ADA 2025 Day #2 Highlights –
Executive Highlights
- ADA 85th Scientific Sessions continued with high heat and energy in Chicago! We appreciated new data results and analyses across diabetes technology and therapy, as well as clinical insights into cardio-renal-metabolic health. In case you missed it, check out our Day #1 Highlights from yesterday and our preview for what’s to come over the next two days!
- In therapy, we especially appreciated new data on incretin-based therapies, including Lilly’s orforglipron (oral GLP-1 RA), tirzepatide, and Novo Nordisk’s semaglutide.
- In a standing room-only hall, Lilly presented the full results of the phase 3 ACHIEVE-1 trial (n=559), which studied Lilly’s oral GLP-1 RA orforglipron in T2D. Results were simultaneously published in NEJM, “Orforglipron, an Oral Small-Molecule GLP-1 Receptor Agonist, in Early Type 2 Diabetes” – notably, orforglipron achieved superiority for the primary endpoint of superior A1c reduction compared to placebo at Week 40, conferring A1c reduction of 1.3%-1.6% from a relatively low baseline of 8%, for the efficacy estimand. In key secondary endpoints, up to 76% of participants taking orforglipron achieved the A1c target of <7.0%, 66% achieved an A1c of ≤6.5%, and 26% achieved <5.7%[1]. Discontinuation for GI reasons was low. These findings support the potential for orforglipron to substantially demonstrate A1c and weight reduction in T2D with a safety profile consistent with the overall GLP-1 RA class.
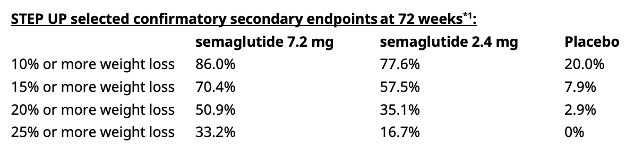
- In obesity, Dr. Sean Wharton (University of Toronto, Canada) presented full results of the phase 3b STEP-UP trial (n=1,407) evaluating high-dose semaglutide (7.2 mg) in people with overweight or obesity without T2D. See Novo Nordisk’s press release. Assuming that all participants adhered to treatment, semaglutide 7.2 mg achieved weight loss of 20.7% at Week 72, compared to 17.5% with semaglutide 2.4 mg and 2.4% with placebo, from a mean baseline body weight of 113 kg (249 lbs). Furthermore, one-third (33.2%) of people treated with semaglutide 7.2 mg achieved a weight loss of ≥25%, compared to 16.7% with semaglutide 2.4 mg and none with placebo. The safety and tolerability profiles were similar to those of semaglutide 2.4 mg.
- In cardiovascular health, we were excited to hear new findings from a post hoc analysis of the STRIDE trial (n=792), which investigated semaglutide 1.0 mg (Ozempic) in people with T2D and early-stage symptomatic peripheral artery disease (PAD). The analysis examined whether maximum walking distance (the primary outcome) and pain-free walking distance were consistent across subgroups. The effects of semaglutide on maximum walking distance and pain-free walking distance were consistent across all subgroups. Additionally, correlation analyses suggested that the functional benefits observed in the study were not correlated with the weight loss and glycemic improvements also seen in the study. Dr. Alice Cheng (University of Toronto, Canada) shared five valuable “key takeaways” for endocrinologists, including the importance of screening for PAD and considering the risk of sarcopenia.
- Dr. Dan Drucker (Mt. Sinai Hospital, Canada) outlined thought-provoking hypotheses for why GIP receptor agonism and antagonism both appear to be effective, complementing high interest in incretin-based therapies. He drew on an article by Duke’s Dr. Jonathan Campbell, which was published just this month. Emerging candidates suggest that both receptor agonism and antagonism (e.g., Amgen’s MariTide) are effective. He shared four hypotheses for this seemingly contradictory finding: (i) chronic agonism (i.e., activation) of the GIP receptor can lead to desensitization of the receptor, thus resembling the effects of a GIP antagonist; (ii) antagonism of GIP receptors on inhibitory GABAergic neurons may result in the disinhibition of neurons with GLP-1 receptors; (iii) GIP receptor agonism increases the activity of inhibitory GABAergic neurons, reducing food intake; and (iv) loss of GIP receptor activity over time (e.g., due to genetic or pharmacological reasons) leads to an increase in GLP-1 receptor sensitivity due to disinhibition or compensation. Moreover, he reviewed the research around the neuroprotective effects of incretin therapies and their potential mechanisms.
- In tech, we saw two related oral presentations addressing GMI-A1c discordance. Previous studies have identified a “glycation gap” in which individuals’ A1c may differ from that expected, with the gap being both positive (i.e., higher A1c than expected) and negative (i.e., lower A1c than expected). Glycation gaps are associated with increased mortality.
- Dr. Rich Bergenstal (International Diabetes Center) presented striking results from an analysis of the GDAC study, which evaluated the agreement of an updated GMI metric (uGMI) with A1c. uGMI accounts for population-based red blood cell factors, unlike the original GMI. In GDAC, uGMI showed greater alignment with A1c. In the overall cohort, original GMI and A1c demonstrated a regression slope value up to 25% away from unity (perfect correlation); however, with uGMI, the regression slope was within 2% of unity. The analysis was also replicated on a real-world dataset in which GMI-A1c discordance was more pronounced. uGMI ameliorated these discrepancies well.
- Prof. Ramzi Ajjan (University of Leeds, UK) presented a similar analysis examining the efficacy of “personalized A1c” (pA1c) to mitigate GMI-A1c discordance, using the same GDAC and real-world datasets. Prof. Ajjan emphasized that uGMI better minimized deviations in the regression slope between GMI and A1c; however, it did not address the spread of the data, which is related to inter-individual variations in red blood cell (RBC) physiology. Personalized A1c (pA1c), which addresses differences in RBC characteristics, was used to tighten the relationship between GMI and A1c. Indeed, correlation to GMI improved with use of pA1c in both the GDAC analysis and the real-world analysis.
- Additional real-world and clinical evidence supporting AID systems continued to dominate discussion:
- MiniMed 780G: Dr. Jennifer McVean (Medtronic) presented real-world outcomes from nearly 375,000 global MiniMed 780G users, up from a dataset of 322,043 global users at ATTD 2025 and the same dataset of 101,629 at EASD 2023. In the overall cohort, mean Time in Range (TIR) was 72% – consistent with previous MiniMed 780G datasets. Time in Tight Range (TITR) was 47%. Time below Range (TBR) was comparable to previous datasets as well at 1.7% with 0.3% Time <54 mg/dL. Use of the recommended settings (i.e., two hours active insulin time and 100 mg/dL) was associated with increased TIR, primarily driven by reductions in Time above Range (TAR). Levels of 78% TIR and 53% TITR for those using the MiniMed 780G were 1.3 hours higher than the overall cohort, driven largely by 1.2 hours less “Time above Range” as well as “Time Below Range” that is slightly lower among MiniMed 780G “recommended setting” users. Dr. McVean emphasized that these results indicate AID should be offered to everyone with T1D.
- Omnipod 5: Dr. Kristin Castorino (Sansum Diabetes Research Institute) presented a sub-analysis of Insulet’s SECURE-T2D study evaluating the impact of Omnipod 5 initiation on diabetes distress in adults with T2D. After 13 weeks, the proportion of participants reporting “moderate” or “high” distress (T2-DDAS score ≥2.0) dropped significantly from 66% to 55% (p<0.001). Improvements were observed across all baseline TIR groups, with the largest reductions in participants with baseline TIR of 30%–60% (-13 percentage points) and ≥60% (-12 percentage points).
- twiist: Dr. Joanna Mitri (Sequel Med Tech) revealed real-world outcomes for early adopters in the twiist Early Experience Program (n=36). All participants in the real-world dataset were adults with T1D. The mean time for device use was 35 days, and presented results were acquired through June 6, 2025. Mean TIR was 80% with TAR of 17% and TBR of 2.9%.
Day #2 of the 85th ADA Scientific Sessions kicked off in Chicago! See below for our top highlights across diabetes therapy, technology, and big picture, and stay tuned for our latest updates over the next few days.
Table of Contents []
-
Diabetes Therapy
- 1. Full results of phase 3 ACHIEVE-1 trial: Orforglipron demonstrates up to 1.6% A1c reduction and 8% weight loss
- 2. GIP receptor agonism and antagonism: Dr. Dan Drucker on why both seem to be effective; impact of incretins on neurodegenerative disease
- 3. Full results of phase 3b STEP-UP trial: High-dose semaglutide 7.2 mg confers greater weight loss than semaglutide 2.4 mg in people with obesity
- 4. Post hoc analyses of COMBINE 1, 2, and 3 trials assess CGM metrics of IcoSema and glycemic benefits across kidney function
- 5. Post hoc analysis of SURMOUNT-5 trial raises concerns over “excessively” rapid weight loss
- 6. Addressing CV and kidney disease in T1D: Guidelines, GLP-RAs, and SGLT-2 inhibitors
- 7. STRIDE trial: New post hoc analyses show consistent functional benefit across subgroups for semaglutide in people with T2D and peripheral artery disease
- 8. GLP-1/glucagon RA pemvidutide reduces cardio-inflammatory lipid levels in people with obesity or overweight
- 9. Further analyses of SURPASS-SWITCH trial show superior A1c and weight reduction in people switching from dulaglutide to tirzepatide
- 10. ADA Presidents’ selected oral presentation features a retrospective cohort study on the association of age at T1D diagnosis with cardiovascular and kidney disease
- 11. Addressing residual cardiovascular risk in people with diabetes and cardiovascular disease
- 12. SGLT-2 inhibitors increase risk for hospitalization from DKA by 40% vs. GLP-1 RAs in patients with T2D and CKD
- 13. Emulation trial shows tirzepatide has superior cardioprotective benefits compared to dulaglutide
- 14. Novo Nordisk’s subcutaneous amycretin confers 24% weight loss at 36 weeks in people with obesity in phase 1a/2b study
- 15. GLP-1 RAs are not yet cost-effective when compared to obesity counseling in T2D prevention
-
Diabetes Technology
- 16. New analysis of GDAC trial and real-world data shows improved alignment between updated GMI equation and A1c
- 17. Personalized A1c improves correlation between GMI and A1c
- 18. Largest real-world dataset of global MiniMed 780G users (n=374,548) shows mean TIR of 72%; TIR increases with recommended settings
- 19. All eyes on T2D and ketone testing: Abbott product theater reviews unmet needs in analyte sensing
- 20. Omnipod 5 users with T2D report less diabetes distress compared to MDI
- 21. Dr. Rich Bergenstal, Dr. Anita Swamy, and Dexcom Warrior Mr. Lance Bass review the present and future of Dexcom CGM
- 22. UNITE RCT of CGM in non-insulin T2D demonstrates significant TIR improvements regardless of onboarding approach
- 23. Preliminary real-world outcomes among early twiist users (n=36) finds mean TIR of 80% with 2.9% TBR
- 24. Personalized diabetes treatment using AI/machine learning for AID and MDI
- 25. Glycemic outcomes among MiniMed 780G users (n=40,975) do not differ by socioeconomic status
- 26. Retrospective study finds association of CGM with improved renal outcomes in adults with CKD and insulin-treated diabetes
- 27. Dexcom G6 Pro shows high bias and reduced accuracy in ICU setting
- 28. MiniMed 780G users of seven-day infusion set report reduced burden with fewer set changes, occlusion rates, and alerts, as well as slightly improved glycemic outcomes compared to three-day set
- 29. ROUTE-T1D pilot trial demonstrated feasibility and acceptability of a CGM behavioral intervention among youth from backgrounds historically excluded from clinical T1D research
- 30. *NEW* SiBio continuous ketone monitor shows acceptable accuracy and detects benign, transient ketosis in T1D
-
Big Picture
- 31. Fireside chat with ADA CEO Mr. Chuck Henderson and actor Mr. Anthony Anderson spotlights importance of T2D activism and advocacy
- 32. Quick Take: Role of patient advocacy and research organizations
- 33. New per-protocol analysis of BURST trial of Bigfoot Unity demonstrates sustained improvements in A1c and TIR 12 months after initiation
- 34. Abbott hosts “Above the Bias” fireside chat on diabetes stigma
- 35. Richard R. Rubin Award Lecture: Dr. Edwin Fisher on prioritizing social context and peer support in diabetes care
Diabetes Therapy
1. Full results of phase 3 ACHIEVE-1 trial: Orforglipron demonstrates up to 1.6% A1c reduction and 8% weight loss
In a standing-room only hall, Dr. Julio Rosenstock (Velocity Clinical Research) presented the highly anticipated full results of the phase 3 ACHIEVE-1 trial (n=559), which studied Lilly’s oral GLP-1 RA orforglipron in T2D. Results were simultaneously published in NEJM and announced in Lilly’s press release. These results follow topline results announced in April 2025. They also build on the 26-week phase 2 trial (n=272) presented at ADA 2023, in which orforglipron demonstrated A1c reductions of 1.2%-2.1% and weight reductions of 3.7%-10%. Discontinuation due to gastrointestinal adverse events occurred in 2.2%-5.7% of participants in the orforglipron groups and no participants in the placebo group.
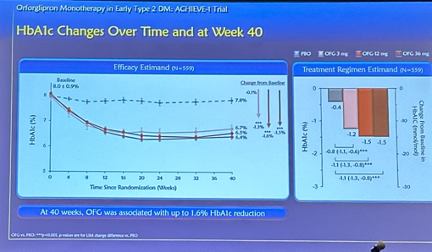
In the phase 3 ACHIEVE-1 trial, orforglipron met the primary endpoint of superior A1c reduction compared to placebo at Week 40, conferring A1c reduction of 1.3%-1.6% from a baseline of 8%, for the efficacy estimand. In key secondary endpoints, up to 76% of participants taking orforglipron achieved the A1c target of <7.0%, 66% achieved an A1c of ≤6.5%, and 26% achieved <5.7%. Notably, improvements in A1c levels were seen as early as Week 4. In another key secondary endpoint, participants taking the highest dose of orforglipron (36 mg) saw a mean weight loss of 8% (16 lbs). These findings support the potential for orforglipron, an oral small molecule and non-peptide GLP-1 RA, to substantially demonstrate A1c and weight reduction in T2D with a safety profile consistent with the overall GLP-1 RA class.
- Molecular characteristics of orforglipron. Dr. Rosenstock explained orforglipron is a differentiated candidate, as a synthetic non-peptide small molecule that activates the GLP-1 receptor. Given orforglipron is not peptide-based, it can be administered orally without fasting or restrictions on food and water intake. Orforglipron’s half-life is 29-49 hours, enabling once-daily administration in people with T2D.
- Given these characteristics and results from the ACHIEVE-1 trial, Dr. Rosenstock expressed strong confidence that orforglipron could become a “first-line” treatment for T2D. Dr. Rosenstock also anticipates the potential for orforglipron to combine well with SGLT-2 inhibitors to improve patient outcomes.
- Study design. This phase 3 trial included people with T2D, A1c of at least 7.0% but no more than 9.5%, and BMI ≥23 kg/m2. Participants were assigned randomly in a 1:1:1:1 ratio to receive orforglipron at one of three doses or placebo: (i) 3 mg (n=143); (ii) 12 mg (n=137); (iii) 36 mg (n=141); or (iv) placebo (n=138). Those who were assigned to receive orforglipron followed a dose-escalation scheme, starting with the 1 mg dose and receiving increased doses every four weeks until reaching the assigned maintenance dose. The primary endpoint was the change from baseline to Week 40 in A1c level. The key secondary endpoint was the percent change in body weight from baseline to Week 40.
- Baseline characteristics. At baseline, participants had a mean duration of T2D for 4.4 years, A1c of 8%, and body weight of 90 kg (198 lbs). The trial participants were 48% women, and participants in this trial were generally representative of the worldwide T2D population. 38% of participants had previously received a glucose-lowering agent (most commonly metformin).
- A1c reduction. At Week 40, orforglipron conferred significant A1c reduction of 1.24% with the 3 mg dose, 1.47% with the 12 mg dose, and 1.48% with the 36 mg dose, compared to 0.41% with placebo. The estimated mean difference from placebo was 0.83% with the 3 mg dose, 1.06% with the 12 mg dose, and 1.07% with the 36 mg dose.
- 68% and 73% of trial participants reached the A1c target of ≤7.0% in the 3 mg and 12 mg groups, respectively, compared to 33% in the placebo group. 57% and 62% of participants reached A1c ≤6.5% in the 3 mg and 12 mg groups, respectively, compared to 15% in the placebo group. 17% and 24% of participants reached A1c ≤5.7% in the 3 mg and 12 mg groups, respectively, compared to 4% in the placebo group.
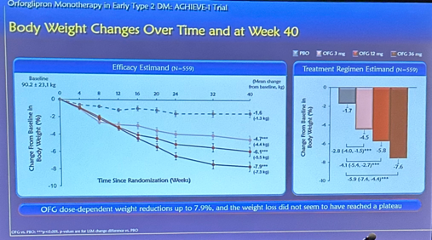
- Weight change. At Week 40, the mean percent reduction from baseline in body weight was 4.5% with the 3 mg dose, 5.8% with the 12 mg dose, and 7.6% with the 36 mg dose, compared to 1.7% with placebo. These percent changes correspond to a mean weight loss from baseline of 4.2 kg (9.3 lbs), 5.2 kg (11.5 lbs), 7.2 kg (15.9 lbs), and 1.5 kg (3.3 lbs), respectively. Orforglipron 12 mg and 36 mg were both superior to placebo, with an estimated difference from placebo in the percent change in body weight of 4.1% with the 12 mg dose and 5.9% with the 36 mg dose.
