance for a second discussion on AI, which has been one of the biggest themes this week! Panelists included Mr. Troy Tazbaz (Director of Digital Health Center for Excellence, FDA), Dr. Kate Sasser (Chief Scientific Officer, Tempus), Dr. Lee Fleisher (Former CMS Chief Medical Officer; CEO, Rubrum Advising), and Ms. Jennifer Leib (Founder, Innovation Policy Solutions). To define AI, Mr. Tazbaz summarized it as using machine learning and algorithms to automate tasks. He shared his clinical focus in ensuring product safety, emphasizing that only predictive AI models, not generative, have been approved by the FDA. Panelists agreed on the need to contextualize AI results, while also sharing optimism about the many possibilities it offers for healthcare.
- Dr. Fleisher deemed AI to be transformative, envisioning huge potential for rural medicine to connect more communities to academic medical centers. He emphasized the need for a new payment model to fund rural health broadly, which should include stipulations to afford AI and properly deploy AI. Dr. Sasser advocated for AI’s potential to transform precision medicine by identifying care gaps and identify targets for novel small molecule therapies.
- Despite the potential of AI, the rapid explosion has left many of the panelists urging for regulation and caution. Drawing upon his background in technology and software, Mr. Tazbaz emphasized the need for post-market monitoring of AI-based technologies, stating “I’ve never deployed an enterprise-based software without having that capability.” He touched on equity as well, advocating for post-market monitoring for all hospitals where AI is deployed, as opposed to simply those with the most resources. Ms. Leib also pushed back on the idea of AI utility being centered in the opinions of payors, imploring the room to elevate the needs of patients and providers first.
13. Investment opportunities to drive innovation in emerging markets
In the afternoon, Ms. Krista Donaldson (Stanford) moderated a standing-room only panel discussion on investment opportunities and risks in emerging markets. Panelists included Ms. Nafisa Jiwani (US International Development Finance Corporation), Ms. Stacy Feld (Johnson & Johnson), Mr. Glenn Rockman (Adjuvant Capital), and Mr. Gregory Rockson (mPharma). Ms. Donaldson acknowledged that healthcare investors perceive significant risks in emerging markets, including limited infrastructure, currency dynamics, and evolving regulatory frameworks. Mr. Rockman argued that the biggest barrier to healthcare investment in emerging markets remains successfully convincing stakeholders that potential investment returns exceed the risks, recommending that manufacturers generate comparative data illustrating healthcare expenditures in high-income countries to convince investors of the need for innovation in emerging markets. Ms. Jiwani encouraged companies to partner with the US International Development Finance Corporation to support and derisk investments in these regions. Importantly, Mr. Rockman emphasized that novel innovation must be delivered on an affordable basis to truly impact patients’ lives.
- Panelists shared what they perceived as the most promising near-term opportunities in healthcare in emerging markets. Ms. Feld highlighted the significant shortage of healthcare workers globally, which contributes to gaps and inequities in care. She advocated for focused development of tech-enabled solutions to empower healthcare workers as the center of care and improve outcomes. Ms. Jiwani said that noncommunicable diseases present a significant unmet need in low-income and middle-income countries, emphasizing an opportunity to reimagine oncology care in these markets. Citing mPharma’s work to improve community pharmacy infrastructure and the prescription supply chain in Africa, Mr. Rockson advocated for novel solutions to scale pharmacy-faced primary care.
--by Nour Khachemoune, Kat Moon, Jeremy Alkire, Elaine Young, Andrew Goyette, Esther Min, Monica Oxenreiter, and Kelly Close
[1] The Inflation Reduction Act requires CMS to negotiate prices with drug manufacturers for brand-name drugs without generic or biosimilar competition that are covered by Medicare Parts B or D. A drug is eligible for price negotiation seven years after FDA approval for small molecules and 11 years after approval for biologics – which incentivizes pharmaceutical and biotech companies to favor biologics over small molecules.
[2] Ionis is also positioning olezarsen for individuals with severe hypertriglyceridemia (SHTG). Olezarsen is currently in the following phase 3 trials for SHTG: (i) CORE (n=617); (ii) CORE2 (n=446); and (iii) ESSENCE (n=1,478). Results from these trials are expected in 2H25. Dr. Monia emphasized that SHTG presents a significantly larger market opportunity than FCS alone. With the approval and upcoming launch of olezarsen in FCS, Dr. Monia said this will serve as a “launch pad” for further progress.
[3] Medtronic’s slide presentation noted that this population samples the top global developed markets by health expenditure per capita, which was predominately included the US, Western Europe, Canada, Japan, Australia, and South Korea.