- Safety. Serious adverse events occurred in 24 participants (4.3%), including eight (5.6%) in the 3 mg group, seven (5.1%) in the 12 mg group, four (2.8%) in the 36 mg group, and five (3.6%) in the placebo group. A total of four deaths occurred during the trial, including three across the orforglipron groups and one in the placebo group. None of the deaths were related to orforglipron or placebo. The most frequently reported adverse events were gastrointestinal (diarrhea, dyspepsia, nausea, constipation, and vomiting). These events were mostly mild to moderate in severity, occurring primarily during the dose-escalation period and decreasing in prevalence over time.
- The adverse events were aligned with the overall GLP-1 RA class. Rates of diarrhea, however, were a bit higher than usual, occurring 18.9% in the 3 mg dose, 21.2% in the 12 mg dose, and 25.5% in the 36 mg dose, compared to 8.7% in the placebo group.
- At Week 40, the change from baseline in mean systolic blood pressure was 3.3-5.7 mmHg with orforglipron, compared to 1.2 mmHg with placebo. There was no effect on diastolic blood pressure.
- During the study, the overall mean alanine aminotransferase (ALT) and aspartate aminotransferase (AST) levels decreased. The percentage of participants with a post-baseline ALT or AST level three times the upper limit of the target range or higher was similar among trial groups. Additionally, levels returned to normal or near the baseline level in all participants treated with orforglipron.
- Translational perspectives. Acknowledging the limitations of interpreting results across trials without a head-to-head study, Dr. Rosenstock provided comparisons of results from orforglipron in ACHIEVE-1 to oral semaglutide in PIONEER-1 and injectable semaglutide in SUSTAIN-1 in A1c and body weight reduction. See the figures below.
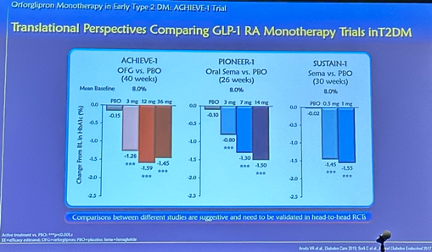
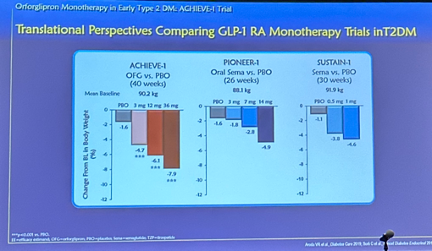
- Ongoing clinical development of orforglipron. In addition to ACHIEVE-1, several additional trials are studying orforglipron, including:
- ACHIEVE-2 (n=962): This trial is studying orforglipron vs. dapagliflozin in people with inadequate glycemic control on metformin.
- ACHIEVE-3 (n=1,698): This trial is studying orforglipron vs. oral semaglutide in people with inadequate glycemic control on metformin.
- ACHIEVE-4 (n=2,749): This CVOT is studying orforglipron vs. insulin glargine in people with overweight or obesity, increased cardiovascular risk, and inadequate glycemic control on one to three oral agents.
- ACHIEVE-5 (n=546): This trial is studying orforglipron vs. placebo in people with inadequate glycemic management on insulin glargine, with or without metformin and/or an SGLT-2 inhibitor.
2. GIP receptor agonism and antagonism: Dr. Dan Drucker on why both seem to be effective; impact of incretins on neurodegenerative disease
Dr. Dan Drucker (Mt. Sinai Hospital, Canada) outlined thought-provoking hypotheses for why GIP receptor agonism and antagonism both appear to be effective, drawing on an article by Duke’s Dr. Jonathan Campbell and he published just this month. Additionally, he reviewed the research around the neuroprotective effects of incretin therapies and their potential mechanisms.
- GIP receptor agonism and antagonism – how can both be effective? Nearly most incretins in development or on the market rely on receptor agonism. Emerging evidence that antagonism is effective (e.g., Amgen’s MariTide) has prompted discussion on these seemingly contradictory approaches. Four hypotheses are summarized in the image below.
- Top left: Chronic agonism (i.e., activation) of the GIP receptor can lead to desensitization of the receptor, thus resembling the effects of a GIP antagonist. However, there is no evidence that tirzepatide, a dual GIP/GLP-1 RA, attenuates activity in neurons with GIP receptors.
- Top right: Antagonism of GIP receptors on inhibitory GABAergic neurons may result in the disinhibition of neurons with GLP-1 receptors (i.e., these GLP-1 receptor-expressing neurons are activated since they are not inhibited by the GABAergic neurons).
- Bottom left: GIP receptor agonism increases the activity of inhibitory GABAergic neurons, and such inhibitory activity may itself be responsible for decreased food intake.
- Bottom right: Loss of GIP receptor activity over time (e.g., due to genetic or pharmacological reasons) leads to an increase in GLP-1 receptor sensitivity due to disinhibition or compensation.
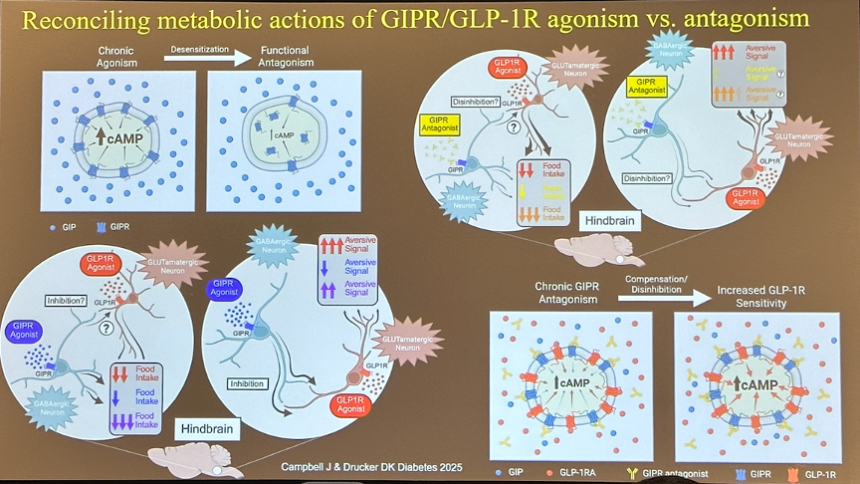
- Potentially illuminating data to come at ADA. Dr. Drucker expressed interest in the full phase 2 results for MariTide, which will be presented on the final day of ADA. Recall that MariTide is a once-monthly dual GIP receptor antagonist/GLP-1 RA. Topline results reported in November 2024 revealed that MariTide conferred ~20% weight loss in people with obesity or overweight and ~17% weight loss in T2D at 52 weeks, with no signs of a plateau in weight loss.
- Evidence for the neuroprotective effects of GLP-1 RAs date back to research over 20 years ago. Dr. Drucker said that research in this area is not new, highlighting research from the early 2000s that found greater seizure severity and brain injury in GLP-1 RA receptor deficient mice. In humans, such a deficiency also leads to seizures and not obesity or diabetes. Further, Dr. Drucker noted that a common observation seen in cardiovascular outcomes trials (e.g., REWIND for dulaglutide) for GLP-1 RAs is stroke risk reduction.
- GLP-1 RAs likely improve brain health through multiple direct and indirect actions on the central nervous system. For example, GLP-1 RAs have been shown in mice to act on receptors in the brain to decrease appetite and reduce neuroinflammation. Another study found that GLP-1 receptors in the brain may reduce systemic inflammation by reducing TNF-α, a cytokine involved in inflammation. The GLP-1 RA exenatide has also been shown to strengthen the blood-brain barrier in aged mice by decreasing leakage.
- GLP-1 RAs for Alzheimer’s disease – upcoming clinical trial completions and evidence from mouse models. Dr. Drucker highlighted Novo Nordisk’s phase 3 EVOKE and EVOKE+ trials investigating semaglutide in people with early-stage Alzheimer’s disease, saying that the results, which are expected by the end of this year, will be “unbelievably informative” for the future of the field. He added that both studies are “not small trials” – each has an estimated enrollment of 1,840 participants.
- One study that used a lipopolysaccharide mouse model to induce neuroinflammation, characteristic of Alzheimer’s disease, found that semaglutide attenuates this inflammation, reducing the number of microglia and the expression of co-regulated inflammatory genes in the hippocampus. Other research has shown that such a mouse model closely mimics the C1 subtype of Alzheimer’s disease in humans, suggesting promise for semaglutide’s efficacy beyond mice. Still, there are other studies that have found that GLP-1 RAs semaglutide and tirzepatide are ineffective in other mouse models of Alzheimer’s disease (e.g., 5XFAD and AAP/PS1).
3. Full results of phase 3b STEP-UP trial: High-dose semaglutide 7.2 mg confers greater weight loss than semaglutide 2.4 mg in people with obesity
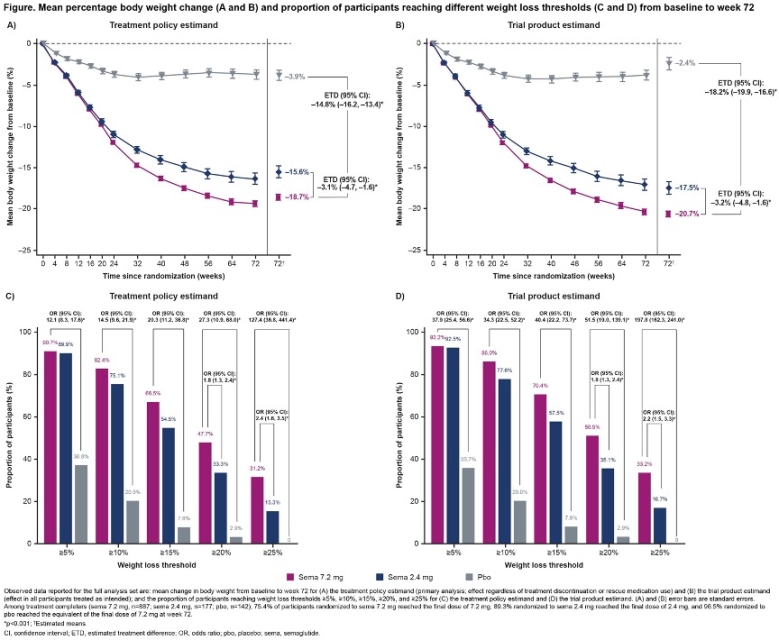
In this late-breaking session, Dr. Sean Wharton (University of Toronto, Canada) presented full results of the phase 3b STEP-UP trial (n=1,407) evaluating high-dose semaglutide (7.2 mg) in people with overweight or obesity without T2D. See Novo Nordisk’s press release. Previously, topline results from January 2025 showed that semaglutide 7.2 mg conferred statistically significant and superior weight loss compared to semaglutide 2.4 mg and placebo, with a safe and well-tolerated profile. Assuming all participants adhered to treatment, semaglutide 7.2 mg achieved weight loss of 20.7% at Week 72, compared to 17.5% with semaglutide 2.4 mg and 2.4% with placebo, from a mean baseline body weight of 113 kg (249 lbs). Furthermore, one-third (33.2%) of people treated with semaglutide 7.2 mg achieved a weight loss of ≥25%, compared to 16.7% with semaglutide 2.4 mg and none with placebo. When applying the treatment policy estimand, people treated with semaglutide 7.2 mg achieved weight loss of 18.7%, compared to 15.6% with semaglutide 2.4 mg and 3.9% with placebo.
- Baseline characteristics and trial design. At baseline, participants were on average 47 years old, with 74% being female. Baseline weight was 113 kg (249 lbs), and waist circumference was 119 cm (47 in). Participants were randomized 5:1:1 to semaglutide 7.2 mg, 2.4 mg, and placebo.
- Results. Most participants reached the maximum dose: (i) 75% for semaglutide 7.2 mg; (ii) 89% for 2.4 mg; and (iii) 97% for placebo. As announced in the topline results, semaglutide 7.2 mg conferred 21% weight loss, compared to 18% with 2.4 mg and 2% with placebo, assuming all participants adhered to treatments. Over 90% receiving either dose of semaglutide achieved weight loss ≥5% (vs. 37% with placebo). Nearly two-thirds of those receiving semaglutide 7.2 mg achieved weight loss ≥15% (vs. 55% with 2.4 mg and 8% with placebo). Finally, over 31% of those taking semaglutide 7.2 mg experienced ≥25% weight loss (vs. 15% with 2.4 mg and 0% with placebo). Waist circumference decreased significantly with semaglutide 7.2 mg vs. placebo (p<0.001).

- Safety and tolerability. The safety and tolerability profiles were similar to those of semaglutide 2.4 mg. Most adverse events were gastrointestinal. 71% of those taking semaglutide 7.2 mg experienced GI side effects (vs. 61% with semaglutide 2.4 mg and 43% with placebo). Discontinuation rates were 3.3%, 2.0%, and none, respectively. Finally, serious adverse event rates were 6.8%, 10.9%, and 5.5%, respectively.
4. Post hoc analyses of COMBINE 1, 2, and 3 trials assess CGM metrics of IcoSema and glycemic benefits across kidney function
Profs. Linong Ji (Peking University, China), Concetta Irace (Università Magna Graecia di Catanzaro, Italy), and Christophe De Block (University Hospital Antwerp, Belgium) presented post hoc analyses of COMBINE 1, 2, and 3 trials, which evaluated Novo Nordisk’s IcoSema (fixed-dose combination of once-weekly insulin icodec + semaglutide) in people with T2D. Previously, full results of these trials were presented at EASD 2024. In COMBINE 1 (IcoSema vs. insulin icodec in T2D; n=1,291) and COMBINE 2 (IcoSema vs. semaglutide 1.0 mg in T2D; n=683), IcoSema conferred a superior A1c reduction vs. insulin icodec or semaglutide alone. In COMBINE 3 (IcoSema vs. insulin glargine U100 + insulin aspart in T2D; n=679), IcoSema conferred a non-inferior A1c reduction to daily basal-bolus treatment. Currently, IcoSema is approved for T2D in Japan and is under review in the EU. Novo Nordisk has not submitted for regulatory approval to the US FDA, which issued a Complete Response Letter in July 2024 regarding the manufacturing process and indication in T1D.
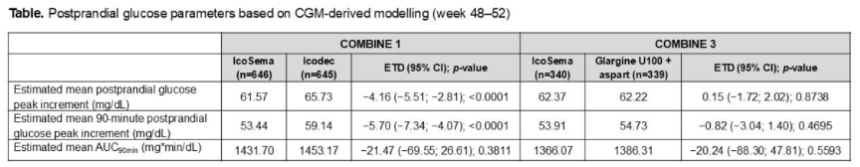
- CGM metrics. Two post hoc analyses of COMBINE 1 and 3 trials, presented by Profs. Irace and De Block evaluated the effects of IcoSema on glucose metabolism (measured by Dexcom G6), compared to other insulin therapies. IcoSema was associated with statistically significantly lower postprandial glucose (PPG) (4.2 mg/dL; p<0.0001) and 90-minute PPG peaks (5.7 mg/dL; p<0.0001), compared to insulin icodec alone. IcoSema and basal-bolus therapy (insulin glargine + insulin aspart) showed no statistically significant difference in PPG. Similarly, the mean percentage of Time in Tight Range (TITR) was higher with IcoSema vs. insulin icodec (8.5% or 2.1 hours; p<0.0001) and similar vs. basal-bolus therapy (p=0.81). The duration of hypoglycemic episodes was similar between IcoSema and comparators. See tables below for full results.
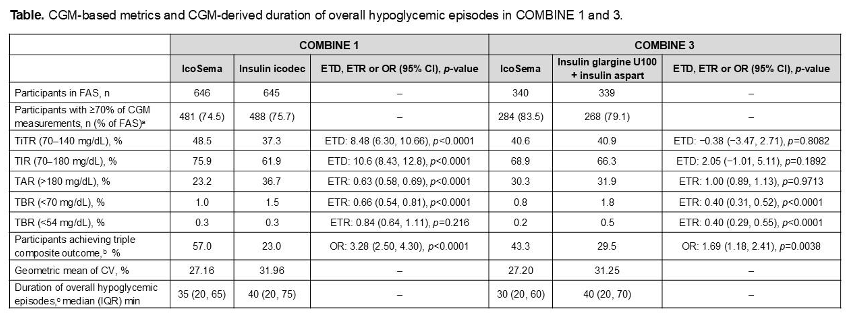
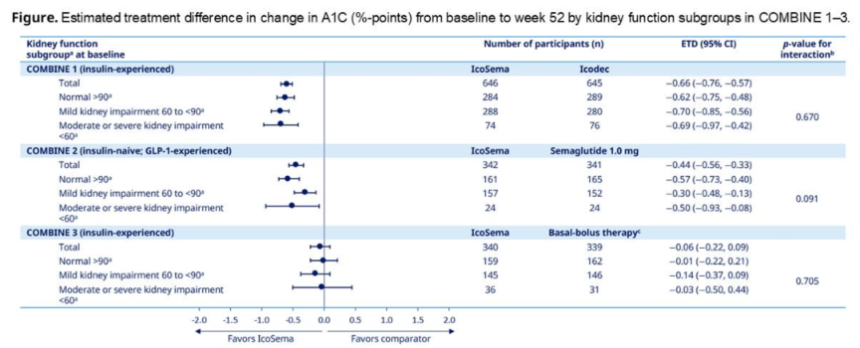
- IcoSema in T2D by kidney function. Another post hoc analysis of COMBINE 1, 2, and 3 trials, presented by Prof. Ji, assessed treatment outcomes of IcoSema vs. comparators (insulin icodec, semaglutide, and basal-bolus therapy, respectively) in adults with T2D by kidney function at baseline (eGFR ≥90; 60-90; 30-60; <30; all mL/min/1.73 m2). The estimated treatment differences for A1c reduction, hypoglycemia, and achievement of A1c <7.0% were all consistent across kidney function subgroups, with no clear trends.
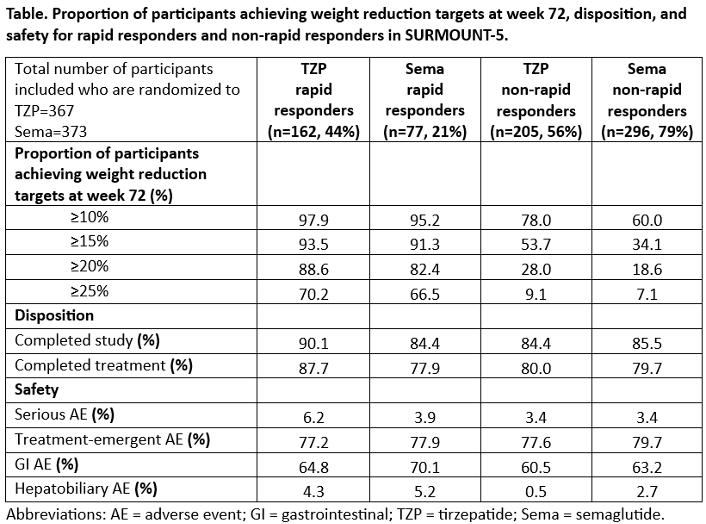
5. Post hoc analysis of SURMOUNT-5 trial raises concerns over “excessively” rapid weight loss
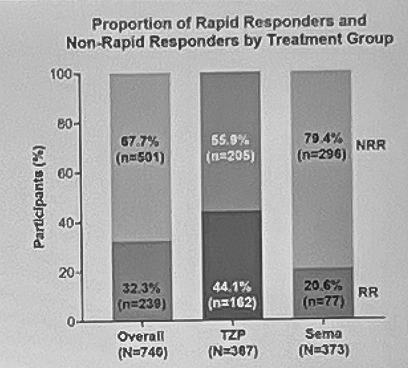
In this standing room-only presentation, Dr. Louis Aronne (Weill Cornell Medical College) shared a post hoc analysis of the phase 3b SURMOUNT-5 trial that characterized rapid responders to tirzepatide and semaglutide. As background, the SURMOUNT-5 trial (presented at ECO 2025 and published on NEJM) found that tirzepatide confers 20.2% weight loss vs. 13.7% with semaglutide in people with obesity or overweight with weight-related comorbidities from a baseline weight of 113 kg (249 lbs). This post hoc analysis further investigated the efficacy and safety of tirzepatide and semaglutide in rapid and non-rapid responders.
- Rapid responders were defined as those who reached ≥15% weight loss by Week 24, which marks the end of the dose titration period. This definition was informed by the top tertile of all participants’ weight reduction. Overall, 32% of participants were considered rapid responders. More specifically, 44% of people taking tirzepatide and 21% of those taking semaglutide were considered rapid responders (see figure below). The median time to reach ≥15% weight loss was 36 and 52 weeks for tirzepatide and semaglutide, respectively.
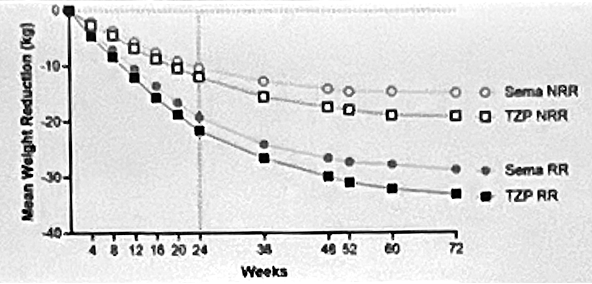
- Rapid responders were more likely to be female and have lower weight at baseline. The average age (44 years) and duration of obesity (15 years) at baseline were similar between rapid and non-rapid responders. However, rapid responders had a higher proportion of females than non-rapid responders (74% vs. 60%). Dr. Aronne said that this follows previous findings that women have been shown to lose more weight from incretin-based therapies. As well, rapid responders had lower average weight (107 kg or 236 lbs vs. 116 kg or 256 lbs) and BMI (38 vs. 40 kg/m2) at baseline. He said that people with lower weight at baseline may have greater sensitivity to these incretins.
- Rapid responders lost more weight than non-rapid responders. As shown in the table below, 94% of rapid responders taking tirzepatide (vs. 54%) and 91% of rapid responders taking semaglutide (vs. 34%) reached ≥15% weight loss. Likewise, 70% of rapid responders taking tirzepatide (vs. 9%) and 67% of rapid responders taking semaglutide (vs. 7%) experienced ≥25% weight loss.
- Safety events were slightly higher for rapid responders. GI side effects were numerically higher in rapid responders by 4%-7%. Hepatobiliary (liver, gallbladder, and bile ducts) adverse events were two to eight times higher among rapid responders. Dr. Aronne noted that rapid weight loss can increase the risk of gallstones and urged that there should be more preventative measures in place for both clinical trials and real-life practice. However, treatment-emergent adverse events were similar in rates, and rapid responders were more likely to complete the study or treatments.
- During the discussion, Dr. Aronne challenged the audience that rapid response may not be necessarily “better,” and proposed that the current titration regimen might even be “excessive” in some patients. There may be diminishing returns on the speed and amount of weight loss. For instance, at some point, patients might lose more lean mass than fat mass. Hence, he emphasized the importance of assessing body composition, which is certainly an evolving field with candidates on the horizon. Dr. Aronne also encouraged slower titration for rapid responders and being comfortable with using non-maximal doses as the highest dosage if patients are responding well to low doses. Finally, he stated that semaglutide and tirzepatide – whether high or low in dosage – offer cardiometabolic benefits, but more trials should be done to study these effects at varying doses.
6. Addressing CV and kidney disease in T1D: Guidelines, GLP-RAs, and SGLT-2 inhibitors
In an afternoon session chaired by Prof. Chantal Mathieu (KU Leuven, Belgium), panelists shared insightful presentations on addressing CV and kidney disease in T1D. Despite major strides in T2D research, the field’s focus on T1D remains underserved, with limited clinical trials and no approved therapies specifically targeting cardiorenal protection. The session highlighted the high prevalence of CVD and CKD in T1D, the potential for emerging therapies (e.g., GLP-1 RAs, SGLT-2 inhibitors, and non-steroidal MRAs), and the importance of translating evidence across populations.
- Dr. Camila Acevedo (University of Missouri) started the symposium with an overview of guidelines for managing CV and kidney disease in T1D. She highlighted the statistic that people with T1D have a life expectancy about 13 years shorter than the general population, primarily due to increased risk of CV events. While there’s been an encouraging decline in overall CV risk in recent years, high risks in T1D remain, with no randomized trials focused specifically on CV risk reduction in this population. Given results from retrospective and observational studies, Dr. Acevedo emphasized the importance of the following:
- Intensive glycemic management. Findings from the DCCT/EDIC cohort showed that intensive glycemic management leads to decreased risk of macrovascular and microvascular complications. Notably, intensive intervention continued to show CV benefits more than 30 years later, known as the “legacy effect.” Early and intensive glycemic management, therefore, sets the foundation for long-term CV health, and ongoing management of traditional CV risk factors remains essential to maintain those benefits.
- Lipid management. A 2016 observational study (n=24,230) showed that the use of lipid-lowering treatment confers a 22% reduction in CVD risk and a 44% reduction in CV death. While guidelines from the ADA, ACC, and ESC vary slightly due to the lack of clinical trials in T1D, most recommendations encourage: (i) lifestyle modification; (ii) at least moderate intensity statin for ages 40-75 and high intensity for those with higher CVD risk; and (iii) consideration of diabetes duration, CVD risk, and LDL levels in younger populations. On statins, Dr. Acevedo explained how, despite negative perception in the public, statins remain one of the most effective treatments for preventing CVD in T1D.
- Blood pressure management. Hypertension affects nearly 40% of people with T1D, often alongside signs of kidney disease with albuminuria and reduced eGFR. As the ADA guidelines suggest, HCPs should tailor treatment to the patient’s initial blood pressure, starting with one or two antihypertensive agents based on baseline levels. HCPs should also reassess patient outcomes after three to six months, monitoring for side effects and ensuring treatments are both effective and well-tolerated. If conditions don’t improve, Dr. Acevedo recommends adding a mineralocorticoid receptor antagonist (MRA).
- Dr. Ildiko Lingvay (UT Southwestern) continued the discussion in T1D, focusing on reducing CVD risk with GLP-1 RAs. Dr. Lingvay started her presentation by asking the audience whether people with T1D should be prescribed GLP-1 RAs for cardiorenal protection. In response, the overwhelming majority of the audience raised their hands, agreeing that the benefits outweigh the risks. A modest number of people shared their inclination to stick with the treatment label, and nobody raised their hands that the risks outweigh the benefits. Setting the stage for her presentation, Dr. Lingvay emphasized that there have been several advancements, including expanding indications for effective treatments (e.g., GLP-1 RAs and SGLT-2 inhibitors), but there hasn’t been a single treatment approved for T1D.
- GLP-1 RAs. Based on clinical trials, GLP-1 RAs have shown a reduction in MACE, all-cause mortality, heart failure-related hospitalizations, and kidney disease progression, independent of glucose lowering. Interestingly, recent data highlight no difference in benefit based on diabetes status, suggesting the cardioprotective effects are based on mechanisms beyond glycemic management, making GLP-1 RAs a compelling off-label option in T1D with high residual risk. For example, in the phase 3 SUMMIT trial (n=731), tirzepatide demonstrated a 38% risk reduction in CV death or worsening heart failure at 52 weeks.
- SGLT-2 inhibitors. While the FDA has rejected SGLT-2 inhibitors for T1D due to DKA risk, this drug class has conferred clinically meaningful improvements in glucose variability, heart failure outcomes, and CKD progression. Dr. Lingvay suggested that DKA risks could potentially be mitigated with continuous ketone monitoring.
- Prescription trends demonstrate a rising unmet need. According to a JAMA study published in October 2024, the percentage of people with T1D prescribed with GLP-1 RAs or SGLT-2 inhibitors increased from 0.7% in 2010 to 8.3% in 2023. While these treatments are not approved for use in T1D, people with T1D continue seeking them for obesity, CVD, and CKD. These findings underscore the need for trials focused on patient outcomes in T1D.
- Prof. Hiddo Heerspink (University of Groningen, Netherlands) concluded the session with a presentation on reducing CKD in T1D with SGLT-2 inhibitors and non-steroidal MRAs. He reminded that CKD affects 27% of people with T1D, and one in four will develop kidney failure within 30 years of diagnosis. Despite this significant burden driven by metabolic, hemodynamic, and inflammatory mechanisms, most clinical trials focus on T2D, creating a treatment gap for kidney protection strategies in T1D.
- Long-term effects of SGLT-2 inhibitors on kidney function. In the T1D subgroup (n=73) of the EMPA-KIDNEY trial, SGLT-2 inhibitor empagliflozin slowed eGFR decline to 0.23 mL/min/1.73 m²/year, compared to 2.78 mL/min/1.73 m²/year with placebo. While the sample size was small, Prof. Heerspink said these findings suggest that empagliflozin may have meaningful renal benefits in people with T1D.
- Ongoing FINE-ONE trial in CKD and T1D. Encouragingly, the FINE‑ONE trial (expected to complete later in 2025) represents a pivotal step toward positioning and non-steroidal MRA finerenone as a kidney-protective treatment for T1D and CKD. Regulators have accepted albuminuria reduction as a surrogate for CKD progression intervention, allowing albuminuria to serve as a bridging biomarker to translate evidence for kidney protection from T2D to T1D. Prof. Heerspink stressed that people with T1D need treatments like those with T2D, and expressed high hopes that finerenone could potentially shift the treatment paradigm.
7. STRIDE trial: New post hoc analyses show consistent functional benefit across subgroups for semaglutide in people with T2D and peripheral artery disease
Drs. Zaina Albalawi (Memorial University of Newfoundland, Canada), Alice Cheng (University of Toronto), Neda Rasouli (University of Colorado), and Subodh Verma (St. Michael’s Hospital, Canada) discussed the STRIDE trial and shared new findings from a post hoc analysis. Recall that STRIDE (n=792) investigated semaglutide 1.0 mg (Ozempic) in people with T2D and early-stage symptomatic peripheral artery disease (PAD), which is characterized by muscle pain in the legs during activity. As background on PAD, the condition affects over 230 million individuals and accounts for 16.2% of all first CV manifestations of T2D. Currently, cilostazol is the only class I treatment available for claudication (i.e., muscle pain during physical activity), and besides its poor tolerability, it is contraindicated in heart failure and offers no CV benefits.
Presented earlier this year at ACC 2025, STRIDE results showed that, at 52 weeks, semaglutide conferred a 21% increase in maximum walking distance from a baseline of 185 meters vs. an 8% increase with placebo from a baseline of 186 meters. Notably, Dr. Subodh pointed out that the ankle-brachial index, one of the supportive secondary outcomes, already showed an improvement at 52 weeks, suggesting its involvement in semaglutide’s mechanism of action.
Today’s post-hoc analysis examined whether maximum walking distance (the primary outcome) and pain-free walking distance was consistent across subgroups. Specifically, the subgroup analysis explored diabetes duration (≥10 vs. <10 years), A1c (≥7% vs. <7%), BMI (≥30 vs <30 kg/m2), and concomitant medication use (SGLT-2 inhibitors or insulin).
- Subgroup distribution. More than half of participants had a diabetes duration ≥10 years (61% vs. 39%) and an A1c ≥7% (56% vs. 44%). Of note, the majority of participants did not have obesity (59% vs. 41%). Most participants were not on an SGLT-2 inhibitor (65% vs. 35%) or insulin (68% vs. 32%).
- Results. The effects of semaglutide on maximum walking distance and pain-free walking distance were consistent across all subgroups. Additionally, correlation analyses suggested that the functional benefits observed in the study were not correlated with the weight loss and glycemic improvements also seen in the study.
- Dr. Cheng offered five key takeaways from STRIDE for endocrinologists:
- Remember to screen patients for PAD – this may involve knowing where ankle-brachial index testing is available;
- Be careful about sarcopenia – while Dr. Cheng expressed concern over the significant decreases in A1c and body weight from low baselines, she noted that participants in the study improved functional outcomes, giving her confidence that sarcopenia risk likely is not a major issue;
- The study findings support semaglutide’s effect in reducing atherosclerosis;
- Functional outcome measures are important to consider – she pointed out that patients often care more about functional outcomes than hard outcomes because they are more practical, patient-centered, and measurable; and
- These findings add to the growing evidence base for GLP-1 RA use in T2D (e.g., STEP-HFpEF DM and FLOW).
- Future analyses of STRIDE. Dr. Verma shared that there will be more new data from STRIDE coming this year, specifically investigating the impact of smoking. For context, smoking is a risk factor for PAD as it can damage blood vessels, promote blood clot formation, and affect circulation. STRIDE included non-smokers, past smokers, and current smokers. Further insights from STRIDE were also published in Diabetes Care.
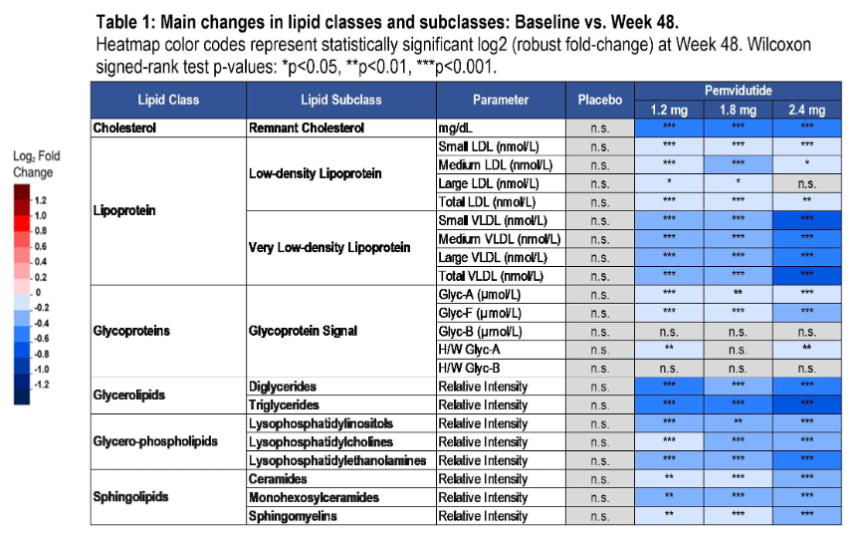
8. GLP-1/glucagon RA pemvidutide reduces cardio-inflammatory lipid levels in people with obesity or overweight
In this oral presentation, Dr. John Suschak (Altimmune) highlighted findings from the phase 2 trial (n=391) of GLP-1/glucagon RA pemvidutide in people with overweight or obesity. Previously, topline results in December 2023 showed that pemvidutide conferred mean weight loss up to 16% at Week 48, with trajectories indicating continued weight loss with continued treatment. The mean baseline BMI was 37.4 kg/m2. This analysis aimed to understand the effects of pemvidutide on lipid panels, which are inflammatory and increase the risk of CVD.
- Pemvidutide lowered total cholesterol, LDL cholesterol, and triglycerides by up to 15%, 11%, and 35%, respectively, in 254 participants whose metabolomic profiling was conducted. Among proinflammatory phospholipids and sphingolipids, ceramides, which are associated with myocardial cell death, decreased by 21% (vs. 0.6% with placebo). Lysophosphatidylcholine, known to contribute to atherosclerotic lesions and hepatic LDL accumulation, was also decreased by 20% (vs. 1% with placebo). Finally, as shown in the table below, NMR spectroscopy showed that pemvidutide reduced small LDL cholesterol and small very low-density lipoprotein (vLDL), which penetrates the arterial wall more easily and creates plaques, and Glyc Acetylation (Glyc-A), a biomarker for heart failure and CVD. Patients who had elevated serum baseline lipid levels experienced even greater reductions. Ultimately, Dr. Sushak emphasized that these improvements in lipid profile suggest cardiometabolic benefits of pemvidutide.
- In November 2024, Altimmune announced a phase 3 VELOCITY program, following a successful end-of-phase 2 meeting with the FDA. Four phase 3 trials will evaluate the efficacy of pemvidutide on: (i) weight loss in overweight or obesity without diabetes; (ii) weight loss and serum lipids in overweight or obesity with elevated LDL cholesterol levels; (iii) weight loss in overweight or obesity and elevated liver fat; and (iv) weight loss, lean mass preservation, and daily functions in obesity or overweight, including elderly participants with sarcopenia, an age or immobility-related muscle loss
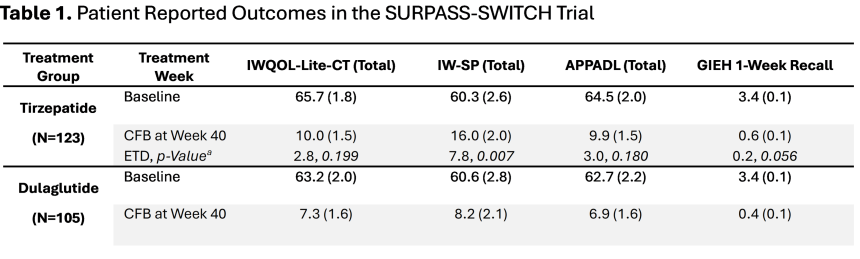
9. Further analyses of SURPASS-SWITCH trial show superior A1c and weight reduction in people switching from dulaglutide to tirzepatide
In this oral presentation session, Drs. Anita Kwan and Kristina Boye (Lilly) shared additional analyses of the phase 4 SURPASS-SWITCH study (n=282), which compared switching from dulaglutide (0.75 mg, 1.5 mg) to tirzepatide 15 mg vs. escalating dulaglutide dose to 4.5 mg[2] in people with T2D taking dulaglutide and three or fewer oral antihyperglycemic agents. Full results of this trial were published in Annals of Internal Medicine this year, showing that switching to tirzepatide confers significantly greater A1c reduction (1.4%) from baseline of 7.8% at Week 40, compared to escalating dulaglutide (0.7%). Weight loss was significantly higher in people taking tirzepatide (10.5 kg vs. 3.6 kg) from baseline of 97 kg (214 lbs). The rates of serious adverse events were similar (7.2% vs. 7.0%), with the most common events being mild-to-moderate nausea and diarrhea.
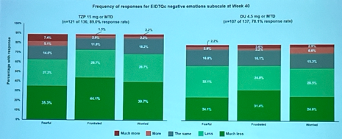
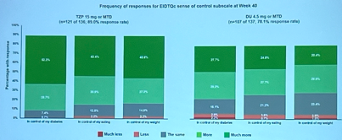
- A subgroup analysis, presented by Dr. Kwan, showed that switching to tirzepatide confers two-to-three times greater A1c and weight reduction than escalating dulaglutide across all baseline subgroups – including by A1c, duration of T2D, baseline dulaglutide dose (0.75 mg, 1.5 mg), and duration of dulaglutide dose. See figure below for the comparison.

- Patient-reported outcomes, presented by Dr. Boye, showed that switching to tirzepatide significantly improved the impact of weight on self-perception (IW-SP, p=0.007) and body weight-related self-perception. While not statistically significant, switching to tirzepatide showed numeric improvements in the impact of weight on quality of life (IWQOL-Lite-CT, p=0.199), ability to perform physical activities of daily living (APPADL, p=0.18), and global impression of emotional health (GIEH, p=0.056). These results demonstrate that switching to tirzepatide significantly improves self-perception related to weight and numerically improves physical ability and emotional health. Dr. Boye especially pointed out that tirzepatide allows patients to worry less and feel a greater sense of agency in managing their disease and weight, according to the Emotional Impact of Diabetes Treatment Questionnaire comparison (EDITQc).
10. ADA Presidents’ selected oral presentation features a retrospective cohort study on the association of age at T1D diagnosis with cardiovascular and kidney disease
Selected by the ADA Presidents, Dr. Araz Rawshani (University of Gothenburg, Sweden) and his team’s retrospective study (n=34,155) focused on the association of age at T1D diagnosis with fatal cardiovascular and kidney events. He emphasized the importance of this work, highlighting that people with T1D have a greater risk of complications and reduced life expectancy compared to those without diabetes. Given this association, Dr. Rawshani has hypothesized that age at T1D diagnosis impacts clinical complications and mortality risk – supporting the case for delaying T1D onset in youth.
- Methods. The nationwide retrospective cohort study included individuals ≥18 years of age with T1D with at least one entry in the Swedish National Diabetes Register (NDR) from 1998 through 2019. The study excluded individuals with T1D with coexisting congenital, metabolic, or neurological disorders. Cox proportional hazards models were used to determine the relationships between age at T1D diagnosis and fatal and nonfatal outcomes (e.g., cardiovascular mortality, acute myocardial infarction, peripheral arterial disease, end-stage renal disease).
- Results. The study participants had a mean age of 31 years and a median follow-up of 13 years. Younger age at T1D diagnosis was associated with a higher risk of mortality and nonfatal outcomes. T1D diagnosis at less than 10 years of age was associated with 16 life-years lost. The hazard ratio (HR) per every one-year increase in age at T1D diagnosis was 0.97 for cardiovascular mortality, indicating a 2.6% lower risk for every one-year increase in age at diagnosis. Additionally, the HRs per one-year increase in age at T1D diagnosis were 0.99 for non-cardiovascular mortality, 0.96 for end-stage kidney disease, 0.97 for peripheral artery disease, 0.97 for acute myocardial infarction, 0.97 for heart failure, and 0.97 for coronary heart disease. Overall, T1D diagnosis before 10 years was associated with the greatest risk of cardiovascular mortality (HR: 5.7).
- Conclusion. Based on these findings, Dr. Rawshani emphasized that the early onset of T1D carries an increased risk of significant complications and mortality. These findings support the growing awareness that delaying T1D onset in youth may reduce the risk of complications and early mortality.
11. Addressing residual cardiovascular risk in people with diabetes and cardiovascular disease
An energetic morning session urged multifactorial approaches to reducing cardiovascular disease (CVD) risk. Prof. Mohammed Al-Sofiani (King Saud University, Saudi Arabia), Dr. Irl Hirsch (University of Washington), and Dr. Vanita Aroda (Harvard University) stressed the need to continue treating CVD in three different cases. Prof. Al-Sofiani discussed the importance of lipid management in addition to achieving LDL cholesterol goals, Dr. Hirsch considered glycemic metrics beyond A1c and their impact on CVD risk, and Dr. Aroda proposed a reframing of residual risk as personal risk, an essential factor.
- Prof. Al-Sofiani discussed lipid management beyond LDL goals in people with diabetes. It has been established that people with diabetes have greater residual cardiovascular (CV) risk even with an LDL cholesterol value less than 30 mg/dL, which is considered very low among the broader population. This indicates that other factors may drive residual CV risk among this population.
- Lipid management should target HDL functionality as opposed to quantity. Previous research has explored the option of increasing HDL levels to reduce CV events, finding no significant difference in CV. However, raising HDL levels does improve HDL functionality, as HDL collects excess cholesterol from tissues and transports it to the liver for breakdown. This effect may improve overall CV health.
- The risk of CVD increases with elevated fasting triglycerides in statin-treated patients. Patients with low LDL cholesterol values as a result of statin use may still have significant CV risk due to elevated triglyceride levels, said Prof. Al-Sofiani. The Reduce-IT trial demonstrated that icosapent ethyl, a purified omega-3 fatty acid, reduces the risk of CVD events in patients also on statin therapy, supporting the combined use of these treatments.
- The field of Lp(a) research continues to expand and provides promise for further reducing CVD risk. Lp(a) is an LDL-like particle with apo(a), a protein, bonded to an apoB protein core. Lp(a) has been shown to be five to six times more atherogenic (promoting the formation of arterial plaques) than LDL and therefore represents a crucial CVD risk factor even at target LDL values. Prof. Al-Sofiani said that niacin, PCSK9 inhibitors, estrogen/lomitapide, and lipoprotein apheresis are effective treatments to lower Lp(a) levels, with many more therapeutics in development. In all, Prof. Al-Sofiani urged the audience to consider broader lipid management beyond lowering LDL levels alone.
- Dr. Hirsch then turned to the effect of glycemic metrics beyond A1c on CV risk among people with diabetes. He began with discordance between glycemia management index (GMI) and A1c. For instance, a 53-year-old African American woman may have an A1c of 7.4% but a GMI calculated at 6.4%. Before intensively lowering A1c, Dr. Hirsch urged the audience to consider that Black individuals may have higher A1c values than white individuals with the same blood glucose values, with a discrepancy of 0.4%-0.5% observed in some cases. Factors contributing to this are individual variations in red blood cell survival times and glycation rates. He emphasized that there is “nothing perfect about GMI,” but in this case, it provides further insight into glycemic management in such a patient. As the patient already experiences high rates of hypoglycemia at 2.8% Time below Range (TBR), providers should instead focus on LDL lowering in this case and deem her glycemic management satisfactory. With regard to cardiovascular risk, hypoglycemia leads to increased heart rates and blood carbon monoxide levels, among many other factors, leading to CVD.
- There is a significant connection between hypoglycemia rates and sudden death rates. Given this relationship, Hirsch firmly stated that A1c is not the ideal metric to assess hypoglycemia. He said that the CGM metric TBR is essential for understanding hypoglycemia and that TBR should be closer to zero, as opposed to the common standard of 4%. A1c management can be “less stringent” to improve rates of hypoglycemia with powerful CV benefits.
- Finally, he discussed a strong relationship between high glycemic variability and hypoglycemia as demonstrated by studies such as Diamond T1D. Dr. Hirsch recommended the use of AID in people with T2D and glycemic variability that approaches an elevated 40% coefficient of variation. Again, the increased risk of CVD caused by hypoglycemia must be aggressively managed, and Dr. Hirsch said that AID is a powerful tool to reduce glycemic variability. He urged the room to act on interventions for long-term CV benefits.
- Dr. Aroda discussed the essential role of inflammation in CVD and approaches to detection and management. Inflammation is often considered a “residual” risk of CVD, defined as a remaining factor after the achievement of targets for inherent risks such as LDL cholesterol, blood pressure, and glycemia. Dr. Aroda said that despite the benefits of LDL cholesterol lowering, 60-80% of the residual CVD risk remains. This large contribution to CVD risk leads Dr. Aroda to deem “residual” a misnomer – residual risk is rather a core, root cause of elevated LDL, blood pressure, and glycemia. She instead encourages the use of “personalized risk” to emphasize the importance of treating residual risk factors.
- Inflammation plays a key pathogenic role in all stages of atherosclerosis (arterial plaque formation). In the formation of new arterial plaques, inflammatory mediators in the blood inhibit collagen synthesis, leading to a greater likelihood of atherosclerotic lesion rupture with serious CV consequences. As the disease progresses, risk factors such as smoking, the gut microbiome, visceral adipose tissue, and diabetes mediate inflammation and drive further plaque progression. Targeting such drivers of inflammation may provide prognostic, diagnostic, and therapeutic benefits.
- Providers should carefully consider what exactly they are targeting with respect to inflammation. Dr. Aroda described obesity as one of the most prevalent proinflammatory conditions worldwide. 2022-2023 studies demonstrated that high levels of weight loss through the use of GLP-1 RAs such as semaglutide and tirzepatide lead to reduced inflammatory markers, and lipid-lowering medications may also reduce inflammation overall.
12. SGLT-2 inhibitors increase risk for hospitalization from DKA by 40% vs. GLP-1 RAs in patients with T2D and CKD
In this poster session, Dr. Panupong Hansrivijit (Harvard University) explored the increased risk of diabetic ketoacidosis (DKA) due to SGLT-2 inhibitors in patients with chronic kidney disease (CKD). The study was a large retrospective cohort study (n=143,858), comparing adults with T2D and CKD stages 3-4 who initiated SGLT-2 inhibitors versus GLP-1 RAs. Using data pooled from three large US claims databases, the study compared new users of SGLT-2 inhibitors and GLP-1 RAs, with 1:1 propensity score matching. Over a median follow-up of 6.5 months, SGLT-2 inhibitor use was associated with a 40% increased risk of DKA hospitalization (HR=1.40), though the absolute risk remained low. As a reminder SGLT-2 inhibitor use has been associated with increased risk of DKA in some T2D patients.
- Baseline characteristics. At baseline, participants were on average 71 years old, and 49% were female. Nearly half had obesity (23%) or morbid obesity (22%), and 34% had documented diabetic neuropathy. Cardiovascular comorbidities were common: 58% had coronary artery disease, 22% had heart failure, and 94% had hypertension. Participants were taking an average of two diabetes medications at baseline, with 58% on metformin and 36% on insulin. Patients were 1:1 propensity score-matched to initiate either an SGLT-2 inhibitor or a GLP-1 RA, with 71,929 individuals in each treatment group.
- Results. Over a median follow-up of 6.5 months, the incidence of hospitalization for DKA was 4.4 events per 1,000 person-years among SGLT-2 inhibitor initiators, compared to 3.1 events per 1,000 person-years among GLP-1 receptor agonist initiators. This translated to a 40% increased relative risk with SGLT-2 inhibitors (HR=1.40) and an absolute risk difference of 1.23 events per 1,000 person-years. The elevated risk remained consistent across subgroups defined by age (<65 vs. ≥65 years), cardiovascular disease status, metformin use, and insulin use within 90 days of treatment initiation.
13. Emulation trial shows tirzepatide has superior cardioprotective benefits compared to dulaglutide
In this oral presentation, Dr. John Ostrominski (Harvard University) shared results of a trial emulation (n=31,550), comparing major adverse cardiovascular events (MACE) between tirzepatide and dulaglutide. This is an emulation of the ongoing phase 3 SURPASS-CVOT trial (n=13,299), a cardiovascular outcomes trial comparing tirzepatide to dulaglutide on cardiovascular outcomes in people with T2D and atherosclerotic cardiovascular disease (ASCVD). Of note, this trial has been expected to complete last month, although the clinicaltrials.gov website has not yet updated from “Active, not recruiting” status. Prof. Mathieu Chantal announced during ADA 2025 that the full results will be presented at EASD 2025, so we greatly look forward to the results!
The emulation trial was conducted using private insurance claims data between June 2022 to August 2024. Propensity-matched participants who initiated tirzepatide or dulaglutide (1:1) were evaluated for major adverse cardiovascular events (MACE; myocardial infarction, stroke, or mortality).
- Baseline characteristics. Participants were on average 68 years old, with 51% being female. Slightly over 60% of participants were White, followed by 14% Hispanic, 14% Black, and 2% Asian. Clinically, baseline A1c was on average 7.8% and eGFR was 74 mL/min/1.73 m2, which is in the range of early-stage kidney disease (ESKD; 60-90 mL/min/1.73 m2). A significant percentage of participants had comorbidities, including obesity (~50%), hypertension (89%), diabetic neuropathy (36%), heart failure (21%), stroke (15%), and myocardial infarction (10%). Most participants were on statin (83%), beta blockers (53%), and metformin (62%), while 34% were on insulin.
- Results. At a median follow-up of six months, tirzepatide was statistically significantly associated with a 37% reduction in MACE, compared to dulaglutide. When adjusted for A1c, tirzepatide was associated with a 23% reduction, compared to dulaglutide. These associations were consistent with and without SGLT-2 inhibitor use. While Dr. Ostrominski did not clarify, we are curious if the data included further glycemic metrics from continuous glucose monitoring (CGM) and how these mediate MACE reduction.
14. Novo Nordisk’s subcutaneous amycretin confers 24% weight loss at 36 weeks in people with obesity in phase 1a/2b study
In the poster hall, full phase 1b/2a results were presented for Novo Nordisk’s once-weekly subcutaneous GLP-1/amylin RA amycretin for obesity. Positive topline results were shared in January 2025. Amycretin is also under study as an oral formulation, for which positive results were presented at Novo Nordisk Capital Market Day 2024 and EASD 2024.
- Study design and baseline characteristics. Participants (n=125) were randomized to amycretin (1.25 mg, 5 mg, 20 mg, or 60 mg) or placebo. They ranged in age from 29 to 42 years old. Body weight ranged from 83.6 to 99.1 kg (184 to 218 pounds), and BMI ranged from 30 to 33.1 kg/m2. Mean A1c ranged from 4.9 to 5.5% - none of the 125 participants had diabetes or pre-diabetes (we would have been curious to see CGM metrics as we imagine despite the A1cs, people were not “in range” 100% of the time).
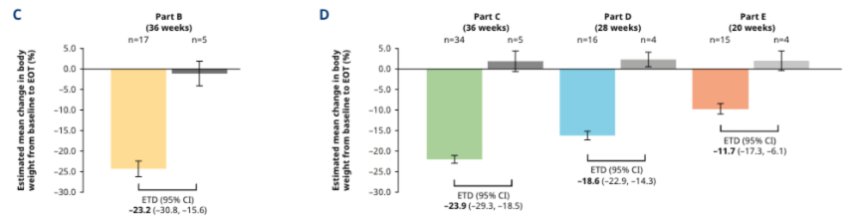
- Body weight loss results.
- At the highest amycretin dose (60 mg), participants saw 24.3% body weight loss versus 1.1% loss in people on placebo at 36 weeks (Part B in figure).
- Participants on 20 mg amycretin (Part C in figure) saw extremely significant weight loss as well at 36 weeks (-22% vs. +1.9% in placebo). That 90% of the weight loss could happen at just one-third of the top dose is very compelling.
- At the 5 mg dose (Part D in figure), weight loss was still high at -16.2% vs. +2.3% in placebo – this part of the study was shorter as well at 28 weeks.
- Finally, in the lowest dose group, 1.25 mg amycretin, weight loss was -9.7% vs. +2.0% in an even shorter period of 20 weeks (Part E in figure). There were no indications of a plateauing in weight loss at the end of the study for any of the doses.
- Safety. Most adverse events were “mild or moderate” in severity, dose-dependent, and GI-related (e.g., nausea, vomiting, diarrhea). Nearly all participants (94%) in the amycretin 5 mg, 20mg, and 60 mg groups experienced a GI-related adverse events, and the majority (63%) of those on amycretin 1.25 mg did as well. The poster noted that while this frequency of GI-related adverse events is high, it is in line with those reported in early-phase studies for GLP-1 RAs, GLP-1/GIP RAs, and amylin RAs. While we would agree it is in line with what has been seen, in “real life,” the combination of challenging (or little to no) titration combined with difficult side effect profile for many has caused major problems with “persistence.” Although there is no shortage of people who would like to try incretin therapy, the percentage who can stay on it successfully is low compared to many other classes. We hope with such successful results for amycretin therapy, there can be lessons learned on side effect profile that can be shared throughout the field.
- Other. With such terrific efficacy, we are also curious what combination therapy might be possible to use that would ameliorate some of the present difficulty of going on therapies with challenging (but perhaps more addressable) side effect profiles.
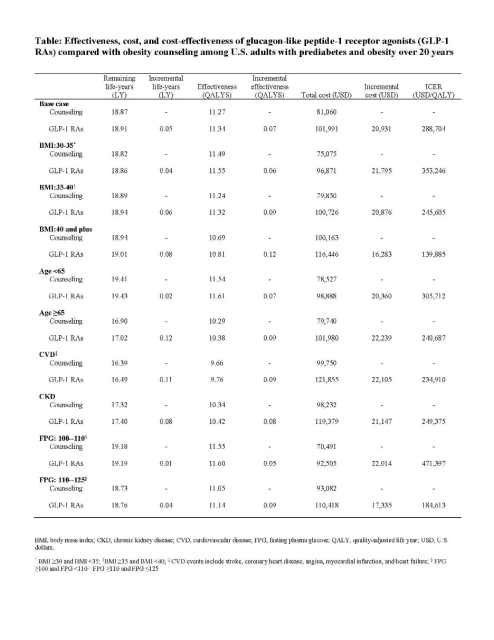
15. GLP-1 RAs are not yet cost-effective when compared to obesity counseling in T2D prevention
Dr. Hui Shao (Emory University) presented a simulation study on behalf of a CDC-led research team, addressing the long-term cost-effectiveness of GLP-1 RAs to prevent T2D in adults with obesity and prediabetes. While it’s well established that GLP-1 RAs are an effective therapy for reducing body weight and the incidence of T2D, this treatment class incurs an annual cost of >$10,000 for diabetes and >$13,000 for weight loss. In the US, where 75% of the population lives with overweight or obesity, evaluating the cost-effectiveness of preventative interventions is paramount to directing public health recommendations and insurance coverage.
- Study design and baseline characteristics. Study investigators used a CDC- and RTI-developed microsimulation model to estimate 20-year clinical outcomes for US adults with prediabetes and obesity. The cohort came from the 2017-2020 National Health and Nutrition Examination Survey (NHANES), with demographic characteristics matching national estimates. The model incorporated literature-based assumptions for the efficacy of GLP-1 RAs, dropout rates, and an annual drug cost of $13,618. GLP-1 RAs were compared with brief obesity counseling, and outcomes were evaluated via incremental cost-effectiveness ratio (ICER) in cost per quality-adjusted life year (QALY).
- Results. The ICER for use of GLP-1 RAs in the study population was $288,704/QALY, which far exceeds the standard US cost-effectiveness threshold of $100,000/QALY. Although the strategy was more favorable with subgroups with higher BMI (≥40 kg/m2), older age (≥65), or elevated fasting glucose (110-125 mg/dL), none of these scenarios lowered ICER to below the standard threshold. The use of GLP-1 RAs also had a slightly lower cumulative incidence of cardiovascular and microvascular disease, but these health benefits did not offset high drug costs. Dr. Shao emphasized that GLP-1 RAs are unlikely to be cost-effective in general prediabetes or obesity populations unless prices decline by more than 50%.
Diabetes Technology
16. New analysis of GDAC trial and real-world data shows improved alignment between updated GMI equation and A1c
In an oral symposium, Dr. Rich Bergenstal (International Diabetes Center) presented additional results from the GDAC study, which evaluated the agreement of an updated GMI metric (uGMI) with A1c. We previously saw results from the GDAC study at EASD 2024 analyzing an updated A1c metric – called “personalized A1c” (pA1c) – to better reflect average glucose levels than lab-based A1c. The study found that pA1c significantly reduced deviations from average glucose and better minimized discrepancies across racial groups compared to lab-based A1c.
Dr. Bergenstal said that this new GDAC analysis was motivated by the common discordance between GMI and A1c. Previous studies have identified a “glycation gap” in which individuals’ A1c may appear higher or lower than expected, and both positive (i.e., higher A1c than expected) and negative (i.e., lower A1c than expected) glycation gaps are associated with increased mortality. The existence of these glycation gaps has been attributed to variation in red blood cell characteristics across individuals and populations, including cells’ lifespan.
- This analysis compared the alignment between uGMI and current GMI with A1c. The updated GMI accounts for population-based red blood cell factors, unlike the original GMI. The study calculated GMI and uGMI with the following equations, where “AG” represents average glucose:
- GMI = 0.02392 * AG + 3.31
- uGMI = (15.36 * AG-1 + 0.0425)-1
- Methods. 257 participants in the GDAC study were included in this analysis with representation across several racial and ethnic groups. The study collected CGM data and bi-monthly A1c for 26 weeks. The analysis was also replicated on a larger real-world dataset.
- In GDAC, uGMI showed greater alignment with A1c. In the overall cohort, original GMI and A1c demonstrated a regression slope value up to 25% outside unity (i.e., one). This discrepancy was more pronounced among Black participants, showing a slope value 40% outside unity compared to 22% outside unity among non-Black participants. In contrast, regression slope values were significantly reduced with uGMI, indicating a better match. In the overall and non-Black cohorts, the regression slope was within 2% of unity with uGMI, while in the Black cohort, the regression slope was within 6% of unity. Notably, for the same average glucose, Black participants demonstrated higher A1c values than the non-Black cohort at all A1c ranges.
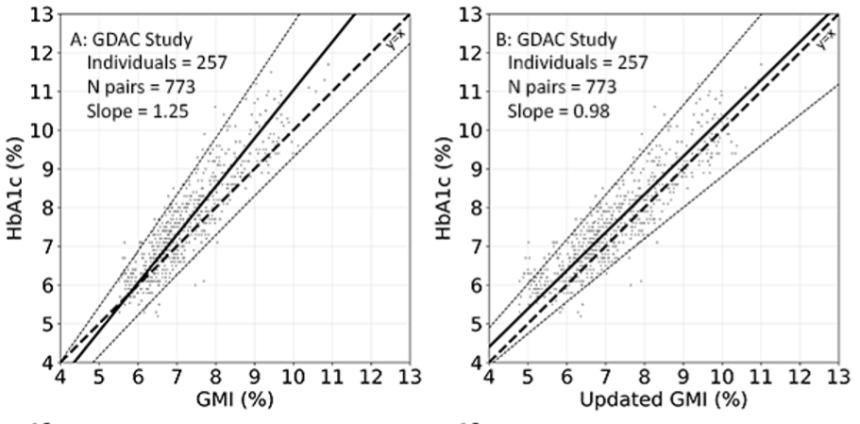
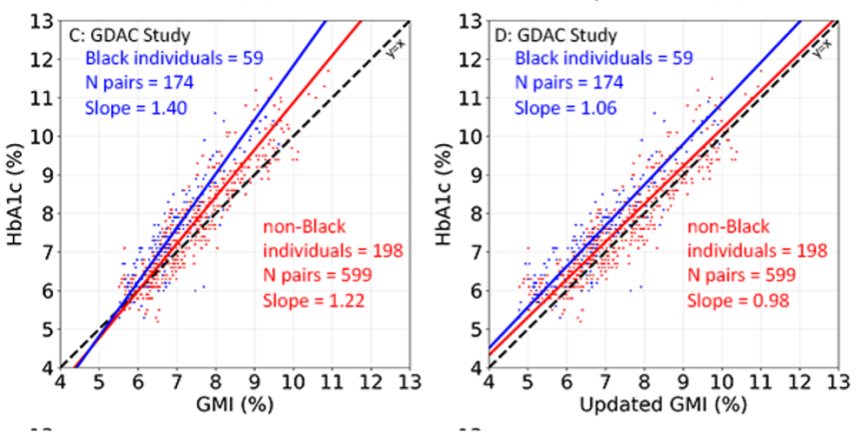
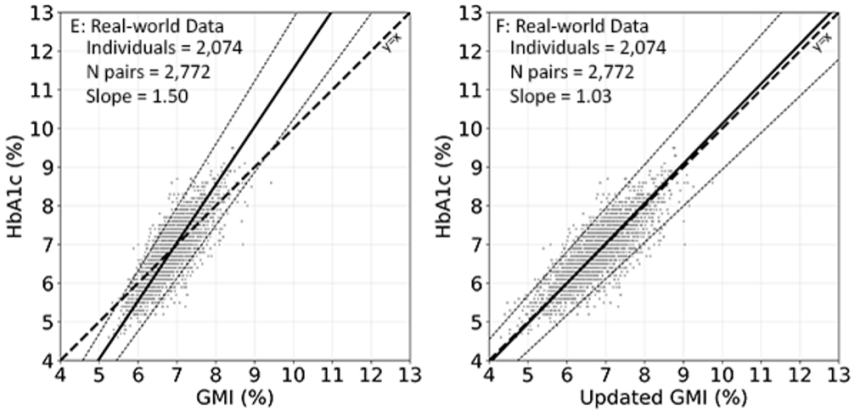
- GMI-A1c discordance was more pronounced in the real-world dataset, but uGMI still ameliorated these discrepancies well. With original GMI, the regression slope value was up to 50% outside unity; however, the slope was only 3% outside unity with uGMI.
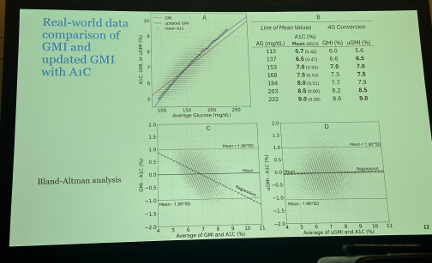
- Dr. Bergenstal said the results suggest uGMI better reflects A1c than original GMI, which could improve clinical decision-making. He compared the agreement of the current GMI (top graph below, red line) and uGMI (top graph, blue line) with the mean A1c (black circles) of all participants for each glucose range, and uGMI demonstrated much stronger agreement with the mean A1c levels. A Bland-Altman plot (bottom graphs below) also confirmed greater alignment between uGMI and A1c than GMI and A1c.
17. Personalized A1c improves correlation between GMI and A1c
Following Dr. Rich Bergenstal’s presentation on updated GMI, Prof. Ramzi Ajjan (University of Leeds, UK) presented a similar analysis examining the efficacy of “personalized A1c” (pA1c) to mitigate GMI-A1c discordance. Like Dr. Bergenstal’s data, this analysis also analyzed the same GDAC and real-world datasets. Prof. Ajjan previously presented results of the GDAC study at EASD 2024. Compared to lab-based A1c, pA1c reduced significant deviations from average glucose. Moreover, pA1c aligned with average glucose across all racial groups unlike lab A1c.
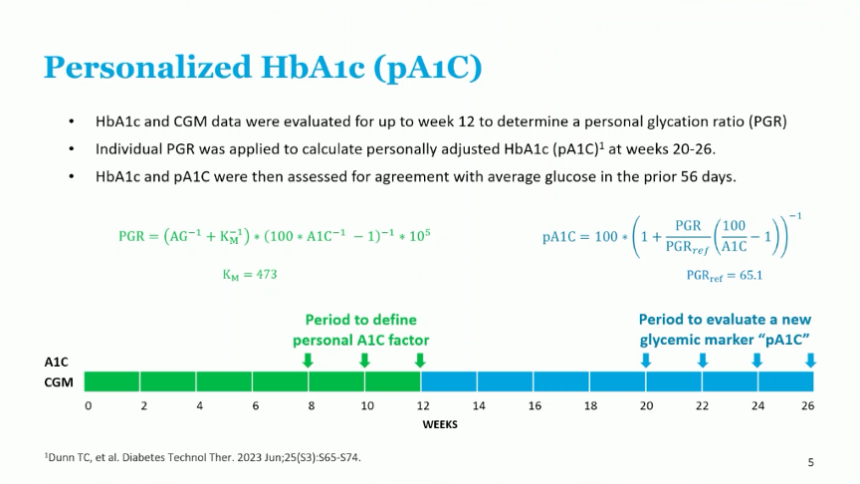
- Prof. Ajjan emphasized that the updated GMI better minimized deviations in the regression slope between GMI and A1c; however, it did not address the spread of the data. Thus, pA1c was applied to the datasets, aiming to tighten the correlation between GMI and A1c by accounting for individual variability in red blood cell physiology via the personalized glycation ratio (PGR), which is derived from A1c and CGM-derived average glucose.
- Correlation to GMI improved with use of pA1c. For GMI and lab A1c, R2 was 0.81, which increased to 0.93 with pA1c – indicating tighter correlation and less variance in the data. Additionally, a greater proportion of data was within 0.5 points with pA1c than lab A1c. Specifically, 60% of datapoints were within 0.5 points with lab A1c compared to three-quarters of datapoints with pA1c.
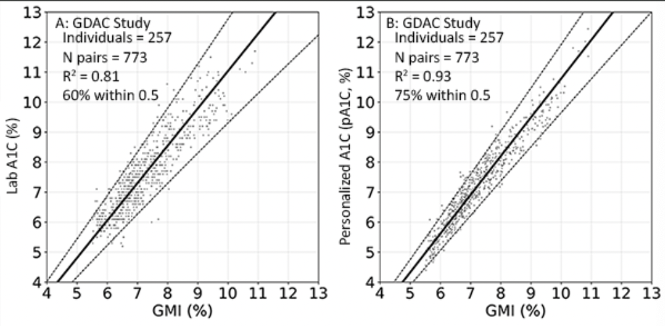
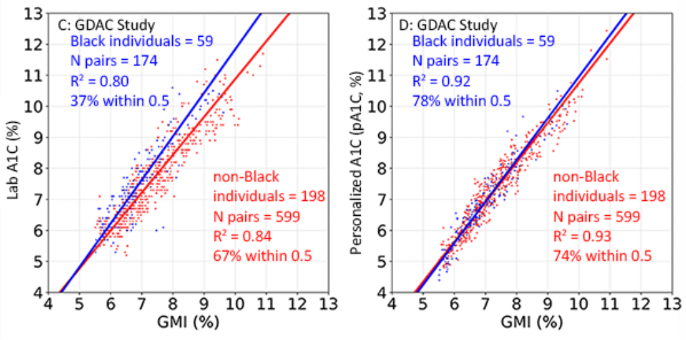
- Prof. Ajjan highlighted that pA1c led to larger improvements in agreement in certain subgroups, particularly among Black individuals. In the Black cohort, R2 increased from 0.80 with lab A1c to 0.92 with pA1c. Additionally, the percentage of datapoints within 0.5 points significantly increased from 37% with lab A1c to 78% with pA1c. The magnitude of increase for both endpoints was greater than the corresponding increases observed with pA1c in the non-Black cohort.
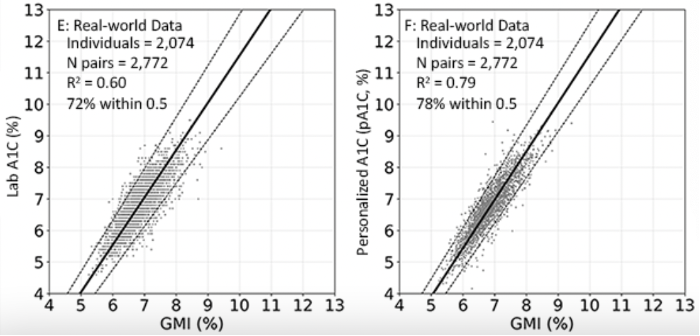
- pA1c also improved correlation in the real-world dataset. R2 improved from 0.60 with lab A1c to 0.79 with pA1c, and 78% of datapoints were within 0.5 points with pA1c compared to 72% with lab A1c.
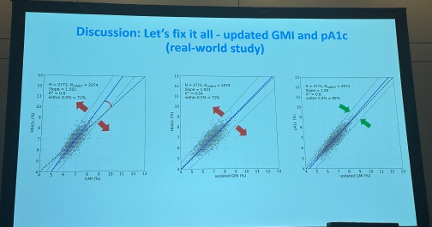
- pA1c and updated GMI showed significantly improved correlation than original GMI and lab A1c. In the real-world dataset, updated GMI with pA1c achieved a slope of 1.09 (within 9% of unity) compared to 1.5 between original GMI and lab A1c (within 50% of unity). R2 was also higher with updated GMI and pA1c compared to original GMI and lab A1c (0.8 vs. 0.6). Prof. Ajjan said these results indicate that for individuals with diabetes who do not require uninterrupted CGM, adjustment for red blood cell factors and use of pA1c should be considered.
18. Largest real-world dataset of global MiniMed 780G users (n=374,548) shows mean TIR of 72%; TIR increases with recommended settings
During Medtronic’s product theater, Dr. Jennifer McVean (Medtronic) presented real-world outcomes from nearly 375,000 global MiniMed 780G users. To our knowledge, this is the largest real-world dataset that Medtronic has presented for MiniMed 780G. Medtronic presented outcomes among 322,043 global users at ATTD 2025, up significantly from a dataset with 101,629 global users at EASD 2023. In both datasets, mean Time in Range (TIR) was 72%, and Time below Range (TBR) was 2.0% or below.
- Medtronic’s dataset included users who voluntarily uploaded their data to CareLink between January 2020 and April 2025. Eligible participants had ≥10 days of CGM data.
- In the overall cohort, mean TIR was 72% – consistent with previous MiniMed 780G datasets. Time in Tight Range (TITR) was 47%. TBR was comparable to previous datasets as well at 1.7% with 0.3% Time <54 mg/dL. Use of the recommended settings (i.e., two hours active insulin time and 100 mg/dL) was associated with increased TIR, primarily driven by reductions in Time above Range (TAR). Users who selected recommended settings (n=59,474) achieved 78% TIR and 53% TITR, which were each 1.3 hours/day higher, respectively, than the overall cohort. TAR was 1.2 hours/day lower (26% to 21%), and TBR was slightly lower at 1.5%.
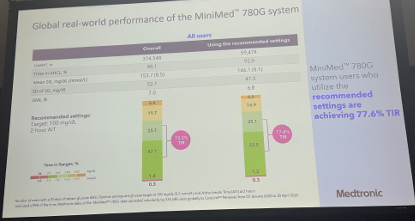
- Dr. McVean emphasized that these results support AID as the standard of care for people with T1D, stating that it should be offered to all individuals with T1D. She also reiterated that AID should be initiated early, even as early as at T1D diagnosis, citing T1D Exchange data presented at ATTD 2025 that associated earlier initiation of AID following T1D diagnosis with lower A1c than delayed, or no, initiation, at two years post-diagnosis.
19. All eyes on T2D and ketone testing: Abbott product theater reviews unmet needs in analyte sensing
Abbott’s morning product theater highlighted the gaps in CGM care in T2D and ketone monitoring. Dr. Diana Isaacs (Cleveland Clinic), Dr. Carol Wysham (Multicare), and Dr. Marc Taub (Abbott) discussed the future of technology for patients with diabetes in this morning panel.
- Dr. Taub examined the potential of Abbott’s dual CGM-CKM in development to improve diabetes care. While no new details were shared on the product or launch timeline, Dr. Taub reviewed its feasibility studies showcasing variable responses in glucose and ketones after cessation of insulin delivery. He argued that continuous ketone monitoring can offer early warning of potential ketosis after insulin delivery failure and reduce the number of items patients must carry in their “diabetes kits.”
- Abbott’s dual CGM-CKM sensor will be based on FreeStyle Libre 3 Plus. It will be a 15-day sensor that sends glucose and ketone data to a smartphone or receiver every minute, with optional alarms for elevated ketone levels. Dr. Taub said that this is especially important given the concerning data around ketone testing rates in people with T1D. With 64% of patients reportedly never testing for ketones, less than 20% testing ketones when experiencing glucose values >300 mg/dL for over an hour, and only 62% of patients testing when nauseated or vomiting, this dual sensor could fill a significant unmet need.
- Abbott has announced several future pump integrations for its dual glucose-ketone sensor in recent weeks. Four companies’ AID systems will be integrated with the sensor upon launch: (i) Sequel’s twiist; (ii) Tandem’s t:slim X2 and Mobi with Control-IQ+; (iii) Beta Bionics’ iLet; and (iv) Ypsomed’s mylife Loop.
- Dr. Isaacs reviewed data demonstrating the clinical benefits of FreeStyle Libre CGM in people with T2D. The US T2D population remains significantly underpenetrated, with just 10% of the ~30 million people using a CGM today. ADA’s Standards of Care continue to recommend CGM for all people with diabetes on insulin therapy, and more recently have recommended considering the technology for those on non-insulin therapy. Dr. Isaacs cited the FRONTIER study as an example of the benefits for this population, as it showed that CGM had an additive reduction in A1c alongside GLP-1 RAs, with an additional 0.6 percentage point reduction compared to GLP-1 RA use alone.
- Dr. Isaacs also reviewed a late-breaking abstract at ADA on the qualitative benefits to self-management engagement with FreeStyle Libre 3 Plus. As a follow-up to the RCT presented at ATTD earlier this year reporting significant increases in self-management engagement (a 0.5 point increase to 4.4 on the Likert 5-point scale; p<0.001) and a 0.9 percentage point decrease in A1c, semi-structured interviews with adult users with T2D found that most reported an improved diet with use of FreeStyle Libre 3 Plus (84%), and approximately two-thirds said effective decision making was made possible with its data (n=31).
- Dr. Wysham highlighted Abbott’s future software innovation with three key upcoming offers:
- Insulin pump integrations with FreeStyle Libre 3 Plus. In addition to its existing integration with Beta Bionics’ iLet and the upcoming integration with Sequel’s twiist, Dr. Wysham highlighted its integration with Tandem’s insulin pumps. Integration with t:slim X2 launched in the US yesterday, while Mobi integration is expected later this year.
- Apple Watch App. Dr. Wysham also highlighted Abbott’s work on the Libre Watch App, in which FreeStyle Libre users will be able to view their glucose readings and trends on their Apple Watch – similar to Dexcom’s offering for G7 users beginning last June.
- Epic EHR integration. First announced in April, Abbott will soon be integrating Libre data with Epic’s EHR system, the leading EHR platform in the US. This often-requested feature will enter CGM-derived metrics as discrete items in a patient’s chart, enabling searchability and streamlined workflows.
20. Omnipod 5 users with T2D report less diabetes distress compared to MDI
Dr. Kristin Castorino (Sansum Diabetes Research Institute) presented a sub-analysis of Insulet’s SECURE-T2D study evaluating the impact of Omnipod 5 initiation on diabetes distress in adults with type 2 diabetes (T2D). Initial results from this trial were presented at ADA last year, demonstrating mean A1c reduction of 0.8% to 7.4% and Time in Range (TIR) improvement of 20% (+4.8 hours/day) to 66% at 13 weeks. No additional severe hypoglycemic events were reported. The positive results contributed to the FDA’s clearance of the system for individuals with T2D on insulin therapy last August.
- Study design. As a reminder, SECURE-T2D was a single-arm, multicenter trial that enrolled adults with insulin-treated T2D (n=305) from 21 institutions across the US. Participants remained on stable anti-hyperglycemic medications and initiated 13 weeks of Omnipod 5 use after a 14-day standard therapy phase. Diabetes distress was assessed at baseline and study end using the Type 2 Diabetes Distress Assessment System (T2-DDAS).
- Baseline characteristics. Mean age was 57 years; 24% of participants were Black and 22% Hispanic/Latino. Most (72%) had public insurance; 21% were on basal-only insulin, and 55% were using GLP-1 RAs. At baseline, mean A1C was 8.2% and 66% of participants reported moderate or high diabetes distress.
- Results. After 13 weeks, the proportion of participants reporting “moderate” or “high” distress (T2-DDAS score ≥2.0) dropped significantly from 66% to 55% (p<0.001). Improvements were observed across all baseline TIR groups, with the largest reductions in participants with baseline TIR of 30%–60% (-13 percentage points) and ≥60% (-12 percentage points). Mean T2-DDAS score declined by 0.3 points. Dr. Castorino concluded that Omnipod 5 use significantly reduced diabetes distress across a diverse T2D cohort of adults regardless of baseline glycemia and prior therapy, reinforcing the system’s potential to alleviate the emotional burden associated with T2D self-management.
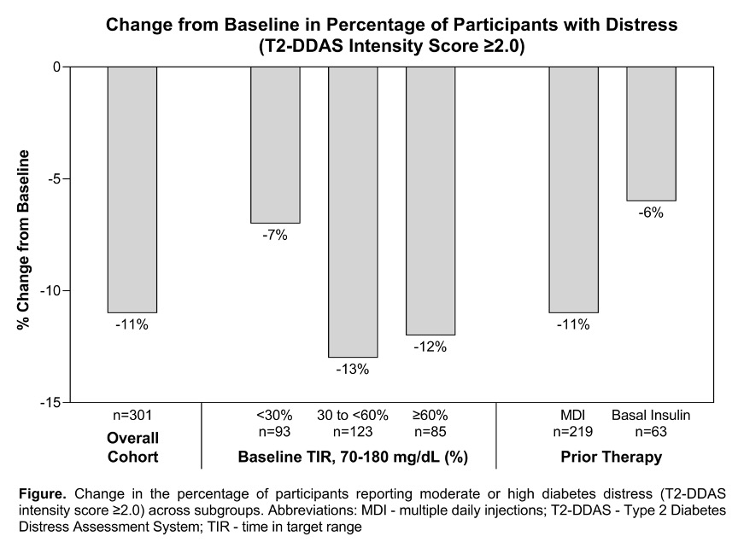
21. Dr. Rich Bergenstal, Dr. Anita Swamy, and Dexcom Warrior Mr. Lance Bass review the present and future of Dexcom CGM
Dexcom’s product theater reviewed the field’s viewpoints on CGM and Dexcom’s current and upcoming offerings. Dr. Rich Bergenstal (International Diabetes Center), Dr. Anita Swamy (Lurie Children’s Hospital), and Dexcom Warrior and singer of *NSYNC acclaim Mr. Lance Bass led this dynamic afternoon panel in front of a packed room in the exhibit hall.
- Dr. Rich Bergenstal reviewed broader perspectives on the importance of CGM. Continuing a common theme from today, he noted that CGM and GLP-1 RAs are “better together” for improving glycemic outcomes and said earlier interventions and personalization of therapy remains critical. Speaking to the emergence of over-the-counter CGMs, he also teased a later presentation on real-world data from the first 30 days of thousands of Stelo users (2018-LB), which will partly improve our understanding of glycemic targets for people with normoglycemia. Dr. Bergenstal also reviewed Dexcom’s new US-focused “State of Type 2” report (n=111 HCPs, n=310 people with T2D), which found that: (i) 96% of HCPs agreed that CGM should be the standard of care for individuals using MDI; (ii) 94% of HCPs agreed that CGM should be standard of care for those on basal-only insulin; and (iii) 59% of HCPs ranked CGM as the single most impactful tool for improving T2D management in the next decade, including current and future medications.
- Earlier this year, Dexcom also unveiled its multi-national “State of Type 2” report (n~2,500), which similarly found that: (i) people with T2D largely reported positive experiences with CGM (93%); (ii) nearly all healthcare providers (96%) said that CGM should be standard of care for people with T2D on MDI; (iii) half of healthcare providers said that CGM should be standard of care for T2D regardless of insulin therapy; and (iv) more healthcare providers said CGM and education access will positively impact T2D management in the next 10 years than improved medications.
- Dr. Swamy reviewed Dexcom’s current and upcoming product offerings:
- Dexcom G7 15 Day. This sensor received FDA clearance in May for people 18 years and older with T1D, T2D, or gestational diabetes, the G7 15 Day increases the sensor’s wear time by 50% compared to Dexcom G7, totaling 15.5 days including the 12-hour grace period. The sensor is expected to launch in 2H25.
- Software:
- Dexcom G7 app. In May, Dexcom unveiled an app feature allowing users to upload photos, including meals, to event logs. The feature includes an AI-generated algorithm to estimate carbohydrates. Dr. Swamy noted that for now, this is not designed to replace users’ own carbohydrate counting, “but that may change.” It also facilitates the visualization of glycemic data immediately before and after events (like exercise) and meals to clarify short-term glycemic impacts. The app also added a seven-day GMI, previously only available for 14 days of data. The company has previously shared that additional Clarity data will be made available for the app “soon,” for which Dr. Swamy did not share any details.
- Follow app. Similar to Clarity data integration, Dr. Swamy did not provide details on upcoming updates to Dexcom’s Follow app. In our interview with Dexcom CEO Mr. Kevin Sayer in March, he remarked that these upgrades will be a “massive revamp.”
- Stelo. Dr. Swamy touted the continued popularity of Stelo, Dexcom’s over-the-counter CGM designed for adults not on insulin therapy that was launched last August. She highlighted its integration with Oura Ring in May, enabling users to see how its metrics – including vital signs, sleep data, signs of stress, heart health, and activity data – correlate with glucose. She identified this integration as a “critical” feature for people with and without diabetes.
- Mr. Lance Bass, former member of *NSYNC and one of Dexcom’s 25,000+ Dexcom Warriors, then took to the stage to share his personal experience with latent autoimmune diabetes in adults (LADA). He recounted how he was initially misdiagnosed with T2D and struggled to manage his glycemia with medication until his care team ultimately corrected the diagnosis. Speaking on Dexcom’s product portfolio, he expressed appreciation for CGM compared to BGM, highlighted the value of Dexcom’s food logging feature, and shared his excitement about the prospect of soon needing to change sensors only twice a month. He said technologies like Dexcom CGM enable him to more easily focus on family, including traveling and activities with his kids.
22. UNITE RCT of CGM in non-insulin T2D demonstrates significant TIR improvements regardless of onboarding approach
In an oral symposium, Dr. Holly Willis (International Diabetes Center) presented results from the Using Nutrition to Improve Time in rangE (UNITE) RCT (NCT05928572) evaluating the impact of CGM initiation in adults with T2D on non-insulin therapy. This trial reflects heightened interest in the efficacy of CGM for people with T2D on non-insulin therapy after the launch of the first CGMs with over-the-counter availability in the US last year – Abbott’s Lingo and Dexcom’s Stelo. Due to increasing evidence supporting CGM’s efficacy in this population, the ADA’s 2025 Standards of Care updated its recommendations to state that healthcare providers should “consider” CGM in adults with T2D on non-insulin therapy. As a result, CGM coverage for people with T2D on non-insulin therapy is expanding in the US – as of this summer, the three largest PBMs will cover Dexcom CGM for this population. Evidence will continue to accumulate since Dexcom has also initiated an RCT in this population: an initial readout is expected by early 2026.
- Methods. This two-month pilot study assessed glycemic outcomes following Dexcom G7 initiation through two approaches: (i) nutrition-focused approach (NFA) or (ii) self-directed approach (SDA). In the NFA arm, participants received CGM education that emphasized using CGM data to not only improve Time in Range (TIR), but also to make healthy food choices that align with evidence-based recommendations. In the SDA arm, participants received CGM education primarily through manufacturer-provided resources, which instead encouraged participants to use CGM in the way that felt most useful to them. Participants wore blinded CGM for 10 days to collect baseline data before initiating Dexcom G7. 124 participants completed the study. In the overall cohort, baseline A1c was 7.9%.
- Significant glycemic improvements were observed regardless of education approach. For the NFA arm, TIR increased 6.0 hours/day from 46% at baseline to 71% at two months (p<0.0001), and in the SDA arm, TIR increased 3.8 hours/day from 55% at baseline to 71% at follow-up (p=0.0007). Both arms also achieved significant improvements in Time in Tight Range (TITR). For the NFA arm, TITR increased 4.3 hours/day from 16% at baseline to 34% at two months (p<0.001), and in the SDA arm, TITR increased 2.9 hours/day from 23% at baseline to 35% at follow-up (p=0.003). These improvements were primarily driven by significant reductions in Time above Range (see table below), and Time below Range remained minimal in both arms.
- The percentage of users achieving over 70% TIR also significantly increased in both arms. Two-thirds of participants in the NFA arm achieved >70% TIR at follow-up compared to 20% at baseline – translating to nearly eight-fold increased odds of achieving >70% with TIR vs. baseline (OR=7.6, CI: 3.3-17.4; p<0.001). Moreover, 61% of participants in the SDA arm achieved >70% TIR at two months compared to 34% at baseline – this represents three-fold increased odds of achieving this target with CGM vs. baseline (OR=3.1, CI: 1.4-6.8; p=0.005). The results indicate that CGM can significantly improve glycemic health in people with T2D on non-insulin therapy regardless of onboarding strategy.
Nutrition-Focused Approach (n=64) | Self-Directed Approach (n=60) | |||||
Glycemic Outcomes | Baseline | Follow-up | p-value | Baseline | Follow-up | p-value |
Time in Range | 46% | 71% | <0.0001 | 55% | 71% | 0.0007 |
Time in Tight Range | 16% | 34% | <0.001 | 23% | 35% | 0.003 |
Time above Range | 54% | 28% | <0.001 | 45% | 29% | <0.001 |
Time below Range | 0.1% | 0.2% | 0.10 | 0.3% | 0.1% | 0.01 |
Mean sensor glucose | 197 mg/dL | 162 mg/dL | <0.0001 | 182 mg/dL | 163 mg/dL | 0.002 |
A1c | 7.9% | 7.3% | <0.001 | 7.8% | 7.3% | <0.001 |
- Participants in the NFA arm achieved dietary improvements, measured by the Healthy Eating Index (HEI). Total HEI score increased five points from 60 to 65 due to increased consumption of healthier foods, including fruits and vegetables, and decreased consumption of refined grain and sugar. In contrast, total HEI score did not change in the SDA arm.

- Both arms also saw benefits in additional patient-reported outcomes. Regarding diabetes distress, both groups experienced statistically significant within-group reductions in regimen-related diabetes distress scores in the Diabetes Distress Scale; there was no between-arm difference. NFA participants also reported increased confidence in their ability to use CGM data to choose foods that both improve TIR and overall health. In contrast, there was no statistically significant increase in confidence in the SDA arm.
23. Preliminary real-world outcomes among early twiist users (n=36) finds mean TIR of 80% with 2.9% TBR
Sequel’s Product Theater unveiled preliminary outcomes among early twiist users. Dr. Joanna Mitri (Sequel Med Tech) presented outcomes from two analyses: (i) an investigator-initiated study of adults with T1D (n=19); and (ii) real-world outcomes for early adopters in the twiist Early Experience Program (n=36). Dr. Mitri announced twiist is expected to launch in the US starting July 7 with FreeStyle Libre 3 Plus compatibility. Eversense 365 integration is expected in 3Q25, and Sequel also plans to offer integration with Abbott’s upcoming dual glucose-ketone sensor. We previously saw data on twiist’s occlusion detection capabilities at ENDO 2024. These analyses are some of the first glycemic outcomes among twiist users that Sequel has presented.
- Investigator-initiated study: This single-arm study initially included 20 adults with T1D, although one participant later opted out. The trial lasted for eight weeks, evaluating CGM metrics at the end of the study period, including Time in Range (TIR), Time above Range (TAR), and Time below Range (TBR). At eight weeks, mean TIR was 70%. Mean TAR was 27% with 8% Time >250 mg/dL. Mean TBR was 2.3% with 0.2% Time <54 mg/dL.
- Real-world outcomes: All participants in the real-world dataset were adults with T1D. The mean time for device use was 35 days, and presented results were acquired through June 6, 2025. Mean TIR was higher in the real-world cohort than the investigator-initiated study, reaching 80%. TAR was subsequently lower in the real-world dataset at 17% with 3% Time >250 mg/dL. TBR was slightly higher in this dataset: 2.9% with 0.2% Time <54 mg/dL. Although this is a small, preliminary dataset, we would be interested to see mean Time in Tight Range for users, especially since TBR appears slightly higher than other real-world datasets of other AID systems (such as Omnipod 5, iLet, or MiniMed 780G). We’re interested to see how twiist’s real-world outcomes evolve once adopted by a much larger userbase.
- Dr. Mitri said that Sequel will share best practices for system settings in the future. We are interested in which settings these users employed. twiist offers a much broader correction range than its competitors in the US (87-180 mg/dL), so we imagine a more aggressive target setting (<100 mg/dL) would predispose people to greater TBR.
24. Personalized diabetes treatment using AI/machine learning for AID and MDI
Dr. Peter Jacobs (Oregon Health & Science University) spoke to a full room Saturday afternoon on the potential of AI and machine learning (ML) to improve AID. Dr. Jacobs said that the primary motivation for personalization of diabetes treatment is that many with T1D are still not achieving glycemic targets regardless of insulin delivery method. In a 2024 survey of 926 people with T1D, half of patients using CGM and MDI or CGM and an insulin pump as well as 28% of people on AID failed to meet consensus Time in Range (TIR) goals. Dr. Jacobs noted that physiological differences significantly impact postprandial glucose response in T1D, which may contribute to challenges in goal achievement. In a study controlling for meal size (n=61), significant postprandial glucose level differences were observed between individuals. When exercising, controlling for meal size and exercise intensity of 70% VO2 max, nearly all participants (n=20) experienced a drop in glucose levels, with substantial differences between participants and between repeated measurements of the same participant.
Addressing these physiological differences presents a significant clinical challenge, which AI can help address. AI can adapt to an individual, quickly responding to forecasted events, such as meals, exercise, or predicted hypoglycemia.
- Reinforcement learning, a type of ML, can be used to personalize meal bolus calculation. In a 2024 study, Dr. Sayyar Ahmed (University of Girona, Spain) et al. demonstrated a Markov Decision Process-based algorithm that incorporates a state space (current glucose state), action space (amount of glucose to administer), and transition probability (likelihood of system moving from current state to improved glycemia). The system optimizes recommended bolus policy based on a reward (the return of blood glucose to normal levels). In silico, the algorithm demonstrated strong TIR results – 71% for full automation and 76% for hybrid AID. Time below Range (TBR) was 1% for full automation and 0.1% for hybrid AID.
- AI-enabled meal detection and insulin dosing reduces Time above Range (TAR) in AID. Dr. Clara Mosquera-Lopez (Oregon Health & Science University) et al. developed an algorithm that uses: (i) two-hour CGM history; (ii) insulin on board 60 minutes before prediction time; and (iii) time of day to detect meals without user input. The system calculates the probability of a meal having been consumed in the past 60 minutes, and estimates carbohydrate amounts in 20-gram ranges. Using this feature, a randomized in-clinic study (n=15) found that the meal detection algorithm had 83% sensitivity and a false discovery rate of 17%. The meal detection algorithm reduced post-prandial TAR by 11%. In a one-week outpatient study, the algorithm demonstrated equivalent performance to AID with 56% TIR compared to 57% with usual care, but with a 38% reduction in required meal bolusing. The streamlining of support offered by the meal detection algorithm reduces the burden of diabetes management for PWD.
- AI can be used with MDI to improve patient outcomes and streamline care. An algorithm, such as DailyDose, may be implemented to provide weekly recommendations of carb ratios, correction factors, and long-acting insulin. Use of the algorithm improved TIR by 6% in eight weeks when used by 24 participants. An automated forecast tool may also predict risk of hypoglycemia before bedtime and prompt the user to eat before going to sleep. The algorithm will recommend different snacks based on a risk of hypoglycemia within four hours of bedtime versus four to eight hours after bedtime. The proportion of nights where patients experienced very low glucose levels less than 54 mg/dL was reduced by 30% with use of the algorithm and MDI. “The future is bright for use of AI in personalized diabetes care,” said Dr. Jacobs. With AI, greater personalization of medicine may be just a few clicks away.
25. Glycemic outcomes among MiniMed 780G users (n=40,975) do not differ by socioeconomic status
In the poster hall (938-P), Dr. Jennifer McVean (Medtronic) presented another real-world analysis of MiniMed 780G users, examining the relationship between glycemic outcomes and socioeconomic status. This analysis classified socioeconomic status according to the area deprivation index (ADI), which evaluates health disparities based on census indicators, including education, employment, housing, and income. Importantly, research has associated higher ADI with increased dysglycemia. Therefore, this analysis intended to elucidate the relationship between ADI and glycemic outcomes among MiniMed 780G users.
- Methods. The study included data from 40,975 US users who uploaded their data to CareLink between May 2, 2023, and March 31, 2025. Eligible participants had at least 10 days of available CGM data. Participants were grouped into four quartiles: (i) 1-≤25; (ii) 26-≤50; (iii) 51-≤75; and (iv) 76-≤100. A higher ADI indicates greater socioeconomic deprivation. The lowest and highest deciles were also directly compared.
- Glycemic outcomes were similar regardless of ADI quartile. All quartiles averaged 73% Time in Range (TIR) and 47% Time in Tight Range. Time below Range (TBR) was identical for all three quartiles at 1.3% with 0.2% Time <54 mg/dL. Each quartile also achieved a mean GMI of 7.0%, and mean sensor glucose was 153-154 mg/dL across quartiles. Roughly half ofparticipants in each quartile achieved the triple composite endpoint of: (i) GMI <7.0%; (ii) TIR >70%; and (iii) TBR <4.0%.
- A greater proportion of users with the recommended system settings achieved the triple composite endpoint: 68-70% of participants in each quartile. These users also achieved 5% higher TIR at ~78% (+1.2 hours/day) and 5% higher TITR at ~52% across all quartiles.
- Glycemic outcomes were comparable between the lowest and highest ADI deciles as well. The least deprived decile achieved 74% TIR compared to 73% TIR in the most deprived decile, while TITR was 47% in both cohorts.
26. Retrospective study finds association of CGM with improved renal outcomes in adults with CKD and insulin-treated diabetes
Dr. Katia Hannah (Dexcom) presented a retrospective analysis of adults with insulin-treated diabetes and CKD (n=65,839), most of whom have T2D, evaluating the effects of CGM initiation on CKD progression. This study was motivated by previous research sponsored by Dexcom, which associated CGM initiation with reduced diabetes-related hospitalizations among people with T2D and CKD. Notably, although all the cohort qualify for CGM use, only 1% were using Dexcom’s CGM, and presumably less than 10% were using any kind of CGM.
- Methods. The study used de-identified Truveta EMR data to compare glycemic and renal outcomes between individuals who initiated Dexcom CGM (between August 2017 and December 2022) and non-users. The index date for individuals not using CGM was defined as their first outpatient encounter with a qualifying eGFR. CKD progression was defined as an increase in CKD stage. Changes in eGFR and A1c were evaluated as the difference between baseline lab values (which occurred within 12 months of the index date) and three-year follow-up.
- Demographics. The cohort consisted of 65,206 individuals who did not use CGM (99% of the cohort!) and 633 individuals using a Dexcom CGM. The vast majority (89%) of non-users had T2D. In comparison to the cohort using Dexcom, over half (56%) of this group had T1D. While CGM users were predominantly white (81%), among non-users, only about two-thirds (65%) were white, and 23% Asian, and 18% Black. Baseline A1c was similar in both cohorts at 8.0% among CGM users and 8.1% among non-users. The mean baseline eGFR was 71 mL/min/1.73 m2 among CGM users, while non-users had a mean eGFR of 66 mL/min/1.73 m2, a statistically significant difference. Most participants were classified as having stage 2 CKD, including 81% of CGM users and 69% of non-users (we assume most non-users had more advanced CKD).
- At three years, fewer CGM users experienced CKD progression. Specifically, 29% of CGM users saw CKD progress by at least one stage compared to 35% of non-users (p<0.0001) – that 17% fewer people with diabetes on CGM would see progression in chronic kidney disease certainly strikes us as a very decisive result. Notably, CGM initiation was also associated with a 14% reduction in progression rate according to eGFR rate of change (p<0.05). Specifically, non-users had a crude three-year decline in eGFR of 8.6 mL/min/1.73 m2 compared to 7.4 mL/min/1.73 m2 among CGM users – that CGM users would experience a 14% lower decline is certainly important evidence in favor of those on Dexcom’s CGM.
- We were also happy to hear data on glycemic outcomes. As expected, CGM users also had improved glycemic outcomes. In a subset of individuals with A1c available at baseline and three years (463 CGM users and 37,218 non-users, reflecting 73% and 57% of the CGM and non-CGM groups), CGM initiation was associated with a 0.25% greater reduction in A1c (p<0.05).
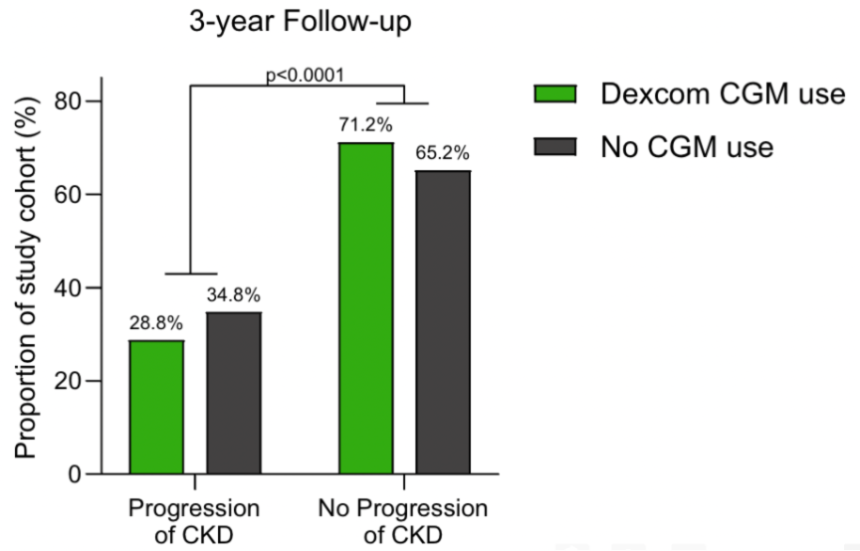
27. Dexcom G6 Pro shows high bias and reduced accuracy in ICU setting
Dr. Jagdeesh Ullal (University of Pittsburgh) presented data evaluating the accuracy and reproducibility of the Dexcom G6 Pro CGM in critically ill patients with diabetes or stress-induced hyperglycemia in the ICU.
- Study design. Participants included 95 people with diabetes and 35 with chronic hyperglycemia, for reasons other than diabetes. The study, which was conducted across five US academic centers, compared 9,121 CGM glucose readings from the Dexcom G6 Pro with venous or capillary blood glucose (BG) values in ICU patients. A total of 124 participants wore two CGM sensors simultaneously to assess reproducibility.
- Results. The median relative absolute difference (RAD) between CGM and BG was 19%, with only 41% of CGM readings falling within the 15%/15 agreement threshold, which was lower than expected compared to outpatient performance. CGM readings were consistently higher than reference values, with a median bias of 25 mg/dL. Accuracy was not significantly affected by insulin type, hypoglycemia status, or ICU setting, though slightly better accuracy was observed in cardiac ICUs.
- Conclusion. The Dexcom G6 Pro sensor exhibited lower accuracy in ICU patients compared to outpatient performance benchmarks, where accuracy was generally better at higher blood glucose levels.It remains unclear whether this is due to the sensor itself or the unique physiological challenges present in critically ill populations. Dr. Ullal noted that newer sensors, such as Dexcom G7, have shown improved accuracy in similar ICU studies, as observed in a recent publication.
28. MiniMed 780G users of seven-day infusion set report reduced burden with fewer set changes, occlusion rates, and alerts, as well as slightly improved glycemic outcomes compared to three-day set
Dr. Tim Kwa (Medtronic) presented a poster (954-P) comparing glycemic outcomes between Medtronic’s seven-day extended infusion set (EIS) and three-day standard infusion set (SIS). The analysis included two components. First, CareLink data was analyzed from 49 MiniMed 780G users who switched from SIS to EIS, who maintained >80% use of each set during the three months before and after the switch. Additionally, CareLink data collected between January and April 2024 was compared between new MiniMed 780G who used EIS (n=948) and SIS (n=642) – users were categorized by infusion set based on which set constituted >80% of orders.
- Among those who switched from the SIS to the EIS, Time in Range (TIR) was slightly higher with EIS, although the difference was not statistically significant (p=0.31). TIR increased 26 minutes/day from 73% with SIS to 75% with EIS. Time below Range was slightly lower with the EIS vs. the SIS (1.2% vs. 1.6%).
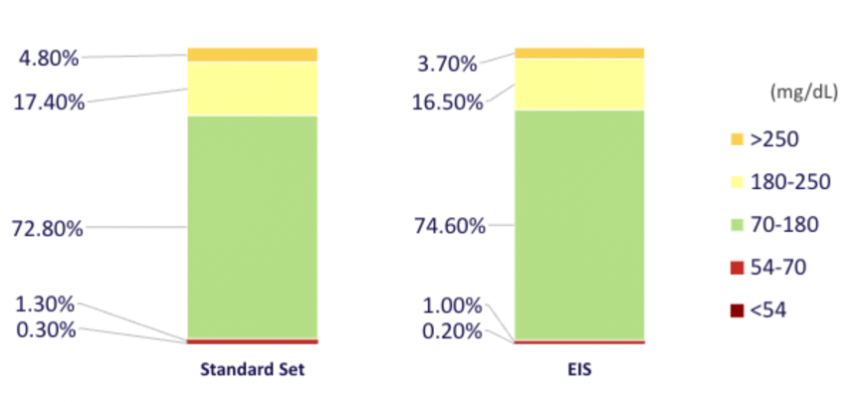
- In the second analysis of a broader cohort of SIS and EIS users, set performance was compared.
- TIR: For both the SIS and EIS, median TIR was lowest on the first day after a set change for both the SIS and EIS (~70%). On the second day of wear, TIR improved to about 76% with both sets. For the EIS, TIR remained roughly stable throughout the remainder of the wear period, decreasing slightly to 75% at Day 7. After Day 1, SIS users also saw TIR decreases to nearly 70% on Days 4 and 7 – corresponding with set changes. The average monthly duration of TIR <70% with the EIS was lower than with the SIS (73 hours vs. 127 hours; p<0.001).
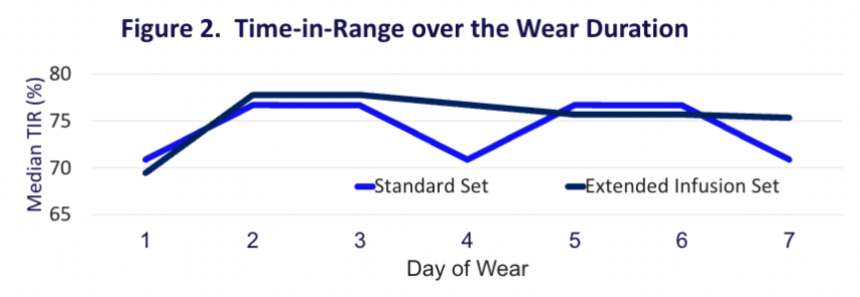
- Occlusion alert rate: Like TIR, occlusion alert rate was highest on the first day of wear for both sets. However, alert rates decreased after the first day, remaining somewhat stable for the rest of wear. Overall occlusion rates were 21% lower with the EIS compared to the SIS (p=0.001).
- Total daily insulin dose: Total daily insulin dose tended to be highest on the first day of SIS use, decreasing significantly each following day of wear.
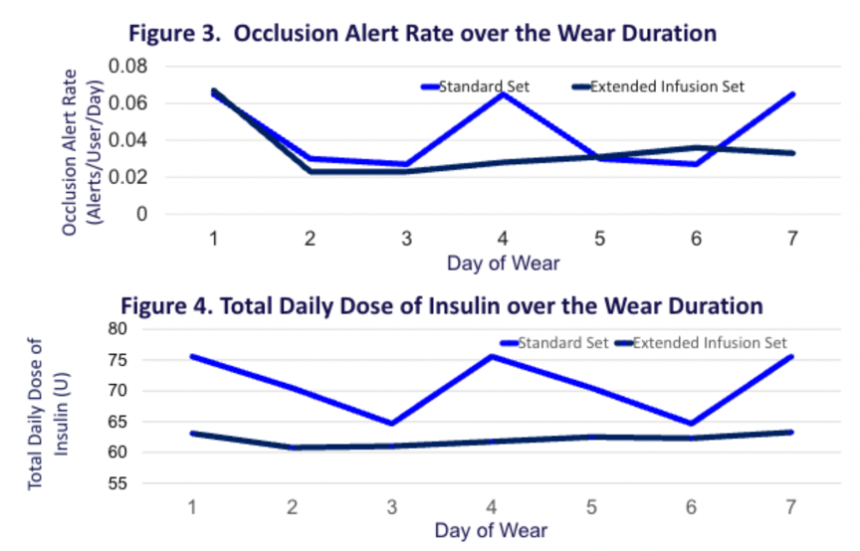
29. ROUTE-T1D pilot trial demonstrated feasibility and acceptability of a CGM behavioral intervention among youth from backgrounds historically excluded from clinical T1D research
Ms. Emma Straton (Children’s National Hospital) presented results from the ROUTE-T1D pilot trial (n=60) evaluating the feasibility and acceptability of a diabetes educator-led behavioral intervention (R01DK121316, PI Randi Streisand, PhD), with an aim of improving CGM wear-time and glycemic outcomes, in racially and socioeconomically diverse youth (aged 10-15 years old) with T1D. The Research on Optimizing the Use of Technology with Education (ROUTE-T1D) trial is a pilot behavioral intervention designed to promote optimal CGM use among racially minoritized youth with T1D. Participants were randomized 1:1 to either “immediate” or “delayed” intervention. The immediate group received three monthly telehealth sessions as youth-caregiver dyads, with a focus on CGM troubleshooting reinforced by positive communication (see figure below). The delayed group continued usual care for six months before receiving the same intervention. Ms. Straton’s presentation included data from the primary outcomes timepoint at 6 months post-randomization which was prior to the delayed group receiving their intervention.
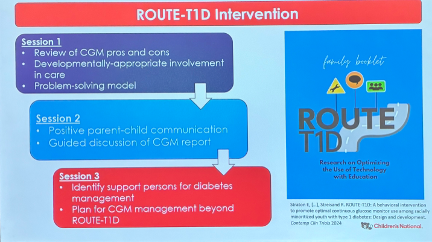
- Baseline characteristics. Participants had a mean age of 13 years and diabetes duration of five years. 58% were male, 54% Black, 27% Hispanic, 8% white, and 72% on public insurance. Mean A1c was 10.6%, and mean Time in Range was 30%. Mean CGM wear-time at baseline was low at 45%. 42% of participants used an insulin pump at baseline.
- Results. Descriptively, A1c dropped from 10.7% to 10.0% in the group receiving immediate intervention compared to 10.3% to 10.4% in the delayed intervention, and CGM wear-time increased from 48% to 59% in the immediate intervention and 44% to 50% in the delayed intervention. The intervention effect was not statistically significant. The interventions achieved 77% overall attendance. Ms. Straton concluded that the ROUTE-T1D pilot behavioral intervention was feasible and acceptable, and showed clinically significant improvements in A1c and CGM wear-time, specifically among youth who experience inequities in diabetes technology access and outcomes.
30. *NEW* SiBio continuous ketone monitor shows acceptable accuracy and detects benign, transient ketosis in T1D
Dr. Ahmad Haider (McGill University) presented findings on the accuracy of SiBionics’ SiBio KS1 continuous ketone monitoring (CKM) system and patterns of ketosis during ketogenic diets in adults with T1D using AID.
- Study design. The study evaluated: (i) CKM accuracy compared to capillary ketone readings; and (ii) real-world ketone dynamics across two types of six-day ketogenic diets – a very-low carbohydrate diet (<50 g/day) or intermittent 12-hour fasting. The study employed a crossover design, enrolling 14 adults with mean age of 36.5 years, baseline A1c 6.9%, and insulin dose 0.72 units/kg. Ketone levels were measured under normal conditions, pump suspension, and with SGLT2i for the T1D participants. One of the notable findings, comparing ketosis events under normal conditions versus during SGLT2i use, generated particular interest among ADA attendees. The analysis collected 181 paired data points comparing SiBio KS1 ketone sensor accuracy to FreeStyle Precision ß-ketone test strip readings. Accuracy was assessed on Day 7 of sensor wear during an eight-hour inpatient insulin suspension followed by capillary ketone testing.
- Results. The overall mean absolute difference (MAD) was 0.2 mmol/L. MARD was 24% for ketone values ≥0.6 mmol/L and 20% for values ≥1.0 mmol/L. Across the two outpatient dietary interventions, CKM readings were <0.6 mmol/L for 98% of the time and ≥0.6 mmol/L for 2.1% (30 minutes/day). There were 51 ketosis events >0.6 mmol/L and 7 events >1.0 mmol/L, each lasting over 30 minutes. The average event duration was 77 minutes, and all events were asymptomatic.
- Conclusions. Dr. Haider characterized this performance as “acceptable” accuracy. Moreover, he said the data suggests that transient, asymptomatic ketosis may occur in individuals with T1D, particularly during ketogenic or fasting regimens. Dr. Haider’s findings support the potential utilization of CKM in better understanding ketone dynamics and guiding safe dietary practices in T1D.
Big Picture
31. Fireside chat with ADA CEO Mr. Chuck Henderson and actor Mr. Anthony Anderson spotlights importance of T2D activism and advocacy
In a compelling and lively fireside chat with ADA CEO Mr. Chuck Henderson and award-winning actor and comedian Mr. Anthony Anderson, who was the first in his family to be diagnosed with T2D – now going on 25 years with the disease. Mr. Anderson shared his T2D journey, highlighting how losing his father to diabetes complications motivated him to take his own diabetes seriously and become vocal in his community about the condition. When his father was finally diagnosed, Mr. Anderson said that the condition had “ravaged his body” for so long that his quality of life was already severely impacted. Not wanting this to happen to others, he, especially as a Black man with a public platform, felt compelled to raise awareness about the importance of recognizing and managing diabetes risk, including partnering with pharmaceutical companies (e.g., Novo Nordisk’s Get Real About Diabetes campaign). As he pointed out, “We go to get our oil changed, but we don’t go to the doctor to get our health checked.”
- “Am I going live with this disease or do nothing and die from it?” Mr. Anderson admitted to being “somewhat cavalier” about his diabetes when he was initially diagnosed. However, after both his parents were also diagnosed, he became more serious and started making simple, but consistent, lifestyle changes.
- Mr. Anderson discussed how he has leveraged his public platform to reach others. As executive producer and star of the hit sitcom black-ish, he incorporated diabetes into the show’s plot (his character has T2D) to spotlight the realities of living with the disease. Knowing that he had half an hour every week (for eight years) on a major television network to impact many – and largely Black – viewers, he took great responsibility in prioritizing diabetes awareness.
- Mr. Anderson encouraged healthcare providers (HCPs) to have grace and be patient and understanding especially when connecting with patients from communities where trust has been broken in the past. He highlighted that some patients might be reluctant to change and unable to accept difficult truths. He urged HCPs to giving these patients information, reassuring them that “eventually a light bulb will go off.” He also emphasized the importance of forming partnerships, which can allow HCPs to better reach certain communities.
32. Quick Take: Role of patient advocacy and research organizations
It’s terrific to see so many nonprofits at work here at ADA’s Sci Sessions and to see the collaboration at work between T1D and T2D. From patient advocacy organizations to research foundations, there is much to celebrate across the diabetes ecosystem.
- As one example, Beyond Type 1 showed us the organization’s “Beginner’s Guide to Thriving with Diabetes,” which is available online here as well as in hard copies. We hope to see screening become an even bigger part of standards of care in the future, and Beyond Type 1 is primed to help contribute to this, given its storytelling prowess. We imagine the toolkit, now already available in English and Spanish, could serve the global community incredibly well if more languages are added. Over the last few years, work toward greater T1D screening has been building; while T2D screening seems more readily available it’s often not as accessible. We’d love to see even more work on this front all toward the end of not only T2D delay and prevention (just like T1D!) but also toward delay and prevention of complications for both types of diabetes.
- Breakthrough T1D celebrated the impact of strategic funding, evidence generation, and partnerships – by bringing together the right stakeholders, the organization hopes to expand therapy options and accelerate the timeline between research labs to clinical practice. Much of this progress has been driven by partnerships: Breakthrough T1D works through direct partnerships (through Industry Discovery Development Partnerships), academic grants (through Strategic Research Agreements), and drug donation (via BT1D-funded academic investigators).

33. New per-protocol analysis of BURST trial of Bigfoot Unity demonstrates sustained improvements in A1c and TIR 12 months after initiation
Dr. John Tillman (Abbott) presented a 12-month analysis of the BURST trial, a prospective, real-world study of the Bigfoot Unity connected pen cap system (1000-P). Bigfoot Unity integrates with FreeStyle Libre 2, leveraging CGM data to provide insulin dose recommendations and real-time alerts. As a reminder, Abbott announced its plans to acquire Bigfoot Biomedical, the original developer of Bigfoot Unity, in September 2023. This analysis evaluated 12-month glycemic outcomes among Bigfoot Unity users. We previously saw three-month outcomes at ADA 2023 and six-month results were published in Clinical Diabetes.
- Methods. This per-protocol analysis included all participants with: (i) available baseline and six-month A1c data; and (ii) ≥50% CGM data availability at six months, representing 125 of the 232 enrolled in the original study. Of these participants, 100 also had 12-month data. The analysis also evaluated a larger cohort with any A1c or GMI data available after three months to determine drop-out bias (n=176).
- Demographics. The cohort’s median age was 58 years, and nearly two-thirds (63%) were female. The vast majority of participants had T2D (86%). Most participants were non-Hispanic white (58%), and 31% were Black or African American. Over half (54%) of the cohort had not previously used CGM, and nearly one-third (31%) had used CGM for at least one year.
- A1c improvements observed at three months were sustained at six months, nine months, and one year. A1c decreased 1.2% from 9.2% at baseline to 8.0% at three months (p<0.001), remaining stable for the duration of the study. Participants also achieved significant improvements in CGM metrics. Time in Range (TIR) increased 6.7 hours/day from 30% at baseline to 58% at six months, which was maintained at 12 months. This was driven by a corresponding 6.7 hours/day reduction in Time above Range (TAR) from 69% at baseline to 41% at six months, which was also sustained at one year. While Time below Range (TBR) data was unavailable at baseline, TBR remained low at both six months (1.0%) and 12 months (1.1%).
- For patient-reported outcomes (PROs), participants reported significant decreases in diabetes distress (measured by the Diabetes Distress Scale). Participants also experienced increased confidence in responding to hypoglycemia (measured by the Hypoglycemia Confidence Scale).
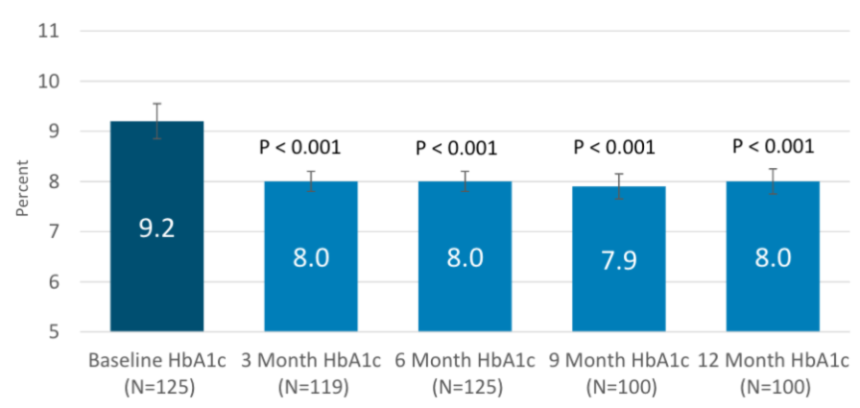
- A1c outcomes tended to be similar regardless of demographic information. Outcomes were relatively similar regardless of: (i) age; (ii) race/ethnicity; (iii) income level; (iv) education level; and (v) prior CGM use. Individuals who were: (i) living in a rural setting; (ii) with shorter diabetes duration (i.e., ≤10 years); or (iii) experiencing higher baseline A1c tended to achieve greater improvements in A1c; however, each of these groups had higher A1c at baseline than their comparators, which may partly explain these discrepancies.
- Glycemic and PROs were similar in the larger cohort compared to the per-protocol cohort. Additionally, when comparing the per-protocol T1D subgroup (n=17) to the per-protocol T2D subgroup (n=108), TBR appeared to be higher among those with T1D (2.0% vs. 0.8%). TAR was also higher among people with T1D (53% vs. 39%). However, the authors noted that the small sample size of people with T1D limits interpretation.
- Bigfoot Unity’s safety profile was reassuring. Six severe hypoglycemia events across four participants were observed in the per-protocol cohort, representing an incidence of 4.5 events per 100 person-years. No DKA or severe hyperglycemia was observed in the per-protocol cohort, compared to eight events across six participants in the expanded cohort. No events were attributed to Bigfoot Unity.
34. Abbott hosts “Above the Bias” fireside chat on diabetes stigma
At Abbott’s busy exhibit hall booth, Dr. Susan Guzman (Behavioral Diabetes Institute, San Diego) moderated a fireside chat on diabetes stigma, featuring patient advocates Ms. Amy Flores and Ms. Jeniese Hosey, who shared moving personal experiences navigating judgment, being dismissed in conversations, and disengagement in care. As a reminder, Abbott launched its enormously successful “Above the Bias” initiative in February, which aims to raise awareness of diabetes stigma and its real-world consequences. The company developed a one-minute film for the initiative, now with 3.1 million views on the company’s US YouTube channel and over 300 million views across all of its various global platforms, that portrays the burden of diabetes through the eyes of a person with diabetes to foster empathy and reduce various stigma associated with the disease. Alongside the initiative, Abbott conducted a survey of over 2,600 people with diabetes over six countries, which found that 40% of respondents had delayed or avoided medical visits due to fear of stigma and judgment.
- Both panelists described moments when stigma from healthcare professionals, peers, or colleagues led to shame, silence, and reluctance to seek care, highlighting the emotional and behavioral toll of diabetes stigma.
- Dr. Guzman and the panelists called on healthcare professionals to practice empathetic, individualized care. They emphasized the need to examine personal biases, listen without judgement, and create respectful, inclusive environments that reduce stigma and support long-term engagement in diabetes care.

35. Richard R. Rubin Award Lecture: Dr. Edwin Fisher on prioritizing social context and peer support in diabetes care
Dr. Edwin Fisher (UNC), recipient of the 2025 Richard R. Rubin Award, delivered an inspiring talk on the transformative role of behavioral science, social context, and peer support in diabetes care. He dedicated the lecture to the late Dr. Richard Rubin and his longtime collaborator Dr. Mark Peyrot, celebrating their shared commitment to viewing diabetes not as something to “control,” but as a complex, lifelong condition requiring sustained management.
- Dr. Fisher’s believed that family and interpersonal dynamics directly impact diabetes outcomes. Studies in adolescents with T1D revealed that low-conflict, collaborative family environments were associated with better outcomes. A career-defining moment came in 1982 while evaluating a self-management program – the results of which solidified his belief that peer-driven environments foster confidence, agency, and sustainable change.
- Global initiatives have demonstrated the power of peer support to improve outcomes across diverse populations. Through Peers for Progress and other global initiatives, Dr. Fisher implemented peer-based interventions in Kerala, India, reducing diabetes progression and cardiovascular risk. In Chicago, a collaboration with Alivio Medical Center provided peer support to over 3,200 Latino adults with T2D, driving significant A1c reductions. Globally, Dr. Fisher emphasized the importance of tailoring peer support to local norms and household decision-making structures.
- Dr. Fisher’s research consistently reinforced that ongoing, personalized support is more effective than standardized education alone. Patients with greater exposure to peer support and community resources showed superior glycemic outcomes, pointing to the need for emotionally attuned, sustainable care systems.
- In closing, he shared his key lesson: prioritize flexibility, emotional care, and human connection over rigid practices. Dr. Fisher argued for a model that respects cultural differences, while promoting accessible, adaptive interventions. Peer support, he concluded, is not a supplement to care, but rather a core element of successful long-term diabetes management. His powerful remarks were greeted with a standing ovation, a fitting tribute to a career that has shaped global diabetes care through insight and relentless advocacy.
--by Riya Chatterjee, Kayla Mathieu, Elizabeth Rose, Jeremy Alkire, Nour Khachemoune, Kat Moon, Elaine Young, Esther Min, Andrew Goyette, Monica Oxenreiter, and Kelly Close
[1] Although this isn’t “technical” remission, since they are taking medicine, we imagine participants would have passed the OGTT (because the A1c drop is secondary to weight loss).
[2] Or maximally tolerated dose.